-
Call Now
1800-102-2727
Chemical Properties – Chemical Properties of p- Block Elements, Practice Problems and FAQ
p-block elements are the ones in which the very last electron occupies the outermost p-orbital. The p-block elements are divided into six groups in the periodic table. p-block elements, including Group 13 to 18 elements. The groups are headed by helium, boron, carbon, nitrogen, oxygen, and fluorine.
On this concept page, we will get to know more about the chemical properties of group 13 to 18 elements in detail.
TABLE OF CONTENTS
- Chemical properties of Group 13 Elements
- Chemical properties of Group 14 Elements
- Chemical properties of Group 15 Elements
- Chemical properties of Group 16 Elements
- Chemical properties of Group 17 Elements
- Chemical properties of Group 18 Elements
- Practice Problems
- Frequently Asked Questions – FAQ
Chemical properties of Group 13 Elements
The property of an element that is seen during a chemical reaction is referred to as an element's chemical properties. Additionally, it establishes how it will interact with other elements.
Group 13 consists of elements boron, aluminium, gallium, indium and thallium. The following are some common chemical properties of group 13 elements.
- Reactivity Towards Air
- Boron is unreactive in its crystalline nature.
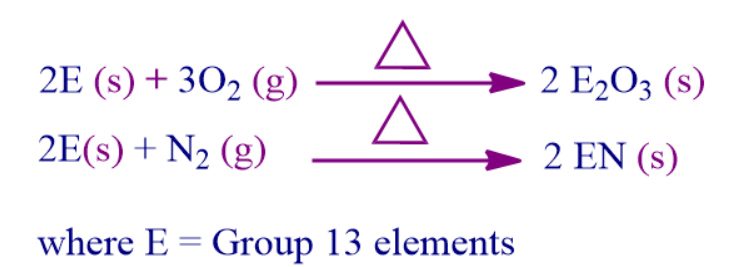
- Aluminium shields itself from further damage by forming a very thin oxide coating on the surface. When heated in air, amorphous boron and aluminium metal transform into B2O3 and Al2O3, respectively.
- At high temperatures, group 13 elements produce nitrides with dinitrogen.
- Air has no impact on gallium or indium. On the surface of thallium, an oxide layer develops. Thallium is kept intact by dipping it in oil or applying vaseline on it.
- Reactivity Towards Acids and Alkalis
- Even at room temperature, boron does not react with acids and alkalies; nevertheless, aluminium dissolves in both and exhibits amphoteric properties.
- In dilute HCl, aluminium dissolves and releases dihydrogen.
- As concentrated HNO3 is an oxidising agent, it forms a protective oxide layer on the surface of aluminium and makes the metal passive. The amphoteric aluminium also dissolves in aqueous NaOH, releasing hydrogen and producing aluminates.
- Other elements also react with dilute mineral acids.
Where M is a group 13 element.
- Reactivity Towards Water
- Boron does not react with water or steam but reacts with red heat steam.
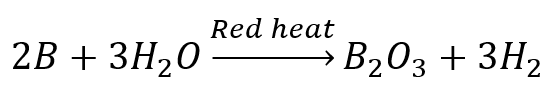
- Without the oxide layer, aluminium decomposes in cold water.
![]()
- Gallium and indium react with water only in the presence of oxygen, and thallium forms hydroxide in moist air.
![]()
- Reactivity Towards Halogens
Trihalides are generated when group 13 elements interact with halogens (except TlI3).
Chemical properties of Group 14 Elements
Group 14 consist of the elements carbon, silicon, germanium, tin and lead. Here are some common chemical properties of group 14 elements.
- Reactivity Towards Oxygen
When heated in oxygen, group 14 elements form oxides of formula MO and MO2. These are the two basic forms of oxides. Only at high temperatures does SiO exist. Higher oxidation state elements often have more acidic oxides than elements with lower oxidation states. While SnO2 and PbO2 are amphoteric in nature, the dioxides CO2, SiO2, and GeO2 are acidic. Monoxides include CO, which is neutral, GeO, which is clearly acidic, SnO, and PbO, which are amphoteric.
- Reactivity Towards Water
Water has no effect on germanium, silicon, or carbon until red heating. Tin decomposes water in its steam form to produce dihydrogen gas and dioxide. Water has no effect on lead, most likely due to the creation of a protective oxide covering.

- Reactivity Towards Halogens
These substances can combine to create halides with the formulas MX2 and MX4 (where X is F, Cl, Br, or I). All elements, with the exception of carbon, directly form halides when the conditions are favourable. The majority of MX4 are covalent in nature. SnF4 and PbF4, which are ionic in nature, are exceptions. There is no existence of PbI4.
- Reactivity Towards Acids and Alkalis
- Carbon and silicon are not impacted by non-oxidising acids.
- Dilute HCl is not destructive to germanium. However, trichlorogermane is produced when metal is heated in a stream of HCl gas.
- In dilute HCl, tin dissolves slowly, but quickly in concentrated HCl.
- Additionally, lead dissolves in dilute HCl and concentrated HCl. It dissolves lead, generating chloroplumbous acid; however, the process eventually ceases due to PbCl2 deposition.
- Carbon remains unaffected by alkalis. Cold aqueous NaOH interact slowly with silicon, while a warm solution reacts rapidly to produce a silicate solution.
- Cold alkalis attack tin and lead slowly, whereas hot alkalis attack them quickly, producing stannates and plumbates. Tina and lead are hence relatively amphoteric in nature.
Chemical properties of Group 15 Elements
Group 15 consist of elements nitrogen, phosphorus, arsenic, antimony and bismuth. Here are some common chemical properties of group 15 elements.
- Reactivity Towards Hydrogen
Group 15 elements all produce hydrides of type EH3; Where E=N, P, As, Sb,Bi.
From NH3 to BiH3, the stability of hydrides declines, and therefore reducing nature of the hydrides increases. Ammonia is merely a weak reducing agent, whereas the strongest reducing agent among group 15 hydrides is BiH3.
Basicity of hydrides follows the order:
- Reactivity Towards Oxygen
- These elements all combine to generate the oxides E2O3 and E2O5, where Where E=N, P, As, Sb,Bi.
- The oxide of these elements is more acidic in its higher oxidation state than in its lower oxidation state.
- Their acidic nature declines down the group. Arsenic and antimony oxides are amphoteric, while bismuth oxides are primarily basic. Nitrogen and phosphorus oxides of type E2O3 are completely acidic.
- Reactivity Towards Halogens
- These substances combine to generate the EX3 and EX5 series of halides, where E=N,P,As,Sb,Bi
- Due to the non-availability of d orbitals in nitrogen's valence shell, pentahalides cannot be formed by nitrogen.
- With the exception of nitrogen, the trihalides of all of these elements are stable.
- Only NF3 is recognised as being stable when it comes to nitrogen. With the exception of BiF3, trihalides are primarily covalent in character.
- Reactivity Towards Metals
All of these group 15 elements are reactive with metals, forming binary compounds with a -3 oxidation state. For example, Ca3N2 (calcium nitride), Ca3P2 (calcium phosphide), Na3As2 (sodium arsenide), Zn3Sb2 (zinc antimonide), and Mg3Bi2 (magnesium bismuthide).
Chemical properties of Group 16 Elements
Group 16 consists of elements oxygen, sulphur, selenium, tellurium and polonium. Here are some common chemical properties of group 16 elements.
- Reactivity Towards Hydrogen
- Group 16 consists of all elements that may form hydrides of the type H2E (E = O, S, Se, Te, and Po).
- Their level of acidity rises from H2O to H2Te . The decrease in bond enthalpy for the breakdown of the H-E bond down the group can be used to explain the increase in acidic character.
- The thermal stability of hydrides from H2O to H2Po likewise diminishes as the enthalpy for the breakdown of the H-E bond lowers along the group. Except for water, all hydrides have reducing properties, and these properties are stronger as you go from H2S to H2Te.
- Reactivity Towards Halogens
- Halides of the types EX6, EX4, and EX2 are frequently formed by Group 16 elements, where E is a member of the group 16 and X is a halogen.
- The stability of group 16 halides is F->Cl->Br->I-. Hexafluorides are the only stable halides among the hexahalides.
- Hexafluorides are all gaseous substances. Their shape is octahedral. For steric reasons, sulphur hexafluoride, also known as SF6, is very stable.
- Reactivity Towards Oxygen
- When E is S, Se, Te, or Po, all of these elements combine with oxygen to generate oxides of the EO2 and EO3 classes.
- While selenium dioxide (SeO2) is a solid, ozone (O3) and sulphur dioxide (SO2) are gases.
- From SO2 to TeO2, the reducing characteristic of dioxide declines; SO2 is a reducing agent, whereas TeO2 is an oxidising one. Sulphur, selenium, and tellurium also produce EO3 type oxides (SO3, SeO3, and TeO3) in addition to EO2 type oxides. The nature of both types of oxides is acidic.
Chemical properties of Group 17 Elements
Group 17 consists of elements fluorine, chlorine, bromine, iodine and astatine. Here are some common chemical properties of group 17 elements.
- Reactivity Towards Hydrogen
Iodine has a lower affinity for hydrogen than fluorine does, although they all react with hydrogen to produce hydrogen halides.
- Reactivity Towards Oxygen
The majority of the oxides that halogens and oxygen may create are unstable in nature. The two oxides formed by fluorine are OF2 and O2F2 . At 298 K, however, only OF2 is thermally stable.
Considering that fluorine has a greater electronegativity than oxygen, these oxides are actually oxygen fluorides. Both are powerful fluorinating agents.
Iodine, bromine, and chlorine all combine to produce oxides, with oxidation states ranging from +1 to +7.
- Reactivity Towards Metals
Metal halides are generated when halogens and metals react. For instance, when bromine interacts with magnesium, magnesium bromide is produced.
- Reactivity of Halogens Towards Other Halogens
XX',XX'3 ,XX'5 ,XX'7 are examples of the interhalogens that are created when halogens interact, where X is a larger size halogen and X' is a smaller size halogen.

Chemical properties of Group 18 Elements
Group 18 consists of elements helium, neon, argon, krypton, xenon and radon. Here are some common chemical properties of group 18 elements. Noble gases are often the least reactive. The following explanations are given as to why they are inert to chemical reactivity:
- All noble gases have a fully filled ns2np6 electronic configuration in their valence shells, with the exception of helium (1s2).
- They have a high enthalpy of ionisation and a higher positive electron gain enthalpy.
There are fewer krypton compounds. Only the difluoride (KrF2) has undergone extensive research. Radon compounds have not been isolated; rather, radiotracer technology has only been used to identify them, such as RnF2. As of yet, no real compounds of Ar, Ne, or He are discovered.
Related Videos
P-Block Elements Group 13 and 14 Class 11 Chemistry One Shot | NEET 2022 Chemistry Exam Preparation
p-Block Elements Group 15 and 16 Class 12 Chemistry One Shot | NEET 2022 Chemistry Exam Preparation
Practice Problems
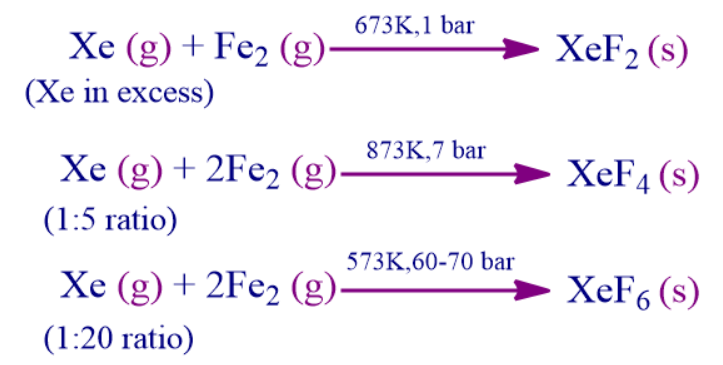
1. When Xe reacts with fluorine in the ratio of 1:5, then the compound formed is _____________.
a. XeF2
b. XeF4
c. XeF6
d. XeF3
Answer: B
Solution: When Xe reacts with fluorine in the ratio of 1:5, then the compound formed is XeF4. When it combines in the ratio of 1:20, then XeF6 will be formed. Xenon when taken in excess forms XeF2.
So, option B is the correct answer.
2. Which statement is responsible for the chemical inertness of nitrogen?
a. The triple bond between the two nitrogen atoms is extremely stable.
b. Nitrogen exists in free as well as combined forms in nature.
c. The dissociation energy of the triple bond present in nitrogen is low.
d. The maximum covalency of a nitrogen atom is four
Answer: A
Solution: Under normal circumstances, nitrogen has no chemical properties. It may be found freely in the atmosphere as a result. The exceptional stability of the triple bond formed by the two nitrogen atoms is what gives the object its chemical inertness. The bond has a high dissociation energy of 225 kcal mol-1.
So, option A is the correct answer.
3. Group 14 halides are generally covalent in nature except __________ and ____________.
a. GeF4 and SiF4
b. PbBr4 and PbI4
c. SnF4 and PbF4
d. CF4 and CCl4
Answer: C
Solution: The majority of MX4, where M is any group 14 element and X is any halogens, are covalent in nature. SnF4 and PbF4, which are ionic in nature, are exceptions.
So, option C is the correct answer.
4. When lead reacts with concentrated nitric acid, ____________ gas is formed.
a. NO
b. NO2
c. N2O
d. N2O5
Answer: B
Solution: When lead reacts with concentrated nitric acid, NO2 gas is formed. When it reacts with dilute nitric acids, NO is formed.
So, option B is the correct answer.
Frequently Asked Questions – FAQ
1. Noble gases are inert in nature. Do they make compounds?
Answer: Yes, noble gases do make compounds irrespective of their inertness. Some of the major examples of noble gas compounds are XeF4 , XeF2 , XeF6 , XeO3 etc. They usually form compounds with fluorine and oxygen.
2. What function do nitrogen compounds serve?
Answer: For all life, nitrogen is a vitally critical element. Numerous cells and activities, including those involving amino acids, proteins, and even our DNA, depend on it. Additionally, it is necessary for plants to produce chlorophyll, which is required in photosynthesis to produce their food.
3. How is oxygen chemically produced?
Answer: In a lab, manganese(IV) oxide and hydrogen peroxide are added to a conical flask to create oxygen. A gas jar that is upside-down and filled with water is used to collect the created gas. The oxygen pulls the water out of the gas jar as it gathers at its top.
4. What is coal's chemical nature?
Answer: Coal is mostly composed of carbon with varying proportions of additional elements, primarily hydrogen, sulphur, oxygen, and nitrogen. Dead plant material decomposes into peat, which is then transformed into coal by heat and pressure from deep burial over millions of years.


