-
Call Now
1800-102-2727
Borax Bead Test – Structure and Properties of Borax, Processes and Reactions of Borax Bead Test, Practice Problems and FAQ
Do you enjoy viewing fireworks?

Obviously, we don't love the noise it makes or the pollution it causes to our environment, but despite all the drawbacks, fireworks are undoubtedly a beautiful sight that we all adore.
Have you ever wondered what the chemicals are that burn and make people happy?
Well, group 1 metal salts in particular make up the majority of the ingredients in pyrotechnics. When exposed to flame, they emit a recognisable colour. These metal salts are used by cracker manufacturers to create vibrant fireworks.
The flame test can be used to quickly identify these salts because of their propensity to produce colours.
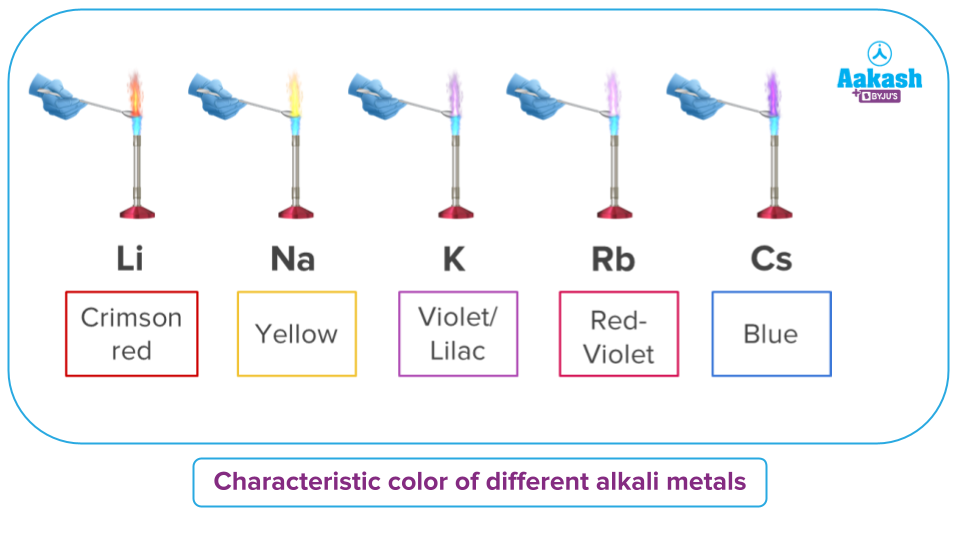
One of these tests is the borax bead test, in which the borax reacts with the appropriate cation or anion of the salt to produce a characteristic colour of the salt.
The Borax Bead Test will be thoroughly explained on this concept page.
TABLE OF CONTENTS
- Borax
- Borax – Physical Properties
- Borax – Chemical Properties
- Borax–Bead Test
- Borax–Bead Test – Chemical Changes
- Practice Problems
- Frequently Asked Questions – FAQ
Borax
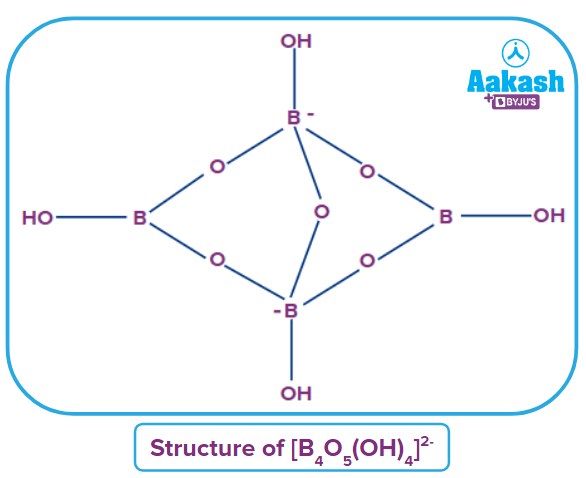
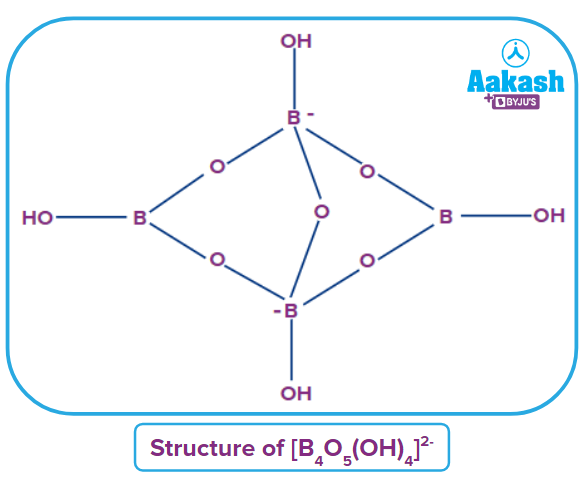
Borax is one of the most important compounds of boron. The chemical formula of borax is Practically, borax is present as the tetranuclear unit . Hence, the formula of borax is correctly represented as
Structure:
Borax – Physical Properties
- White crystalline solid
- In its anhydrous form, borax has a molar mass of 202.2 g/mol and 381.38 g/mol (decahydrate).
- Borax has a density of 1.7 g/cm3
- Borax has a refractive index of 1.472.
- 1,575°C is the boiling point of borax.
- Borax has a melting point of 743 °C.
Borax – Chemical Properties
- Borax burns with a yellowish-green flame and is ignitable or flammable.
- Borax is a substance that dissolves in water at a rate of 31.7 g/L.
- In comparison to acetone, ethylene glycol has a high solubility for borax.
- Boric acid is created when borax and hydrochloric acid react. The involved reaction is listed as,
Borax–Bead Test
This test is used for coloured salts because borax reacts with the corresponding metal salts to form metal borates, which show their characteristic colours.
- The platinum wire is made into a loop at one end, and borax powder is placed inside. The reddest part of the flame is used to heat the platinum wire that is holding the borax powder.
- Borax powder expands as it loses its water if crystallisation, then contracts to form a transparent, colourless bead made of sodium metaborate and boric anhydride.
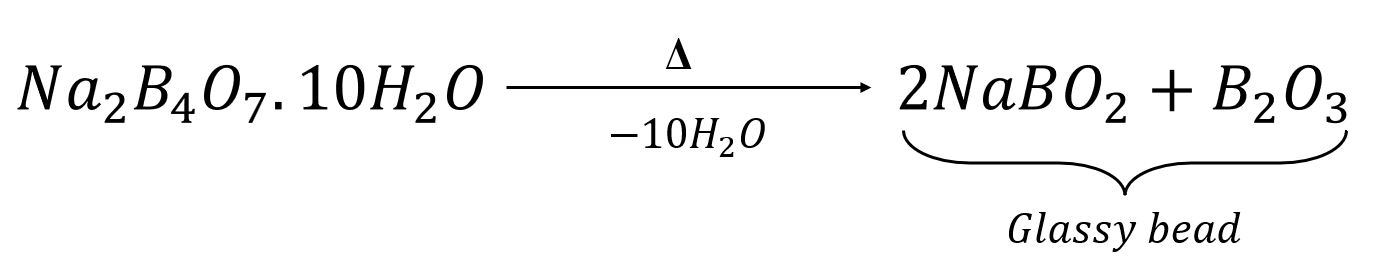
- The hot bead is placed in the salt sample, heated in the flame, and its colour is noted for both the luminous and non-luminous flames, both when they are hot and when they are cool.
Borax–Bead Test – Chemical Changes
- Borax loses its crystallisation water when heated, forming sodium metaborate and boric anhydride.
- Treatment with metal salt:
Blue colour bead of Co(BO2)2 (cobalt metaborate) formation takes place.
For various metals, the colour of the borax bead is listed below.
|
Metal |
Oxidising Flame |
Reducing Flame |
||
|
Hot |
Cold |
Hot |
Cold |
|
|
Cr |
Yellow
|
Green
|
Green
|
Green
|
|
Mn |
Violet
|
Violet
|
Colourless |
Colourless |
|
Fe |
Yellowish brown
|
Yellow
|
Green
|
Green
|
|
Co |
Blue
|
Blue
|
Blue
|
Blue
|
|
Ni |
Violet
|
Reddish brown
|
Grey
|
Grey
|
|
Cu |
Green
|
Blue
|
Colourless |
Opaque |
Practice Problems
1. What colour does a chromium salt give in the borax-bead test?
- Blue
- Green
- White
- Yellow
Answer: B
Solution: The term "borax bead test" refers to the use of borax beads for the detection of coloured basic radicals. Heat causes sodium metaborate and boric anhydride to form from borax, which then react with a basic radical to produce a glassy bead and a colour.
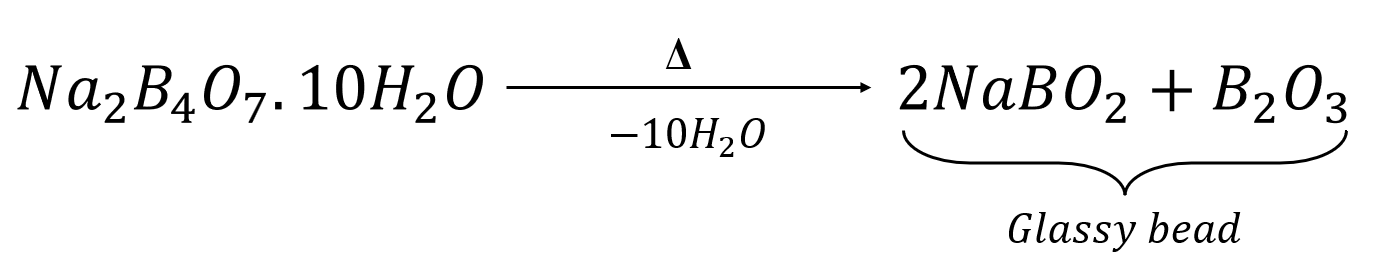
The reaction of chromium salts with the glassy bead produces chromium metaborate, which is green in colour.
![]()
Chromium Metaborate (Green)
So, option B is the correct answer.
2. What colour does a nickel salt give in the borax-bead test?
- Reddish brown
- Yellowish brown
- Greenish Blue
- White
Answer: A
Solution: The term "borax bead test" refers to the use of borax beads for the detection of coloured basic radicals. Heat causes sodium metaborate and boric anhydride to form from borax, which then reacts with a basic radical to produce a glassy bead and a colour.
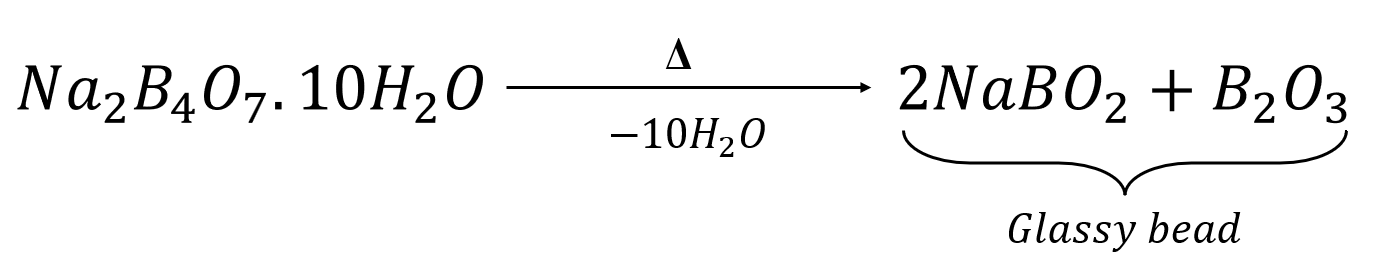
The reaction of a nickel salt with the glassy bead produces nickel metaborate, which is reddish brown in colour.
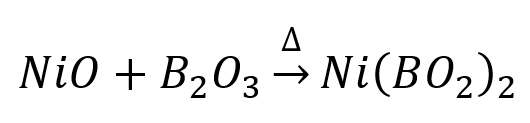
Nickel Metaborate (Reddish brown)
So, option A is the correct answer.
3. What colour does a manganese salt give in the borax-bead test?
- White
- Green
- Violet
- Blue
Answer: C
Solution: The term "borax bead test" refers to the use of borax beads for the detection of coloured basic radicals. Heat causes sodium metaborate and boric anhydride to form from borax, which then reacts with a basic radical to produce a glassy bead and a colour.
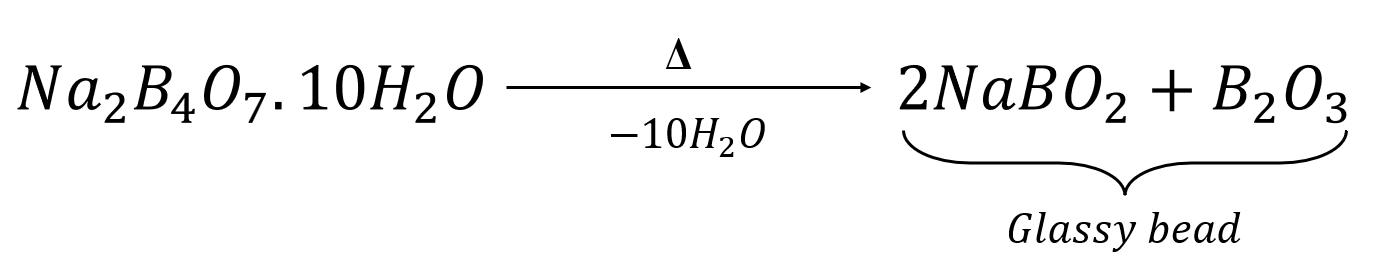
The reaction of a manganese salt with the glassy bead produces manganese metaborate, which is violet in colour.
![]()
Manganese Metaborate (violet)
So, option C is the correct answer.
4. The molecular formula of borax is
Answer: A
Solution: The chemical formula of borax is Practically, borax is present as the tetranuclear unit . Hence, the formula of borax is correctly represented as
So, option A is the correct answer.
Frequently Asked Questions – FAQ
1. Why the borax bead test is invalid for white salts?
Answer: Only coloured salts or white salts that produce coloured residue when heated can be tested with borax beads. Coloured salts break down into coloured metal metaborates in the borax bead test. Borax is heated to produce a glassy bead of boric anhydride, which reacts with the basic component of coloured salt to produce metaborates with distinctive colours. Hence, the borax bead test is invalid for white salts
2. What kind of bond do inorganic salts contain?
Answer: In general, the neutralisation reaction between a mineral acid and a base produces an inorganic salt.
When added to water, acids are chemical substances that release hydrogen ions, and bases are the chemical substances that release hydroxyl ions. Hence, there will be electrovalent or ionic bonds present in inorganic salts.
3. How can salt be easily distinguished in salt analysis?
Answer:
Step1: Always check the salt's colour before using it. Salts of some cations are coloured. You can immediately perform a preliminary test to determine the cation that creates salts of a particular colour if the salt is coloured.
Step2: A flame test should be done first if the salt is colourless.
Step3: Continue with group-wise preliminary tests for cations if the flame test does not reveal anything about the cation. With the sulphate anion , Sr2+, Ba2+, Ca2+, and Pb2+ do not form salts. With the phosphate anion , only group 2, group 1, and group 0 cations can form salts.
You do not need to run tests for the corresponding anions if one of these cations is found in the salt analysis.
4. What do you understand by qualitative analysis?
Answer: Qualitative analysis is the process of identification or determination of specific substances or elements from a given sample by going through the physical and chemical reactions of the sample.






