-
Call Now
1800-102-2727
Breaking of Bonds– Homolytic Bond Cleavage, Heterolytic Bond Cleavage, Ring Opening, Practice Problems & FAQs
Consider a dam built with rods and concrete to be a molecule of a certain compound. A dam majorly acts as a physical obstruction that prevents or limits the movement of subsurface or surface streams of water. Dams create reservoirs for a variety of uses, such as flood control, irrigation, food production for humans and animals, industrial use, aquaculture, and navigability.
Dams and floodgates are used to control or prohibit water flow into particular geographical zones, dams typically serve the primary function of retaining water.
But at times of heavy rain, or a glacier melt down, when water overflows in the reservoirs, the force of water can be so high that it can just break apart the dams! Just as depicted in the image below.

Did you just notice how the dam broke into two parts by the extreme force of water! Now depending on the position and magnitude of the force application (as well as the nature of force) the dam could equally break into two parts or into two unequal portions (as shown in the image here)!
In reality, a bond that creates a molecule of any compound, can also be broken by the application of energy. And breaking a bond usually happens for the creation of something new!
You must struggle against the bond in order to break it, just like you would apply hydro pressure to a dam, until it breaks. It takes energy to do this.
So Let’s dive in and find out in detail how bonds break and what it facilitates!
TABLE OF CONTENTS
- Breaking of Bonds
- Homolytic Cleavage
- Heterolytic Cleavage
- Ring Opening
- Practice Problems
- Frequently Asked Questions-FAQs
Breaking of Bonds
Some old bonds dissolve and some new bonds form when an organic substance reacts. When two atoms share electrons, a bond is created; when they do not, a connection is broken. How readily a bond forms and how easily it breaks depend on the particular electrons that are shared, which, in turn, depend on the atoms to which the electrons belong.
Bond cleavage occasionally needs catalysts. For example, The C-H single bond has a high bond-dissociation energy of about 100 kcal mol-1(420 kJ mol-1), which means that it would take a lot of energy to separate the hydrogen atom from the carbon. It always takes energy to break bonds. The following would show on a free energy diagram for the dissolution of a covalent bond between atoms A and B.
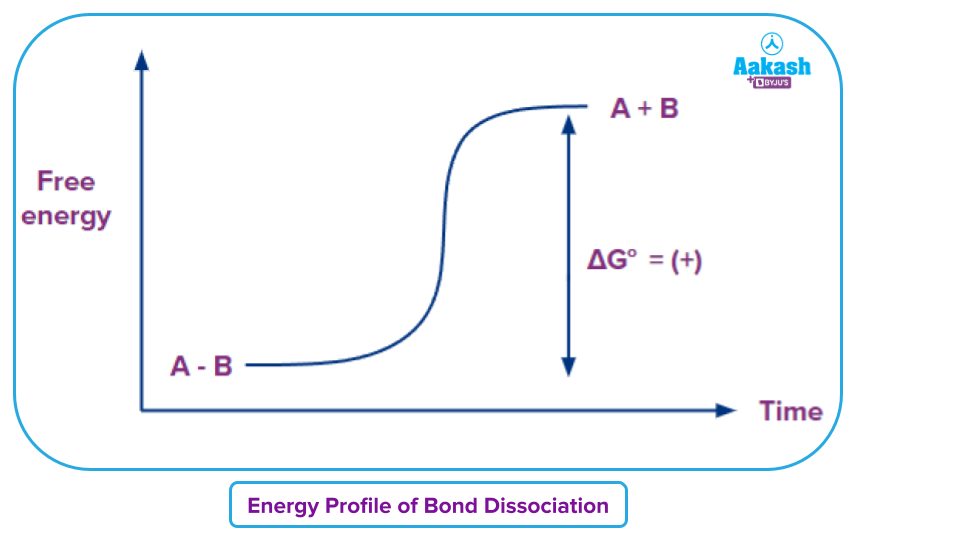
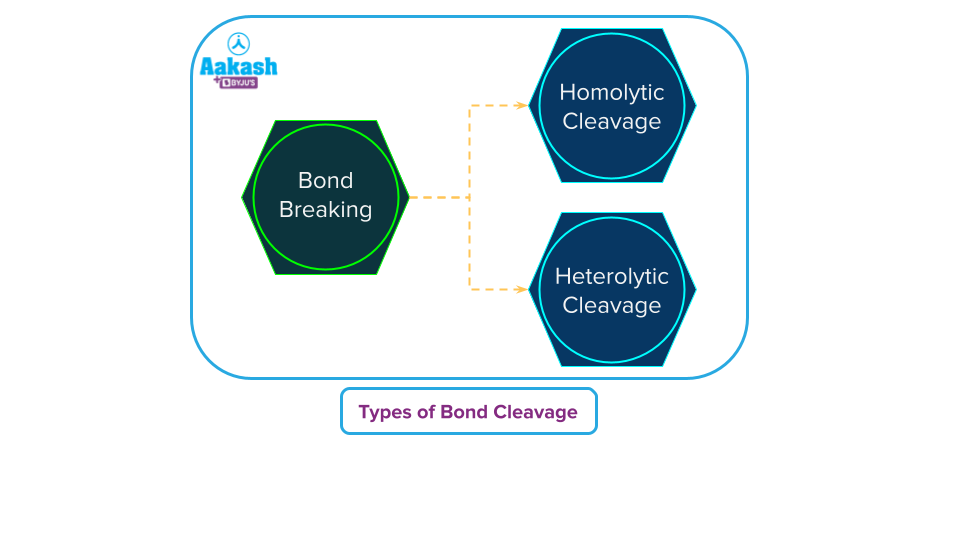
Bond fission is a common term for the breakdown of a chemical bond, most frequently a covalent bond. Homolytic fission and heterolytic fission are the two main forms of bond fission or bond cleavage.
Homolytic Cleavage
The process of a bond breaking in which each atom keeps one of the bonding electrons is known as homolytic bond cleavage or homolysis. Each linked atom shares one of the shared pairs of electrons in a covalent bond. The energy needed to homolytically cleave a bond is known as the bond-dissociation energy. One indicator of the strength of a bond is the enthalpy change.

It should be noted that bond homolysis and homolytic cleavage are other names for homolytic fission. These phrases, which essentially translate to "equal breaking," are derived from the Greek word "homo."
When a particular molecule is dissociated through homolytic fission, which is also known as homolysis, Each atom of the molecule now have one extra electron which these atoms acquired when the bond is broken homolytically. As a result, when a molecule undergoes homolytic fission, the end result is the formation of two free radicals.
The homolytic bond dissociation energy of the molecule is the term used to describe the energy needed to achieve homolytic fission in a molecule.

Here, the homolytic cleavage in Cl2 molecule is shown. Initially, the two Cl- atoms are bonded by the one electron pair and after the homolytic cleavage, the two chlorine radicals are formed.
A lot of energy is typically needed to start a molecule's homolytic fission. This explains why some circumstances, such as those listed below, only result in this kind of bond fission.
- When exposed to UV light, the molecule spilt into the free radicals.
- When the molecule is heated to the necessary level to dissipate the bond dissociation energy needed for homolytic fission.
- Homolysis of bonds mostly occur at temperatures above 200°C.
- When the molecules are heated at higher temperatures without oxygen in order to hasten the breakdown of the molecule i.e., for pyrolysis of the compound.
- In some instances, homolytic fission can be accomplished by giving the molecule a very little heat. One instance is the homolytic fission of the O-O bonds in peroxides. These bonds are weak, which suggests that their bond dissociation energies are low. Therefore, only a minimal amount of heat energy is required to pass through this barrier.
- Free radical intermediates are obtained as products on homolysis.
Factors Controlling Homolytic Cleavage
Electronegativity: The electronegativity of the species that are bound affect the bond dissociation energy.
A bond between two electronegative atoms will have a larger bond dissociation enthalpy than a similar molecule with two less electronegative atoms because fewer electronegative atoms are stronger stabilisers of radicals. For example, BDE of H-H molecule is 104.2 kcal mol-1 and the BDE of H-F molecule is even greater due to higher electronegativity difference i.e., 134.6 kcal mol-1.
Polarisability: An atom can stabilise the free radicals produced after homolysis better if its electron cloud is bigger and more polarizable. Iodine, which is highly polarizable and a radical stabiliser, is an example of an atom with a big electron cloud and high polarisability.
Hybridisation of Orbitals: The s- character of an orbital describes how near the electrons are to the nucleus. The s- character of a radical more precisely describes the distance between the single electron and the nucleus. Radicals become less stable as they get closer to the nucleus because of the orbital's increased electron affinity. Hybridizations with the lowest s-character tend to boost radical stability while lowering bond dissociation energy (i.e. sp3 hybridization is most stabilising)
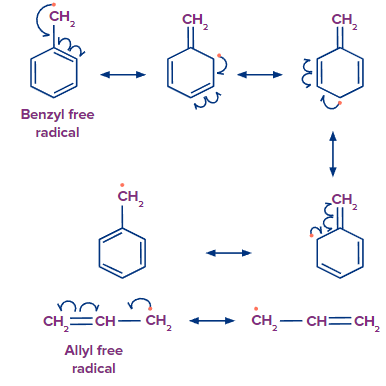
Resonance: Resonance can stabilise free radicals, thereby supporting homolytic fission. By donating negative charge from resonance, or electron delocalization, radicals can be kept stable.
For example, this BrO2 radical is stabilised by resonance.

Resonance also explains the stability of benzyl and allylic free radicals.
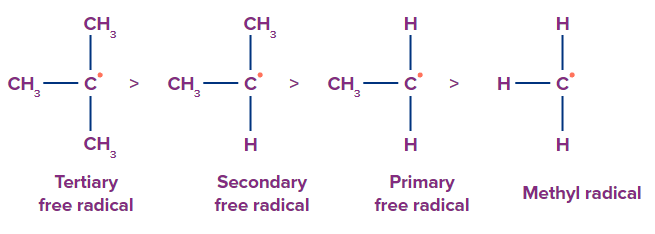
Hyperconjugation: Hyperconjugation stabilises carbon radicals, making more substituted carbons more stable and having less bond dissociation energies (BDE) as a result. Free radicals have the same order of stability as carbocations, namely 3° >2° >1°.
This stability order can be explained using hyperconjugation: The number of alkyl groups attached to the carbon atom carrying the odd electrons increases the delocalization of the odd electrons, making the alkyl free radical more stable. For example, as shown below, the presence of a maximum of three CH3 groups which means it has 9 alpha hydrogens, gives it 9 hyperconjugative structures and hence it is the most stable.
Captodative Effect: . By working together, electron-withdrawing group and electron-donating group substituents can stabilise radicals. This is called the captodative effect. The term "captor group" refers to the electron-withdrawing group (EWG), whereas "dative substituent" refers to the electron-donating group (EDG).
Electron-withdrawing groups attached to the free radical carbon destabilise the free radical and the electron-donating group on the other hand stabilises the carbon free radical.
Heterolytic Cleavage
Heterolytic cleavage occurs when the bond breaks such that one fragment takes away both the electrons of the bond. It produces one anion and leaves the other fragment with an empty orbital (so basically an anion and a cationic species).
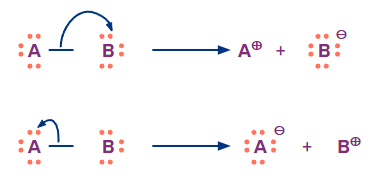
When a covalent bond between two atoms is broken unevenly during heterolytic fission, one of the chemical species retains the bond pair of electrons, which will have one extra electron on it and attains a negative charge (known as anion). The other product of heterolytic fission will have a positive charge (known as cation).
Heterolysis is a phrase of Greek origins that approximately translates to "unequal breaking." Additionally known as homolytic cleavage.
Below is a diagram showing the possible way that a molecule CH3Cl can experience heterolytic fission:
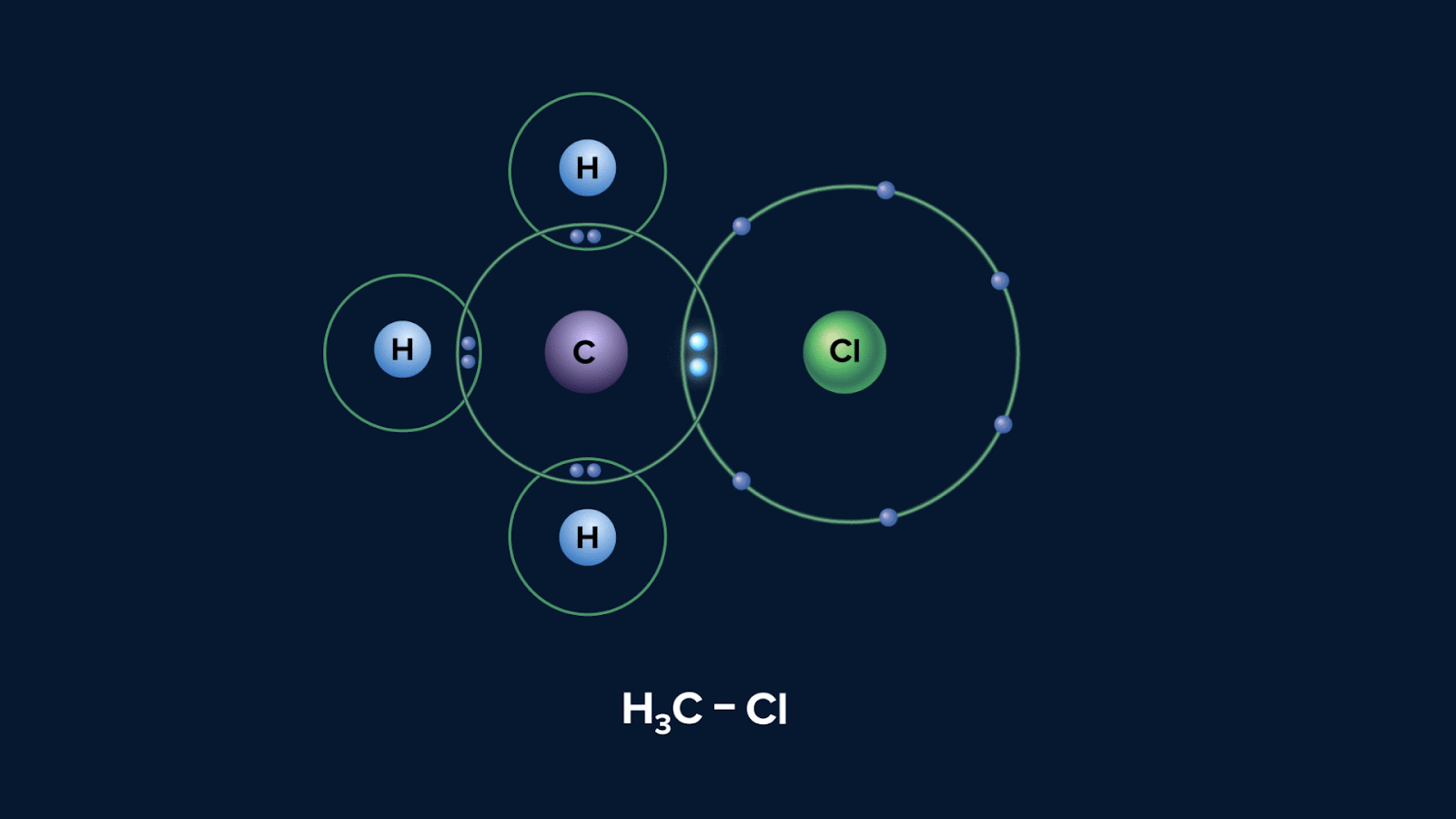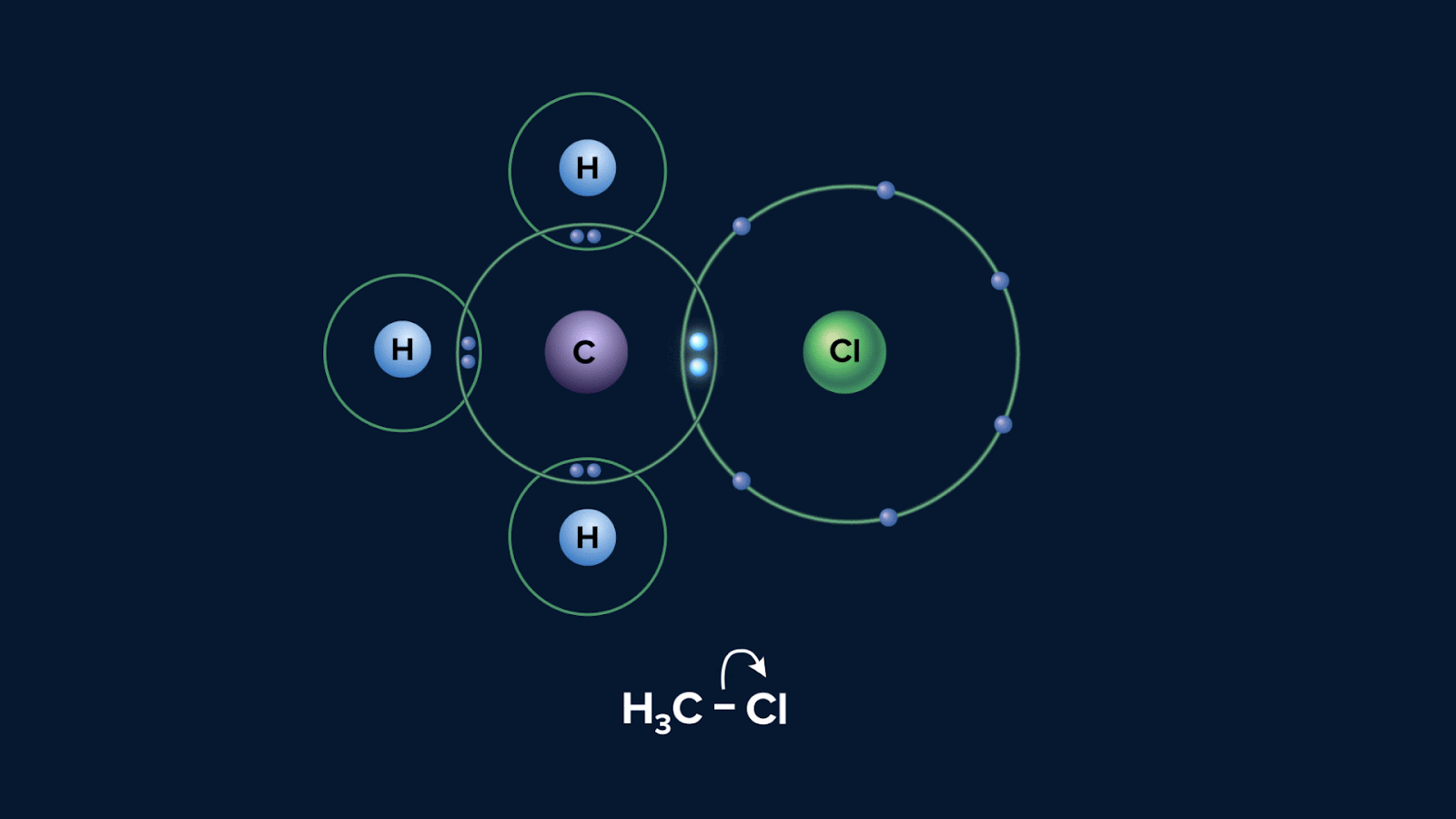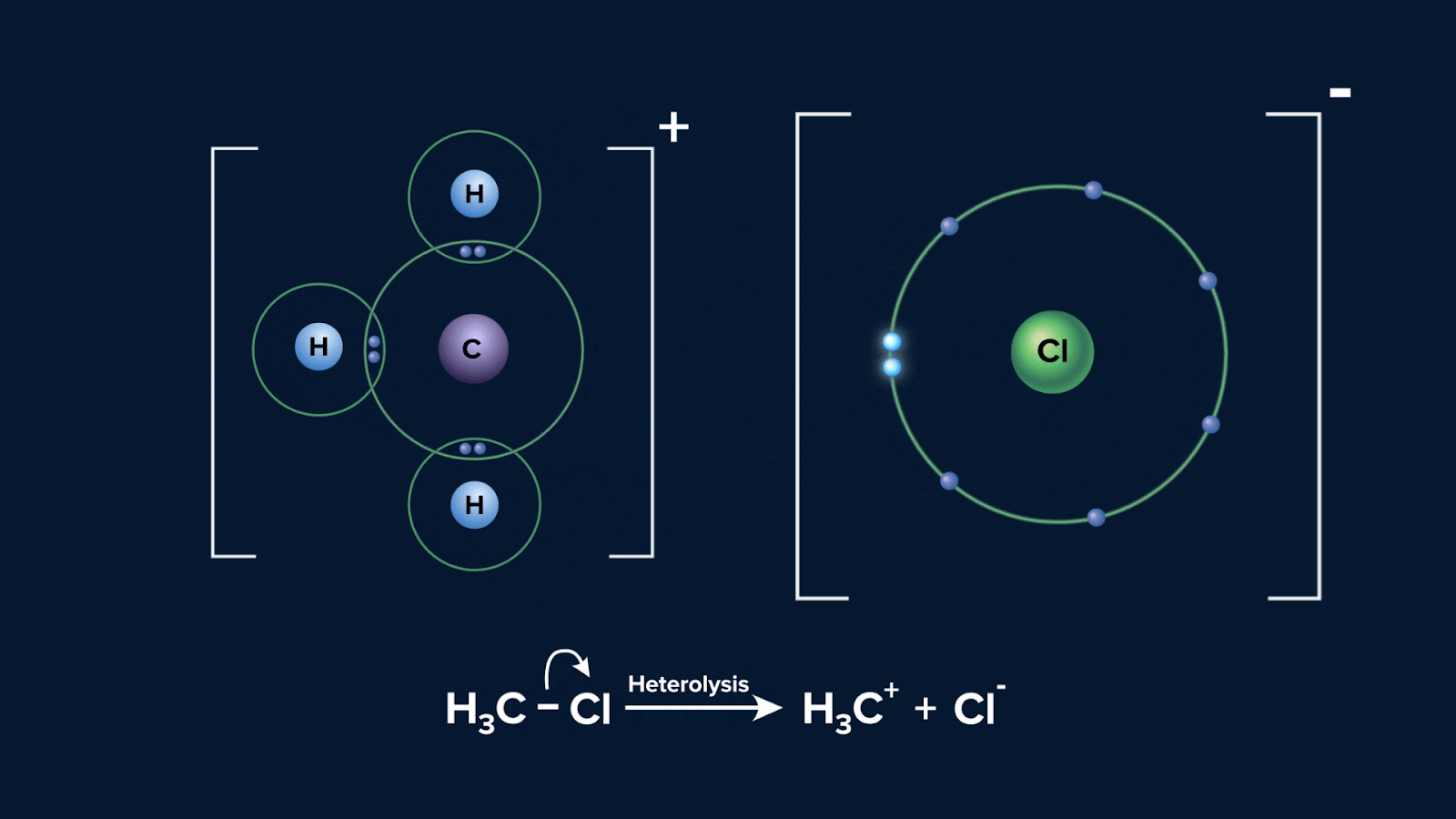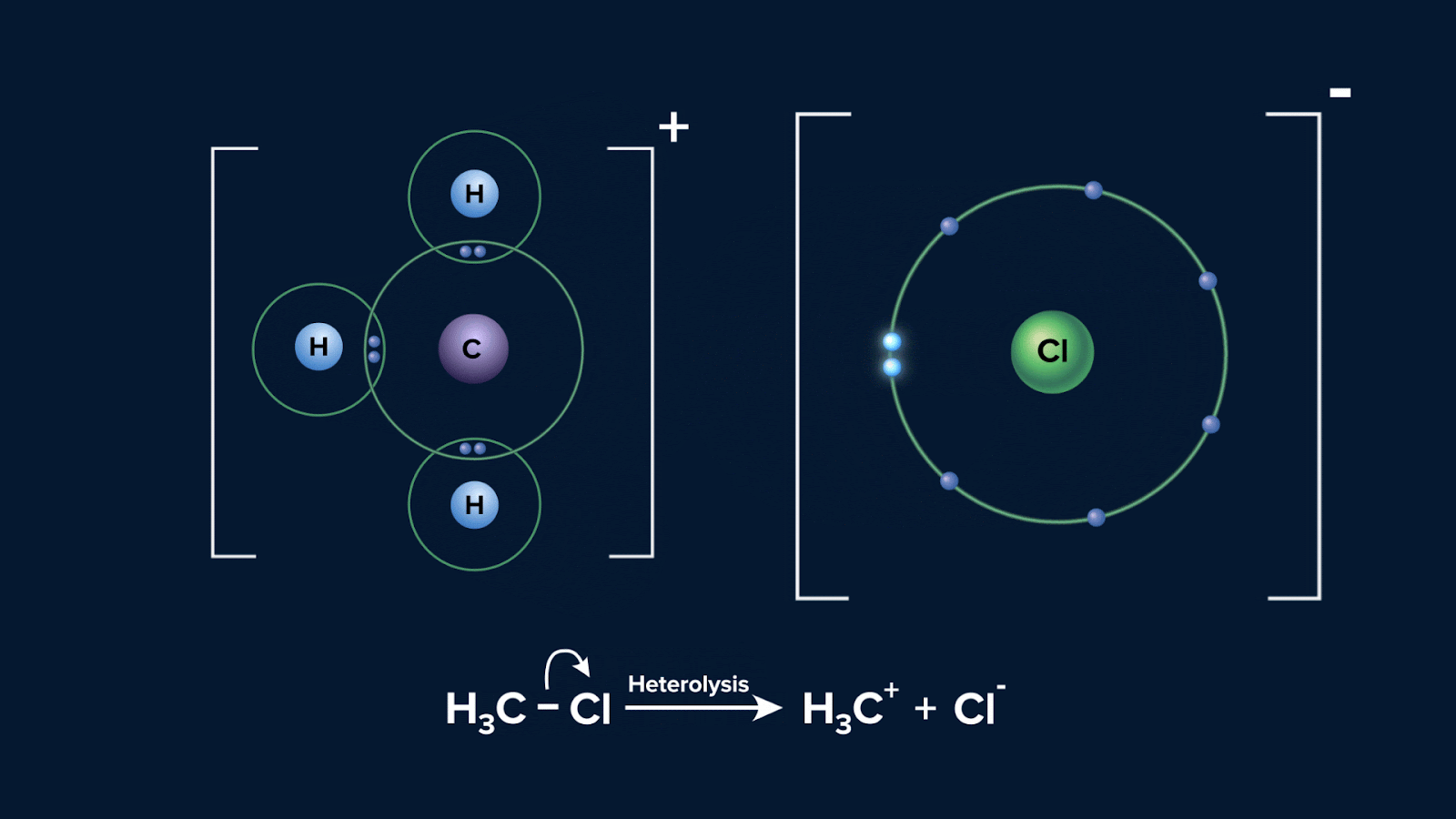
Polarisation of a bond usually results from different electronegativities of the atoms joined by the bond. The greater the difference in electronegativity, the greater the polarisation. In the given instance, atom Cl is more electronegative than C, hence electron will be pulled towards Cl.
The bonded species with the higher electronegativity is often the one that retains the bond pair of electrons and acquires a negative charge when a covalent bond is subjected to heterolytic fission. On the other hand, the more electropositive species, typically does not hold onto any electrons and develops a positive charge.
The heterolytic bond dissociation energy is a common name for the quantity of energy needed to scission a covalent bond by heterolytic scission. The hydrogen chloride molecule is a good example of heterolytic fission, as shown in the chemical reaction below.
H-Cl H+ + Cl-
Due to its stronger electronegativity than hydrogen in this situation, the chlorine atom is able to keep its bond pair of electrons. Chloride anion and hydrogen cation are the end results.
- Single bonds nearly invariably undergo heterolytic fission, which typically results in two fragment types.
- The heterolytic bond dissociation energy, which is comparable to but not the same as the homolytic bond dissociation energy typically employed to indicate the energy value of a bond, is the amount of energy needed to break the bond.
- The leaving group departs with the shared pair of electrons when the bond between carbon and the atom linked to it is severed heterolytically, leaving the carbon as a carbocation, which is an electron-deficient species.
- The likelihood of bond cleavage or the development of a more stable carbocation increases with decreasing activation energy.
- The rate of ionisation of the covalent bond has a significant impact on the rate of reaction for many reactions involving unimolecular heterolysis. They discovered that the type of solvent has a significant impact on the rate of heterolysis.
- For instance, switching from hexane to water accelerates the heterolysis of tert-Butyl chloride (t-BuCl) by 14 orders of magnitude. Strong solvation of the transition state is what is to blame for this. The solvent's polarity, electrophilicity, and ionising power are the primary parameters that influence heterolysis rates.
Ring Opening
Ring-opening is basically electrocyclic reactions in which cyclic organic molecules open up under the application of heat or certain specific attacking reagents to produce new acyclic molecules. Three-membered rings are highly strained and so by attack or nucleophiles or by application of heat, they can be easily cleaved.
The cleaved molecule stays as a single unit during a ring-opening. Although the link is broken, other structural elements still hold the two fragments together. One of the polar carbon-oxygen bonds in an epoxide ring, for instance, can be cleaved by heterolysis to create a single acyclic structure.
For example- Epoxide ring opening reaction
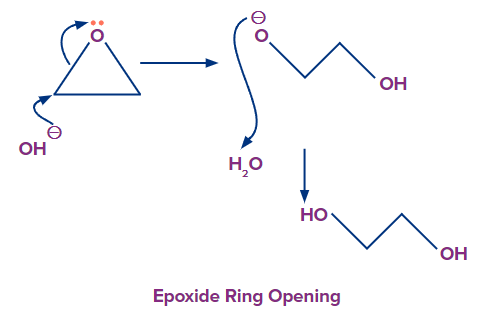
Let’s consider an example: Epoxide ring opening using a basic nucleophile.
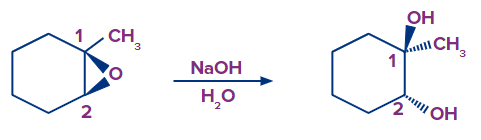
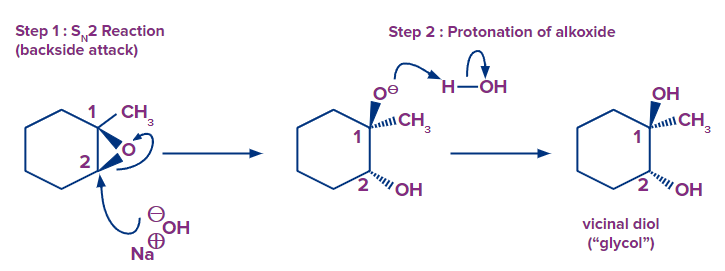
Ring-opening of epoxides with basic nucleophiles. The nucleophile (OH- ) attacks the epoxide at the least substituted position (C2) and results in inversion of stereochemistry at this position. The mechanism proceeds through SN2 followed by the transfer of a proton from the weakly acidic solvent (H2O) to the alkoxide (RO- ) providing neutral alcohol.
Practice Problems
Q.1. Name the species obtained on homolysis.
- Free Radical
- Carbocation
- Carbanion
- Carbene
Answer: (A)
Solution: Homolysis breaks the bond pair equally into two parts such that each bonding atom gets one each electron of the bond pair. Hence homolysis produces free radicals.

So option A is the correct answer.
Q.2. Homolytic fission is favoured by:
- Polar solvents & low temperature
- Non-polar solvent & high temperature
- Polar solvent & high temperature
- Non-polar solvent & low temperature
Answer: (C)
Solution: Free radicals are neutral species, thus non-polar solvents encourage their production. The presence of radiation with a high temperature or energy (such as ultraviolet rays) also encourages homolytic fission.
Q. 3. Which among the following is/are characteristics of a free radical?
- Presence of a negative or positive charge
- Presence of an unpaired electron
- Presence of an odd number of electrons
- Produced by homolytic fission of a bond
Answer: (B), (C), (D)
Solution:
Since a free radical does not have any charge, So A is an incorrect option.
Since a free radical has an unpaired or odd electron, So B is a correct option.
Since a free radical has an unpaired electron, the total number of electrons must be odd. So, C is the correct option.
Since radicals are produced when a covalent bond is cleaved homolytically, So D is the correct option.
Hence, options (B), (C), and (D) are the correct answers.
Q.4. C-Cl will be more readily cleaved by:
- Homolytic Cleavage
- Heterolytic Cleavage
- Both A & B
- None of these
Answer: (B)
Solution: Since there is a considerable electronegativity difference between carbon and chlorine atoms, with chlorine being electronegative, it attracts the bonded pair of electrons towards itself and hence heterolytic cleavage of the bond will happen. So option B is the answer.
Frequently Asked Questions-FAQs
Q.1 Give a comparison between homolysis and heterolysis.
Answer: The heterolytic bond dissociation energy is significantly larger than the homolytic dissociation for the same type of bond when comparing the bond dissociation energies. A neutral molecule undergoes heterolysis, producing both a positive and a negative ion. However, it takes a lot of energy to separate these oppositely charged particles. Bond dissociation takes place more easily in the gas phase through a process called homolysis. However, heterolysis is the preferred type of breaking in an ionising solvent.
Q.2 Mention some natural sources of free radicals in our body.
Answer: Everybody's body naturally produces some free radicals as a result of physiological processes like exercise or inflammation. This is typical and a necessary component of the body's complex self-maintenance system. Free radicals in the environment could possibly come into contact with you which can be extremely harmful.
Free radicals are also produced by other external sources such as exposure to X-rays, ozone, smoking, air pollution, and industrial toxins.
Q.3. Name some common bonds easily cleaved by homolysis.
Answer: The C-O, C-Cl, C-Br, C-I, C-C, and C-H common bonds that cleave to produce free radicals in organic chemistry are shown. Most carbon free radicals are produced by photolysis (light action), such as acetone alpha cleavage.
Other radical initiators exist, such as N-allylic bromosuccinimide's bromination (NBS).
Q.4. State some reactions undergone by carbocations.
Answer: Even weak nucleophiles, like water, will cause carbocations to react and produce a new bond. In order to generate a double (or, in some circumstances, triple) bond from the carbocation, carbons alpha to the carbocation sometimes lose a proton. To remove the proton in this process, only a mild base is needed, such as chloride.
So carbocations can undergo elimination (of a hydrogen) to form a new -bond. Before the ion is stabilised by one of the aforementioned events, a secondary carbocation may change the configuration to generate a tertiary carbocation.


