-
Call Now
1800-102-2727
Cationic and Anionic Hydrolysis- Salts, Anionic Hydrolysis, Cationic Hydrolysis, Practice Problems and FAQs
You might have studied that acids and bases react to form a salt. You must expect,t all salts like sodium chloride made from the reaction of sodium hydroxide and hydrochloric acid to be neutral in nature.
Have you heard of ammonium nitrate? It is used as a fertiliser to improve plant growth and productivity.
It is one of the largest industrial manufactured chemicals.
. 
This, ammonium nitrate is formed by the reaction of the nitric acid and ammonium hydroxide base, This ammonium nitrate is not neutral, but an acidic salt.
But how is Ammonium Nitrate acidic in nature?
And do we have salt that can behave like a base? Yes, some salts exhibit basic properties in aqueous solutions too.
How can we identify the salt nature as acidic, neutral or basic from the reaction of an acid and a base?
Let us find out answers to all these questions.
In this article, we'll go over how these salts are made as well as some fascinating properties, particularly of salts that go through a process called hydrolysis. Let's take a closer look at them.
Table of Contents
- Salts
- Anionic Hydrolysis
- Cationic Hydrolysis
- Practice Problems
- Frequently Asked Questions
Salts
There are four major categories of salt that exist in nature or that we use in our daily lives. Baking soda, washing soda, and ammonium nitrate in fertilisers are some examples of these salts.
These four categories are
|
Acid |
Base |
Salt |
Examples |
Nature of the salt |
|
Strong Acid (SA) |
Strong Base (SB) |
Salt of SASB |
(HCl+NaOH) NaCl |
Neutral |
|
Strong Acid (SA) |
Weak Base (WB) |
Salt of SAWB |
(HCl+NH4OH) NH4Cl |
Acidic |
|
Weak Acid (WA) |
Strong Base (SB) |
Salt of WASB |
(CH3COOH+NaOH) CH3COONa |
Basic |
|
Weak Acid (WA) |
Weak Base (WB) |
Salt of WAWB |
(CH3COOH+NH4OH) CH3COONH4 |
, Neutral , Acidic , Basic |
In this topic, we will discuss how these salts formed and some interesting aspects of salts that undergo cationic and anionic hydrolysis.
First and foremost, we will go over the fundamental terms that will be used on this concept page.
- Hydrolysis: Hydrolysis is the process of breaking down compounds with water. It's fascinating that an ion from a weak electrolyte (a weak acid or a weak base) undergoes hydrolysis.
- Hydration: Hydration is the electrophilic addition reaction in which the original molecule is not cleaved. Water molecules are added to the substance during hydration. It’s amusing that an ion from a strong electrolyte (a strong acid or a strong base) undergoes hydration rather than hydrolysis.
Let us understand this concept more deeply by taking examples of four different categorised salts:
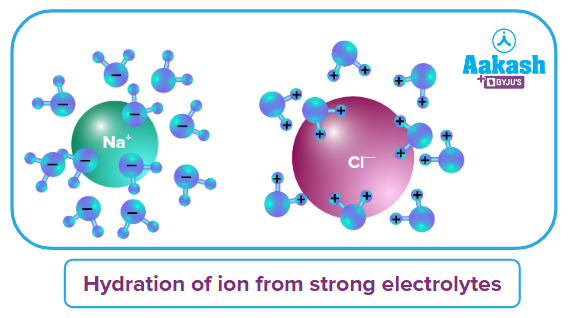
(i) Salt of a Strong acid and Strong Base (SASB): Let us take one example of Salt of SASB; NaCl.
It is a combination of strong acid (HCl) and strong base (NaOH). So, the ion Na+ comes from a strong base (NaOH) and Cl- comes from strong acid (HCl). As per the concept, these ions get hydrated rather than hydrolysed.
|
Salt |
Examples |
Hydration of |
Hydrolysis of |
|---|---|---|---|
|
Salt of SASB |
NaCl(HCl+NaOH) |
Na+ & Cl- |
Nil |
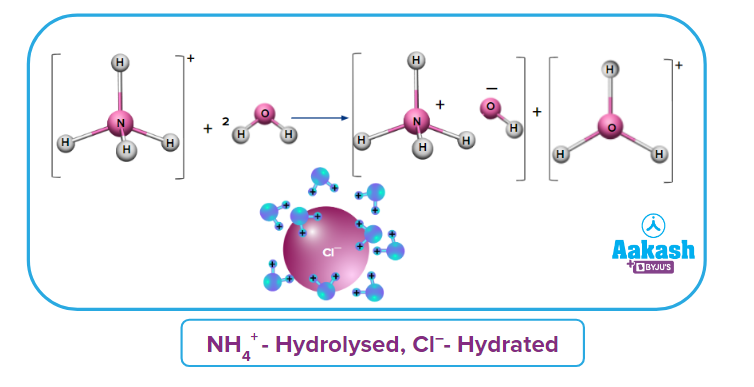
(ii) Salt of a Strong Acid and Weak Base (SAWB): Salt of SAWB; NH4Cl is a combination of strong acid (HCl) and weak base (NH4OH). So, the ion comes from a weak base (NH4OH) and Cl- comes from strong acid (HCl). As per the concept, ions get hydrolysed and Cl- get hydrated.
|
Salt |
Examples |
Hydration of |
Hydrolysis of |
|---|---|---|---|
|
Salt of SAWB |
NH4Cl(HCl+NH4OH) |
Cl- |
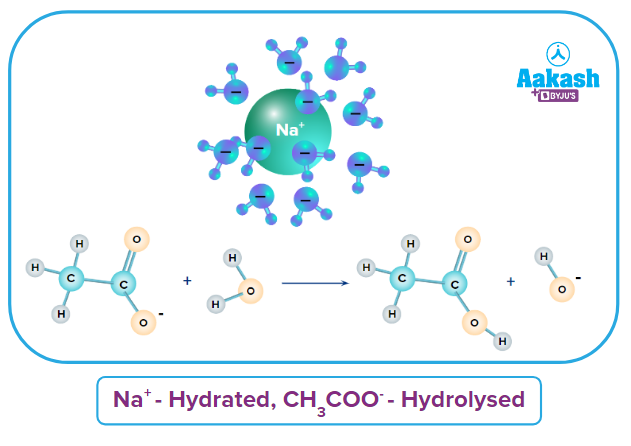
(iii) Salt of a Weak Acid and Strong Base (WASB): Salt of WASB; CH3COONa is a combination of weak acid (CH3COOH) and strong base (NaOH). So, the ion Na+ comes from a strong base (NaOH) and CH3COO- comes from weak acid (HCl). Hence, Na+ ions get hydrated and CH3COO- get hydrolysed.
|
Salt |
Examples |
Hydration of |
Hydrolysis of |
|---|---|---|---|
|
Salt of WASB |
CH3COONa(CH3COOH+NaOH) |
Na+ |
CH3COO- |
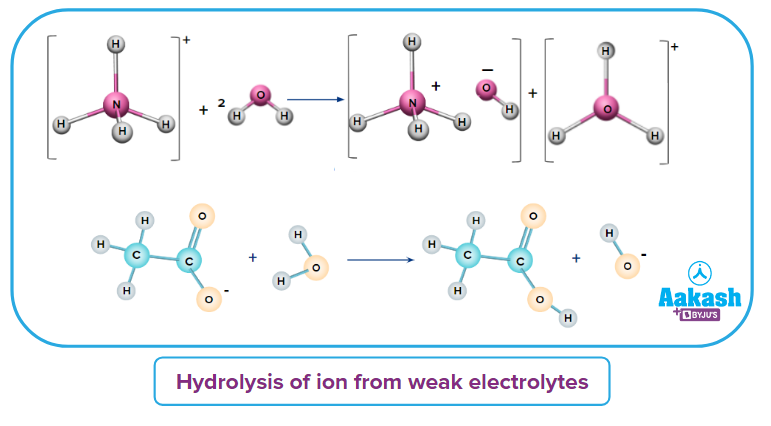
(iv) Salt of a Weak Acid and Weak Base(WAWB): CH3COONH4 (WAWB salt) is a combination of weak acid (CH3COOH) and weak base (NH4OH). So, the ion comes from a weak base (NH4OH) and Cl- comes from weak acid (CH3COOH). As per the concept, these ions get hydrolysed rather than hydrated.
|
Salt |
Examples |
Hydration of |
Hydrolysis of |
|---|---|---|---|
|
Salt of WAWB |
CH3COONH4(CH3COOH+NH4OH) |
Nil |
CH3COO-& |
Anionic Hydrolysis:
Anionic Hydrolysis occurs in the salt of WASB and WAWB when the anion of a weak acid is hydrolysed. Let us discuss some important points regarding Anionic Hydrolysis in the salt of WASB.
- Let's take the example of salt
As an ion CH3COO- comes from a weak acid, it is hydrolysed and ion Na+ comes from a strong base, it is hydrated.
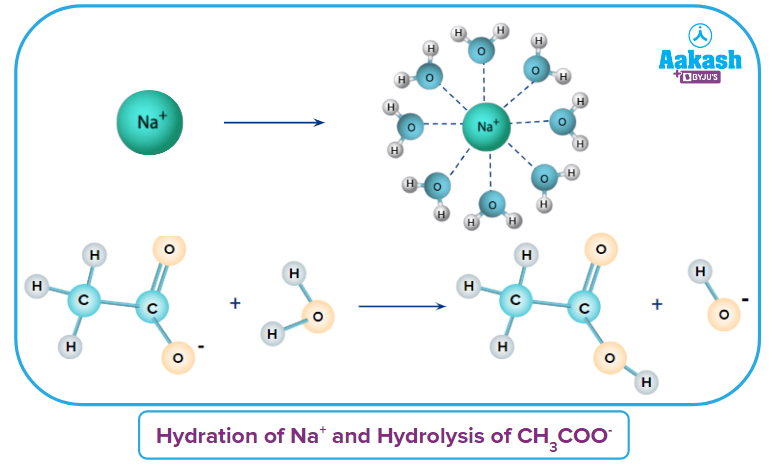
- When an anion from weak acid CH3COOH is considered
The anion from a weak acid will react with water to produce OH- ions.
- In nature, the solution becomes basic. As a result, the pH of the solution rises due to an increase in OH- ions.
Some more examples
|
Salt |
Acid |
Nature of acid |
Anion |
Hydrolysis |
|
NaCN (WASB) |
HCN |
Weak acid |
CN- |
yes |
|
KNO2(WASB) |
HNO2 |
Weak acid |
yes |
|
|
CH3COONa(WASB) |
CH3COOH |
Weak acid |
yes |
|
|
CaCO3(WASB) |
H2CO3 |
Weak acid |
yes |
Anionic Hydrolysis also occurs in the salt of WAWB when the anion of a weak acid is hydrolysed.Some examples are
|
Salt |
Acid |
Nature of acid |
Anion |
Hydrolysis |
|
FeS(WAWB) |
H2S |
Weak acid |
yes |
|
|
Zn(HCOO)2(WAWB) |
HCOOH |
Weak acid |
yes |
|
|
(C5H5NH)2CO3(WAWB) |
H2CO3 |
Weak acid |
yes |
Cationic Hydrolysis:
Cationic Hydrolysis occurs in the salt of SAWB and WAWB when the cation of a weak base is hydrolysed. Let us discuss some important points regarding Cationic Hydrolysis in the salt of SAWB.
- Let's take the example of salt
As an ion comes from a weak base, it is hydrolysed and ion Cl- comes from a strong acid, it is hydrated.
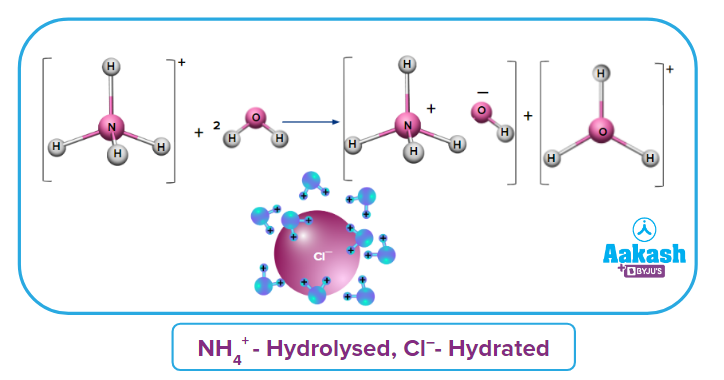
- When a cation from a weak base NH4OH is considered
.
The cation from a weak base will react with water to produce H+ or ions.
- In nature, the solution becomes acidic. As a result, the pH of the solution decreases due to an increase in H+ or ions.
Some more examples
|
Salt |
Base |
Nature of base |
Cation |
Hydrolysis |
|
ZnSO4(SAWB) |
Zn(OH)2 |
Weak base |
yes |
|
|
NH4Cl (SAWB) |
NH4OH |
Weak base |
yes |
|
|
FeCl2(SAWB) |
Fe(OH)2 |
Weak base |
yes |
|
|
(C5H5NH)2Cl(SAWB) |
C5H5NHOH |
Weak base |
yes |
Cationic Hydrolysis also occurs in the salt of WAWB when the cation of a weak base is hydrolysed.Some examples are
|
Salt |
Base |
Nature of base |
Cation |
Hydrolysis |
|
FeS(WAWB) |
Fe(OH)2 |
Weak base |
yes |
|
|
Zn(HCOO)2(WAWB) |
Zn(OH)2 |
Weak base |
yes |
|
|
(C5H5NH)2CO3(WAWB) |
C5H5NHOH |
Weak base |
yes |
Practice Problems
Q1. Comment on the type of hydrolysis and nature of Na2S salt when added to water.
A. Cationic Hydrolysis, Acidic salt
B. Cationic Hydrolysis, Basic salt
C. Anionic Hydrolysis, Acidic salt
D. Anionic Hydrolysis, Basic salt
Answer: (D)
Solution: Na2S is a combination of the weak acid and strong base.
|
Ions |
Origin |
Hydrolysis |
|
S2- |
Weak acid |
Yes, |
|
Na+ |
NaOH - Strong base |
No |
In this salt, upon reaction with water produce OH- ions. This will increase the concentration of OH- in in the solution and hence pH of solution increases. Being a mixture of a weak acid and a strong base, it is basic in nature.
There is the hydrolysis of anion from weak acid. Hence it is an Anionic Hydrolysis.
Hence, the correct answer is the option (D).
Q2. Predict Zn(CN)2 salt undergoes which type of hydrolysis ?
A. Cationic Hydrolysis
B. Anionic Hydrolysis
C. Both (A) and (B)
D. None of these
Answer: (C)
Solution: Zn(CN)2 salt is a combination of a weak acid and weak base.
|
Ions |
Origin |
Hydrolysis |
|---|---|---|
|
CN- |
HCN - Weak acid |
yes |
|
Zn2+ |
Zn(OH)2 - Weak base |
yes |
Being a mixture of a weak acid and weak base, both ions undergoes hydrolysis. There is the hydrolysis of anion from weak acid HCN and hydrolysis of cation from a weak base Zn(OH)2. Hence it undergoes Anionic as well as Cationic Hydrolysis.
Hence, the correct answer is the option (C).
Q3. Comment on the nature of table salt when added to a beaker full of water.
A. Acidic Salt
B. Basic Salt
C. Neutral Salt
D. None of these
Answer: (C)
Solution: The chemical formula for Table salt is NaCl.
|
Ions |
Origin |
Hydrolysis |
|
Cl- |
HCl- Strong acid |
No |
|
Na+ |
NaOH - Strong base |
No |
NaCl is a strong acid and strong base compound. The respective ions are from strong electrolytes, hence they should be hydrated rather than hydrolysed. There should be no excess of H+ or OH- present in hte solution. As a result, the pH of the solution should be close to 7 and the salt is neutral in nature.
Hence, the correct answer is the option (C).
Q4. Arrange the solution of salt NaCN, MgCl2 and FeSO4 in order of decreasing pH.
A. NaCN< MgCl2 <FeSO4
B. NaCN> MgCl2 >FeSO4
C. NaCN< MgCl2 >FeSO4
D. NaCN= MgCl2 <FeSO4
Answer: (B)
Solution: FeSO4 is a combination of the strong acid and weak base.
|
Ions |
Origin |
Hydrolysis |
|
Fe2+ |
Fe(OH)2 - Weak base |
Yes, |
|
H2SO4- Strong acid |
No |
In this salt, upon reaction with water produce H+ ions. This will increase the concentration of H+ in the solution and hence pH of solution decreases. Being a mixture of strong acid and weak base, it is acidic in nature.
NaCN is a combination of a weak acid and strong base.
|
Ions |
Origin |
Hydrolysis |
|
CN- |
HCN - Weak acid |
Yes, |
|
Na+ |
NaOH - Strong base |
No |
In this salt, upon reaction with water produce OH- ions. This will increase the concentration of OH- in in the solution and hence pH of solution increases. Being a mixture of a weak acid and a strong base, it is basic in nature.
|
Ions |
Origin |
Hydrolysis |
|---|---|---|
|
Cl- |
HCl - Strong acid |
No |
|
Mg2+ |
Mg(OH)2 - Strong base |
No |
MgCl2 is a strong acid and strong base compound. The respective ions are from strong electrolytes, hence they should be hydrated rather than hydrolysed. As a result, the pH of the solution should be close to 7 and the salt is neutral in nature.
Acidic salt - FeSO4 (lowest pH), Neutral Salt - MgCl2, Basic Salt - NaCN (Highest pH)
The correct order of pH is: NaCN> MgCl2 >FeSO4
Hence, the correct answer is the option (B).
Frequently Asked Questions
Q1. What is the nature of salt used to puff edible cakes?
Answer: We all love to eat yummy puffed cakes. But do you know, to puff the edible cakes, baking soda is used which contains salt NaHCO3. It is a mixture of a strong base (NaOH)and a weak acid (H2CO3). As a result, it is a basic salt.

Q2. Is washing soda an acid, a base, or a salt?
Answer: Washing soda (Na2CO3) which is used in laundry, is formed by the reaction of a strong base (NaOH) and a weak acid (H2CO3). As a result, it is a basic salt.
Q3. Why are strong acid and strong base ions hydrated rather than hydrolyzed?
Answer: Because strong acid and strong base ions are stable in ionic form and do not want to react further, they are hydrated rather than hydrolyzed.
Q4. What exactly are neutral salts?
Answer: Neutral salts are salts formed by the reaction of a strong acid and a strong base. An example: NaCl is a common example of a neutral salt.
Q5. How Ammonium nitrate salt is acidic in nature?
Answer: Ammonium Nitrate (NH4NO3) is a combination of a weak base (ammonium hydroxide) and strong acid (nitric acid). Hence it is an acidic salt.


