-
Call Now
1800-102-2727
Catalyst – Catalysts, Their Types, Theories on Catalysis, Enzymes, their Characteristics, Catalysts Used in Industries
Imagine you want to cross a sizable mountain to get to the other side. Two methods are used. The first is through what appears to be a pretty lengthy trail (the orange line). The alternate route is a shortcut through the tunnel.
In general, it would be impossible to tunnel through a mountain, but chemistry makes this possible.
Confused? I'll explain.
Consider a reaction where there is a large energy barrier between the reactants and products (like the orange pathway in the figure), meaning that a reactant must overcome this barrier in order to become a product. We, therefore, need more energy. The energy barrier of the reaction can be lowered by employing a material (tunnel) so that less energy is needed for the reactants to be transformed into the products.
This substance is referred to as a catalyst, which offers an alternative route to the reactant similar to the Imaginary Tunnel in a mountain.
On this concept page, we will get to know about catalysts, their types, enzymes, their characteristics and the catalysts used in industry.
TABLE OF CONTENTS
- Catalyst
- Catalyst – Types Based on Phase
- Catalyst – Types Based on the Effect on Rate of the Reaction
- Theories on Catalysis
- Zeolites
- Catalyst – Characteristics
- Enzymes
- Enzymes – Mechanisms
- Enzymes – Characteristics
- Catalysts Used in Industry
- Practice Problems
- Frequently Asked Questions – FAQ
Catalyst
Catalysts are substances that alter a pathway of reaction in order to vary the rate at which the reaction occurs.
Catalysis is the technique of speeding up a chemical reaction by adding materials that are not consumed during the reaction and are still present after it has finished.
The important points that should be kept in mind before discussing catalysts in detail are:
- A catalyst is frequently used to quicken or speed up a reaction.
- A catalyst is used as one of the reactants in a catalytic reaction in order to change the reaction's rate from that of an uncatalyzed reaction.
- With the reactant, the catalyst creates an intermediate that requires less activation energy, which subsequently breaks down to create the product.
- After the reaction is finished, the catalyst is renewed, sometimes in a different physical form.
Example: Formation of NO in presence of Pt(s) as a catalyst.

Catalyst – Types Based on Phase
Homogeneous Catalysts
When both the catalyst and the reactants are present in the same phase, the catalyst is referred to as a homogeneous catalyst. This type of catalysis is referred to as homogeneous catalysis.
Examples:
- Hydrolysis of ester, in an acidic medium. Here, acid acts as a catalyst.

- Oxidation of sulphur dioxide in the presence of NO(g) as catalyst.

- Hydrolysis of sugar in an acidic medium. Here, acid acts as a catalyst.
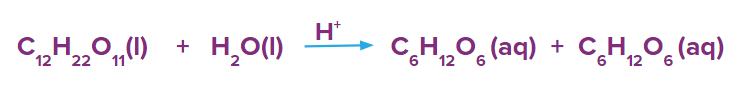
Heterogeneous Catalysts
When the catalyst and the reactants are present in different phases, the catalyst is referred to as a heterogeneous catalyst. This type of catalysis is referred to as heterogeneous catalysis.
Examples:
- Formation of SO3 in presence of Pt(s) as a catalyst.
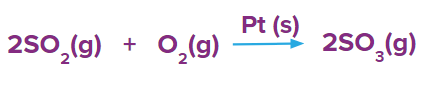
- Reduction of vegetable oil in presence of Ni(s) as a catalyst.
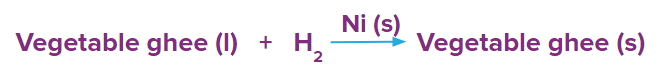
- Formation of ammonia in the presence of Fe(s) as a catalyst.

Catalyst – Types Based on the Effect on Rate of the Reaction
Positive Catalyst
Positive catalyst is the particular kind of catalyst used in the chemical reaction that reduces the activation energy and hence speeds up the reaction.
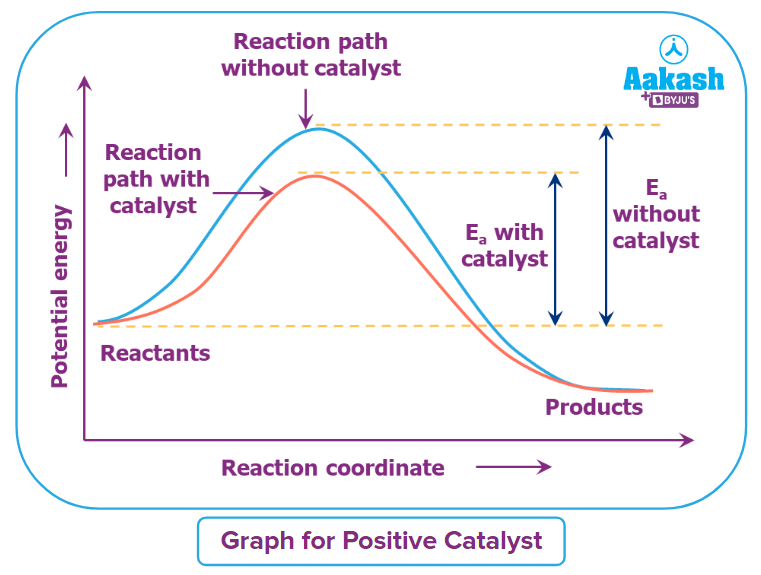
Example:

Here Fe(s) acts as a positive catalyst during the formation of ammonia.
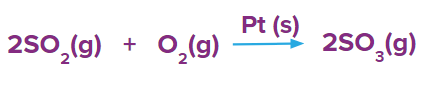
Here Pt (s) acts as a positive catalyst during the formation of sulphur trioxide.
Promoter
It is described as a substance that, although not directly acting as a catalyst, enhances the activity of the catalyst. As a result, when the promoter is given to the reaction, it increases the activity of the catalyst without becoming involved in the chemical reaction.
For instance, Mo acts as a promoter along with Fe (s) catalyst in the formation of ammonia.

Catalytic Poison
These are the substances that do not operate as catalysts yet whose presence lowers the activity of the catalyst. The preferential adsorption of a chemical species, or poison, on the surface of the catalyst results in poisoning. These poisons such as H2S, CS2, PH3 etc or elements such as As, Pb, Hg etc., might be an impurity in the reaction or they might develop as by-products of the reaction.
For instance, Co/H2S acts as a poison in the formation of ammonia, along with a catalyst Fe(s).

Negative Catalyst or Inhibitor
Negative catalyst is employed in chemical reactions to slow down the rate of reaction by raising the activation energy and slow down the speed of reaction. When the negative catalyst is utilised, it engages in a series of reactions with the reaction intermediate, slowing the rate of reaction.
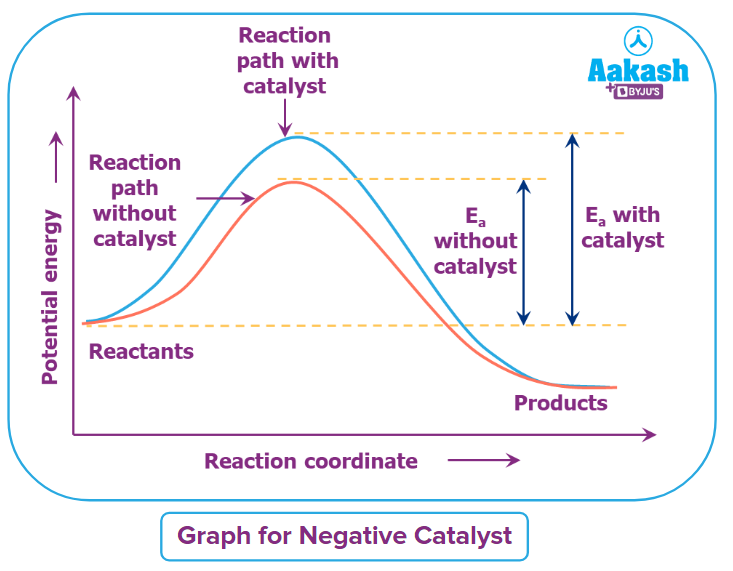

Here alcohol acts as a negative catalyst to decrease the rate of oxidation of sodium sulphite.

Here glycerine acts as a negative catalyst to decrease the rate of decomposition of hydrogen peroxide.
Theories on Catalysis
Intermediate Compound Formation Theory
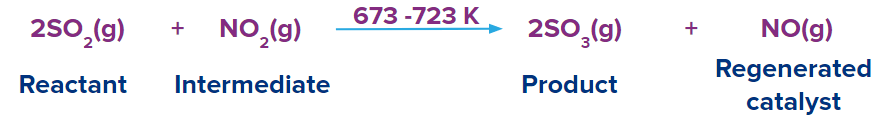
This theory which successfully explains homogeneous catalysis was proposed by Clement and Desormes in 1806. According to this theory, one of the reactants and the catalyst combine to create an intermediate. Because the intermediate component is unstable, it either breaks down or combines with the other reactants to create the product, which regenerates the catalyst.
Reactant A reacts with the catalyst to form an intermediate complex, which further reacts with other Reactant B to give the product and catalyst get regenerated.
Step-1: Reactant A+[Catalyst][Intermediate]
Step-2: [Intermediate]+Reactant BProduct + [Catalyst Regenerated]
Let us discuss intermediate compound theory with an example:

Step 1:

Step 2:
The Modern Adsorption Theory
This theory explains the mechanism of in heterogeneous catalysis. It is a combination of the earlier adsorption theory and the intermediate complex formation theory. According to the earlier version of the adsorption theory of heterogeneous catalysis, the reactants in gaseous or aqueous forms get adsorbed on the solid catalyst, and the increase in the concentration of the reactants on the surface increases the rate of the reaction. This did not explain the specificity of a catalyst. The Modern Adsopion theory has five steps in its mechanism.
- Diffusion of reactant to the catalyst's surface.
- Adsorption of reactant molecules on the catalyst surface.
- Chemical reaction on the catalyst's surface that results in the formation of an intermediate.
- Desorption of reaction products from the catalyst surface, making the surface available for further reaction.
- Diffusion of product from the catalyst's surface.
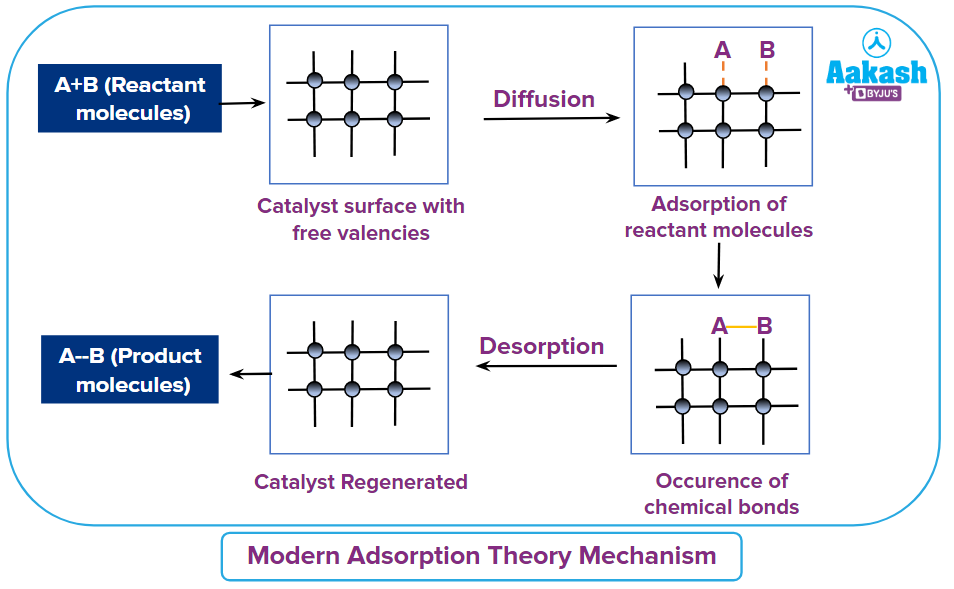
Conclusion: When the gas comes into contact with a surface with free valencies, its molecules are held up due to a loose chemical combination. When different molecules are adsorbed next to each other, they may react, resulting in the formation of new molecules. Following that, formed molecules may evaporate, exposing the surface to new reactant molecules. The free valencies are responsible for the catalytic activity. Therefore, with the increase in the valencies of the catalyst, the rate of the reaction can be increased. The valencies in a catalyst can be increased by fine division of the catalyst and by the rough surface of the catalyst.
This theory explains why the catalyst, even in small quantities, remains unchanged in mass and chemical composition at the end of the reaction.
Zeolites
Zeolites are natural or synthetic microporous aluminosilicates of the formula Mx/n [(AlO2)x (SiO2)y]. zH2O.
Where M=Na+, K+, Ca2+, etc,.
n = Valency of metal cation
z = Water of crystallisation
Shape selective catalysis refers to a catalytic reaction that is influenced by the pore structure of the catalyst as well as the size of the reactant and product molecules. In Greek, zeolite means "Boiling Stone." It was named after the water that became trapped in the pores of the catalyst. In the petrochemical industry, for example, a zeolite catalyst known as ZSM-5 (zeolite sieve molecular porosity 5) converts alcohol to gasoline or hydrocarbons. The alcohol dehydrates in the cavities, forming hydrocarbons.

Some essential points about zeolites:
- These are aluminosilicates with the general formula Mx/n [(AlO2)x (SiO2)y]. zH2O.
- These are three-dimensional silicate networks in which some silicon atoms have been replaced by aluminium atoms.
- Zeolites have honeycomb-like structures and are shape-selective catalysts.

- Zeolites can allow molecules of a certain size to enter and exit the active regions within the holes.
- These are porous and have molecular-sized cavities. A zeolite's internal structure is a network of tunnels and cavities. As a result, zeolites have a massive surface area.
- Reactions proceed in a specific manner depending on the size of the molecules of reactants and products as well as the sizes of the pores of zeolites.
Catalyst – Characteristics
Effectiveness
One catalyst molecule can change one million molecules of the reactant per minute, demonstrating the remarkable efficiency of the catalyst. The mixture of H2 & O2 can be kept in storage for as long as necessary, but when platinum is present, the reaction happens violently and explosively.

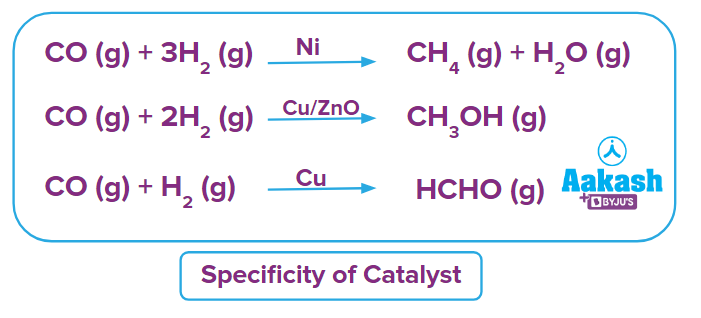
Specificity
The nature of catalysts is very specific. Almost all reactions are regulated by unique catalysts. Different catalysts given for the same reactant will produce different products.
Enzymes
Enzymes are specialised catalysts that are used to catalyse all biological reactions. These are globular proteins. Chemically, all enzymes are proteins that form colloidal solutions and have molar masses ranging from 15,000 to 1,000,000 g/mol. These are created by the living tissue of animals and plants and are also known as biological catalysts or biochemical catalysts.
Examples:
Inversion of cane sugar:

Conversion of glucose into ethyl alcohol:

Enzymes – Mechanisms
There are two main models or mechanisms of enzyme action. They are the lock and key mechanism and the induced fit model.
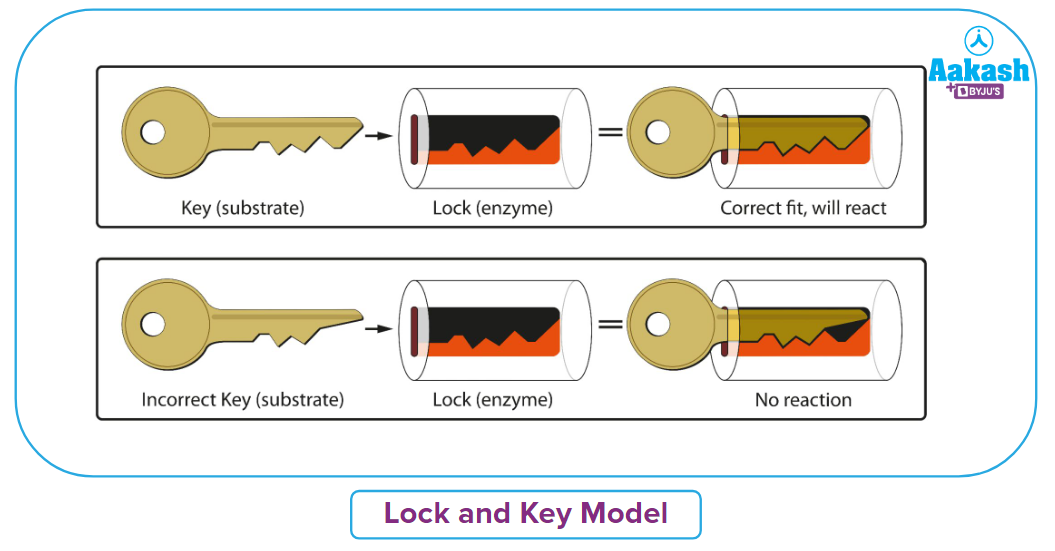
Lock and Key Model
Similar to how a key fits into a lock, the substrate or the molecule on which the enzyme acts fits into the space. Every enzyme has an active site that can only accommodate a certain substrate, similar to how one key can only open a certain lock. This is due to the shape of the active site.

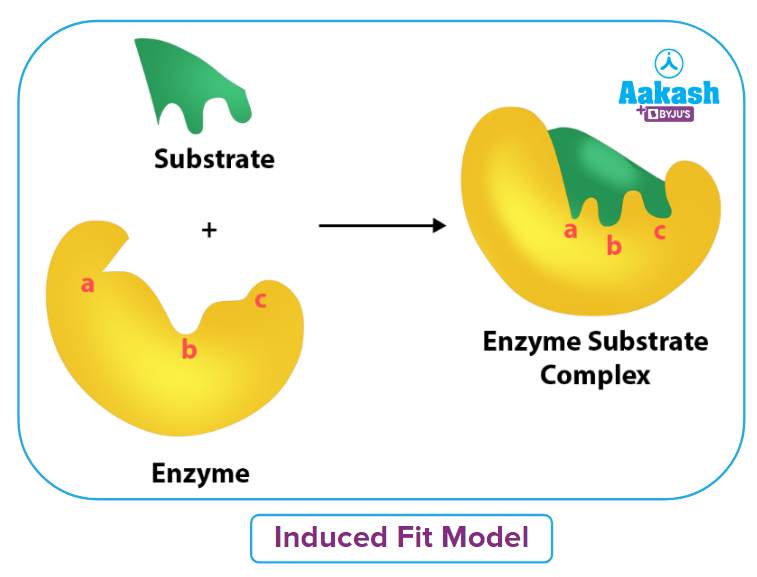
Induced Fit Model
Daniel Koshland proposed the induced-fit model in 1958. It differs from the lock-and-key model, which is also used to explain how an enzyme interacts with a substrate. The substrate and the active site of the enzyme both undergo conformational changes in the induced fit model up until the substrate is fully bound to the enzyme, at which time the final shape and charge are established.
The induced fit model, in contrast to the lock-and-key concept, demonstrates that enzymes are rather flexible structures, with the active site continuously changing form as a result of interactions with the substrate until the substrate is completely bound to it.
The enzyme-catalysed reactions may be considered to proceed in two steps.
- Binding of the enzyme to substrate to form an activated complex.
E+SES#
Where E is the enzyme and S is the substrate.
- Decomposition of the activated complex to form the product.
ES#P+E
Where ES# is the enzyme-substrate activated complex, P is the product and E is the enzyme.
Enzymes – Characteristics
- Effectiveness
The extraordinary effectiveness of the catalyst is demonstrated by the fact that one catalyst molecule can alter one million molecules of the reactant every minute.
- Extremely little amounts
The pace of a reaction can be increased by factors of 103 to 106 by using extremely small amounts of catalysts as little as one-millionth of a mole.
- Specificity
Catalysts have a highly unique nature. Unique catalysts control nearly all reactions. When multiple catalysts are used with the same reactant, different products are created.
- Optimum temperature
At a specific temperature known as the optimal temperature, the rate of a catalytic reaction increases to its maximum. For instance, processes catalysed by enzymes are suitable for human body temperature (310 K).
- Optimum pH
At what is known as the optimal pH, the rate of an enzyme-catalyzed reaction is at its highest. The pH here is in the range of 5 to 7.
- Enhanced activity in the presence of activators or coenzymes
The presence of activators significantly increases the enzymatic activity.
- The effect of inhibitors and poisons
When certain substances known as poisons or enzyme inhibitors are present, the enzymatic activity might be decreased or inhibited.
Catalysts Used in Industry
|
Process |
Catalyst |
|
Haber’s Process: In industry, ammonia is manufactured by Haber’s process. |
In this process, finely divided iron with molybdenum as a promoter is used. The conditions required are 200 bar pressure and 723-773 K. |
|
Contact Process: Sulphuric acid is manufactured in the industry by the contact process. |
In this process, platinised asbestos or vanadium pentoxide (V2O5) as a catalyst, and the temperature required is 673-723 K. |
|
Ostwald’s Process: Nitric acid is manufactured in the industry by Ostwald’s process. |
In this process, platinised asbestos is used as the catalyst and the required reaction temperature is 573 K. |
Recommended Videos
Surface Chemistry Class 12 Chemistry One-Shot (Full Chapter Revision) Concepts+Ques | JEE Main 2022
Top 10 JEE Main Most Important Questions of Surface Chemistry Class 12 | JEE Mains Chemistry 2022
Practice Problems
1. Which of the following accurately describes Haber’s process?
- In the formation of ammonia, , Co/H2S acts as a poison along with catalyst Fe(s).
- In the formation of ammonia, N, Mo acts as a promotor along with catalyst Fe2O3 (s).
- In the formation of ammonia, , Mo acts as an inhibitor along with catalyst Fe(s).
- In the formation of ammonia, , Co/H2S acts as an inhibitor along with catalyst Fe(s).
Answer: A
Solution: Haber’s Process helps in the formation of ammonia on an industrial scale. In this process of formation, , Co/H2S acts as a poison or Mo acts as a promotor along with catalyst Fe(s). Co/H2S is not an inhibitor or negative catalyst, but it helps the negative catalyst to slow down the reaction and hence acts a poison.
So, option A is the correct answer.
2. Which of the following are the correct steps to represent the mechanism of the modern adsorption theory?
- Reactants are desorbed from the catalyst surface, exposing the surface to further reactions.
- The chemical reaction takes place after desorption from the surface of the catalyst.
- Molecules from the reactants get adsorbed to the catalyst surface.
- All of these
Answers: C
Solution: Modern adsorption theory has five steps in its mechanism and these are:
- Reactants diffuse to the surface of the catalyst.
- Molecules from the reactants adsorb to the catalyst surface.
- Chemical reaction that takes place on the catalyst's surface and produces an intermediate product.
- Reaction products are desorption from the catalyst surface, exposing the surface to further reactions.
- Product diffuses from the surface of the catalyst.
Hence, options (A) and (B) are incorrect as the product gets desorbed from the surface of the catalyst, not a reactant molecule and the chemical reaction takes place on the surface of the catalyst, not after the desorption.
So, option C is the correct answer.
3. Which of the following statements is correct about the Zeolite catalyst?
- The catalytic reaction of zeolites is affected by the size of the molecules in the reactant and product.
- The catalytic activity of zeolites depend on the pore structure and are referred to as shape-selective catalysis.
- Thse are three-dimensional honeycomb-like structures in which some silicon atoms have been replaced by aluminium atoms.
- All of these
Answer: D
Solution: The size of the molecules in the reactant and product have an impact on the catalytic reaction of zeolites. They are known as shape-selective catalysis and depend on the pore structure of the catalyst. These are silicon networks in three dimensions that resemble honeycombs, with some silicon atoms substituted by aluminium atoms. So, all the given statements are correct.
So, option D is the correct answer.
4. Can you predict which enzyme is employed in the breakdown of glucose to give alcohol?
- Invertase
- Zymase
- Maltase
- Lactase
Answer: B
Solution: Each and every biological reaction is catalysed by Enzymes. These globular proteins, sometimes referred to as biological or biochemical catalysts, are made by the living tissue of both animals and plants. In the conversion of glucose into ethyl alcohol, zymase is used as an enzyme.

So, option B is the correct answer.
Frequently Asked Questions – FAQ
1. What distinguishes a catalyst from catalysis?
Answer: A catalyst can be given to a process, then once the reaction has taken place, it can be removed and utilised again. Catalysis is the procedure or action through which a catalyst quickens a reaction.
2. What exactly does autocatalysis mean?
Answer: A reaction that is catalysed by one of the reaction's products is referred to as autocatalysis. In this kind of reaction, the rate of reaction will rise as the catalyst is created.
3. What do you mean by induced catalysis?
Answer: Induced catalysis is the term used to describe when one chemical reaction speeds up another chemical reaction.
4. Why is it preferable to use heterogeneous catalysts?
Answer: Catalysts that are heterogeneous play a big part in industrial chemical processing. They are preferred because of their durability and lower operating costs, especially because it is simpler to recover or separate from the products, which enables the simplification of chemical processes.
5. What function does poison play as a catalyst in the Rosenmund reaction?
Answer: In the Rosenmund reaction, hydrogen gas is reduced with acid halides in the presence of palladium to produce aldehyde. The reaction does not end at the aldehyde level, which is a slight reduction of alcohol if a catalyst is not poisoned to halt at the level of aldehyde. With barium sulphate, palladium is poisoned.


