-
Call Now
1800-102-2727
Charge of Electron – Theory, Experiment, Apparatus, Procedure, Calculations, Conclusions, Practice Problems and FAQ
Bricks is the repeating unit of walls. We know that everything around us consists of matter, and that matter is made up of atoms. Atoms, in turn, are composed of electrons, protons, and neutrons. During the calculation of atomic mass, the mass of an electron is neglected and for numerical simplicity, we treat the mass of a neutron as equal to the mass of a proton. So, we can say the basic unit of mass is the mass of a single proton. In the same way, the smallest unit of charge is the charge of an electron. All charges present are an integral multiple of the charge of an electron.
On this concept page, we will get to know the experiment by which the charge of electrons was calculated in detail!
TABLE OF CONTENTS
- Experiment to Determine the Charge of Electron
- Millikan’s Oil Drop Experiment - Principle
- Millikan’s Oil Drop Experiment - Theory
- Millikan’s Oil Drop Experiment - Experiment
- Millikan’s Oil Drop Experiment - Procedure
- Milliken’s Oil Drop Experiment - Calculation
- Millikan’s Oil Drop Experiment - Mass of Electron
- Practice Problems
- Frequently Asked Questions – FAQ
Experiment to Determine the Charge of Electron
To ascertain the charge of an electron, Robert A. Millikan and Harvey Fletcher performed the Millikan's Oil Drop Experiment in 1909. This experiment was conducted at the Ryerson Physical Laboratory, which is part of the University of Chicago. This experiment has been shown to be remarkable for identifying the quantum nature of charge.
- An easy way to gauge the little electric charge that many oil mist droplets have was developed by American physicist Robert A. Millikan.
- The product of the charge and the electric field determines the force acting on any electric charge in the field.
- By measuring the amount of electric force and the size of the electric field on the tiny charge of a solitary oil droplet, Millikan was able to ascertain the charge's magnitude.
- This astounding experiment earned Robert Millikan the 1923 Physics Nobel Prize.
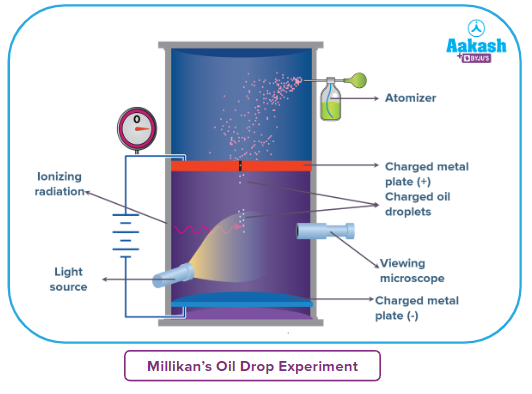
Millikan’s Oil Drop Experiment - Principle
By enabling charged little oil droplets to travel through a hole, Millikan produced an electric field. By varying the electric field's strength, which was always represented as an integral value of 'e,' the charge over an oil droplet could be calculated.
- A spherical drop of oil falling through a viscous medium, such as air, will quickly approach a constant velocity.
- When the drop reaches this state of equilibrium, other forces acting on it, such as gravity, air buoyant forces, electrical forces, and so on, balance the viscous force.
- In this experiment, an ionisation source is used to introduce an electrical force of varying size and alter the speed of the falling drop.
- You will use this experiment to determine the quantum nature of the charge. For this outstanding experiment, Robert Millikan received the Nobel Prize in Physics in 1923.
- By keeping an eye on the oil drop's velocity under various circumstances, it is possible to calculate the amount of charge on the drop.
- If the charge on the drop is an integer multiple of the basic unit of charge, the quantization of charge can be verified (the electron).
Millikan’s Oil Drop Experiment - Theory
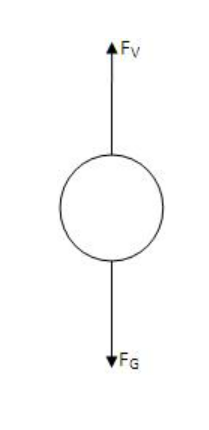
The oil drops are initially permitted to fall between the plates while there isn't an electric field. Gravity causes them to accelerate initially, but air resistance causes them to gradually slow down.
- The terminal velocity 'v1' is estimated in the absence of an electric field as
v1=
where 'l1' represents the oil drop's distance travelled and 't1' represents the time elapsed. The drag force exerted on the descent is calculated using Stokes' law, which is given as
- The apparent weight (Actual weight - Thrust) of a perfectly spherical body is given by
FG =
- The total force imparted on the oil drop must be zero since it is not accelerating at terminal velocity. Hence,
r2=
Where,
g - Acceleration due to gravity
ρ - Density of liquid
r - Oil droplet’s radius
ρair - Density of air
η - Air viscosity
v1 -Terminal velocity
- A field is now produced in the bottom chamber thanks to the voltage source. A likely-looking drop is identified and held in the centre of the field of view by altering the voltage. If the electric forces Fe balance the gravity force, FG, the drop will stay in the air.
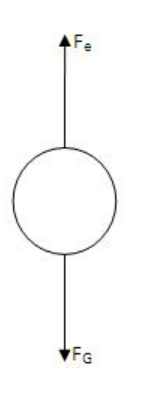
Then, Fe=FG
=mg
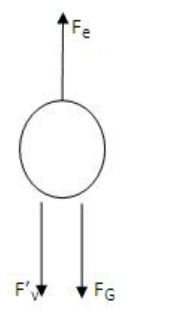
- V represents the balancing potential, while d represents the distance between the plates. If the applied electric force, Fe, is more than the downward force, some of the droplets (the charged ones) will begin to ascend. The viscous and gravitational forces now operate downhill, whereas the electric force now acts upward. The terminal velocity v2 is calculated in the following way:
v2=
where l2 is the oil drop's distance travelled and t2 is the time taken. is now the entire force acting on the drop.
- So, , where Fv' is the new viscous force under the action of the applied electric field.
- Millikan performed the experiment several times, each time increasing the intensity of the X-rays used to ionise the air. The number of electrons attracted to the oil drop varied as a result. Then he experimented with several values for q, which turned out to be .
Millikan’s Oil Drop Experiment - Experiment
The equipment for the experiment was conceived and manufactured by Millikan and Fletcher. It consists of two metal plates separated by an insulated rod. In addition, the plate has four holes, three of which enable light to pass through and the fourth of which permits viewing via the microscope.
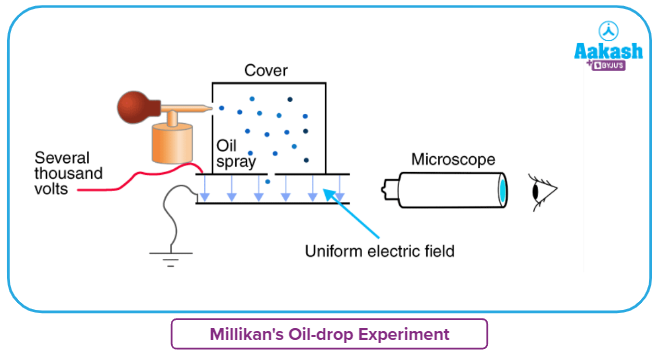
Ordinary oil was not used in the experiment because it would evaporate owing to the light's heat, possibly leading to Millikan's Oil Drop Experiment error. As a consequence, a low vapour pressure oil that is typically used in vacuum equipment was used.
Millikan’s Oil Drop Experiment - Procedure
- After passing through the atomizer in the form of minute droplets, the oil is atomized. The droplets travel through the perforations in the upper plate of the device.
- When the downward movements of oil droplets are identified using a microscope, the terminal velocity of the droplets is calculated.
- The air within the chamber is ionised by sending an X-ray beam across it. These oil droplets get their electrical charge from collisions with gaseous ions generated by air ionisation.
- Between the two plates, an electric field is formed, which can affect the mobility of charged oil droplets.
- Gravity pulls the oil below, while the electric field pulls the charge upward. The electric field intensity is adjusted such that the oil droplet is in a gravity-balanced position.
- The charge over the droplet, which is dictated by the intensity of the electric field and the mass of the droplet, is estimated at equilibrium.
Millikan’s Oil Drop Experiment - Calculation
Where
E= Electric Field
q= charge on electron
Where
m= Mass of Oil droplet
g= Acceleration due to gravity
The method for detecting electron charge devised by Millikan may thus be observed. According to Millikan, all of the drops included charges that were multiples.
- The charge of any oil droplet is always an integral value of electron of . As a result of Millikan's Oil Drop Experiment, the charge is quantized, meaning that the charge on each particle will always be an integral multiple of e-.
- The values of the electric charge (q) on individual oil droplets are always an integral multiple of the electrical charge (e) when this approach is used repeatedly, that is, q=ne, where n=1,2,3...
Millikan’s Oil Drop Experiment – Mass of Electron
From the cathode ray discharge tube experiment conducted by J.J Thomson, ratio for electron was calculated ( = 1.75882010-31C kg-1).
So, the mass of the electron (me) was determined by combining these results with Thomson’s value of the ratio.
Where me is the electron's mass in kilograms and e is the charge magnitude on the electron in Coulombs (C). The charge of an electron is negative because electrons are negatively charged.
Practice Problems
Q1. Calculate the charge on 10 moles of electrons.
A. -964720 C
B. -1.60210-19C
C. 964720 C
D. -96472 C
Answer: A
Solution: The charge on 1 electron =
Charge on 1 mole of electrons =
Charge on 10 moles of electrons
So, option A is the correct answer.
Q2. During an oil drop experiment, the following charges (in units) were discovered on a sequence of oil droplets: , , , , and . Compute the charge on an electron (in the unit specified).
Answer: C
Solution: Magnitude of electrical charge, q, on the droplets is always an integral multiple of the electrical charge, e, that is, q=ne, where n=1,2,3....
q1= 7.5 × 10-18 unit = 3 × 2.5 × 10-18 unit
q2= 5 × 10-18 unit = 2 × 2.5 × 10-18 unit
q3= 10 × 10-18 unit = 4 × 2.5 × 10-18 unit
q4= 12.5 × 10-18 unit = 5 × 2.5 × 10-18 unit
q4= 15 × 10-18 unit = 6 × 2.5 × 10-18 unit
So, e = 2.5 × 10-18
So, option C is the correct answer.
Q3. The oil drop device for Milikan's experiment was built by:
A. Thomson and Millikan
B. Millikan and Fletcher
C. Millikan and Boiler
D. Millikan and Dalton
Answer: B
Solution: Robert A. Millikan and Harvey Fletcher built the equipment and performed the Millikan's oil drop experiment to measure an electron's charge in 1909.
So, option B is the correct answer.
Q4. The charge on an oil drop was discovered to be during Millikan's oil drop experiment. The number of electrons in a single drop of oil is
A. 300
B. 600
C. 900
D. 120
Answer: A
Solution: Charge on 1 electron
Number of electrons = = 300
So, option A is the correct answer.
Q5. The oil drop experiment of Millikan is used to determine the charge on a:
A. Proton
B. Neutron
C. Electron
D. All of these
Answer: C
Solution: Robert A. Millikan and Harvey Fletcher built the equipment and performed the Millikan's oil drop experiment to measure an electron's charge in 1909.
So, option C is the correct answer.
Frequently Asked Questions – FAQ
Q1. In the Millikan oil-drop experiment, how can they assure that each oil drop has only one electron?
Answer: There was an atomizer in the first part of the experiment that could separate the drips to the lowest feasible size. Despite the fact that it did not create any oil molecules. Everything is based on the likelihood that at least one drop will contain a single ion. The initial electric fields are kept at their lowest powers at the start of the experiment. The voltage is gradually raised until a few drips begin to rise. Different terminal velocities may then be achieved by varying voltages, and e/m can be determined. There's also the possibility that e/m will turn out to be multiples of each other.
Q2. What is the charge of an electron electrostatic unit?
Answer: esu is the unit of charge in the CGS unit. The magnitude of the charge of electron = 4.8 × 10-10esu. Esu is substantially smaller than coulombs. . The speed of light in decimeters per second is really mathematically equal to the ratio of esu to Coulomb.
Q3. Why did Milliken not use water in place of oil?
Answer: Millikan has experimented with numerous droplet forms. That was their first try, and J.J. Thomson had utilised water droplets in his previous tests. The tiny droplets, however, quickly evaporated due to the light source's heat in less than two seconds.
Mercury, glycerin, and oil were among the potential additional liquids that Millikan and Fletcher discussed. Fletcher created a rudimentary device after purchasing watch grease from the drugstore. It succeeded!
Q4. Why did some oil drops rise and fall quicker than others in Millikan's experiment?
Answer: When thrown from the same height in a vacuum, all items fall at the same speed. However, because mass and charge are not evenly distributed, when an electric field is applied, the downward acceleration of all the drops is the same, but the upward acceleration (due to the field) varies. This indicates that the accelerations of the drops vary significantly depending on the size of the electric field applied.


