-
Call Now
1800-102-2727
Basicity – Introduction, Types, Examples, Practice Problems and FAQ
When we were kids, we all played with Legos, right?
Have you paid special attention to each variety of Legos? If so, you may have observed that different Lego piece types contain different numbers of studs, or bumps, that allow us to connect one Lego piece to another.

Why are we talking about Legos right now? Is there a relationship between the subject under discussion and Legos?
There is a connection. There are acids in chemistry that have a varying number of replaceable protons (H+), just like different Lego parts have different studs that allow us to join the Lego pieces. The number of replaceable H+ ions in an acid is called the basicity of the acid.
We shall learn more about basicity, its types, examples, and the factors impacting them on this concept page.
TABLE OF CONTENTS
- Acids and Bases
- Basicity – Introduction
- Basicity – Types With Examples
- Finding the Basicity of Oxoacids of Phosphorus
- Practice Problems
- Frequently Asked Questions – FAQ
Acids and Bases
Before getting to know basicity, it is highly important to understand acids and bases.
Arrhenius Concept of Acids and Bases
According to Arrhenius’ concept, acids are substances that dissociate in water to given H+(aq) ions, whereases bases are substances that dissociate in water to given OH- (aq) ions.
H+ ions are unstable and hence highly reactive in nature. So, they do not exist freely in aqueous solutions and combine with the oxygen atom of a water molecule to form hydronium ion (H3O+) that has a trigonal pyramidal structure.
A molecule of base hydrolyses in water as shown below.
Bronsted – Lowry Concept of Acids and Bases
Johannes Bronsted, a Danish chemist, and Thomas M. Lowry, an English chemist, proposed the Bronsted–Lowry concept of acids and bases. According to this theory, acids are substances that donate H+ ions and bases are the substances that accept H+ ions.
Example:
Base Acid Conjugate Acid (CA) Conjugate Base (CB)
In the reaction given above, H2O acts as a proton donor and NH3 acts as a proton acceptor. So, H2O and NH3 are Bronsted–Lowry acid and base, respectively.
Basicity – Introduction
The number of replaceable hydrogen ions (H+) that can be produced by one molecule of acid in its aqueous solution is called the basicity of the acid.
Basicity – Types With Examples
Based on the number of replaceable hydrogen ions (H+) that can be produced, acids can be classified into monobasic, dibasic, tribasic and polybasic acids.
- Monobasic Acid: When an acid contains only one replaceable hydrogen ion (H+), it is called a monobasic acid. Examples include nitric acid (HNO3), hydrochloric acid (HCl), etc.

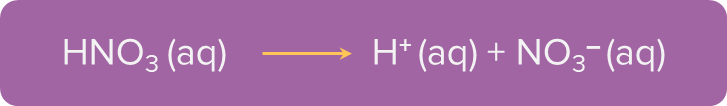
- Dibasic Acid: When an acid contains two replaceable hydrogen ions (H+), it is called a dibasic acid. Examples include sulphuric acid (H2SO4), carbonic acid (H2CO3), etc.


- Tribasic Acid: When an acid contains three replaceable hydrogen ions (H+), it is called a tribasic acid. Example: Phosphoric acid (H3PO4).

Acids which contain more than one replaceable or ionisable hydrogen ions (H+) are generally called polybasic acids. H2SO4, H2CO3, H3PO3, H3PO4, H4SiO4 are some examples of polybasic acids.
Finding the Basicity of Oxoacids of Phosphorus
The basicity of an acid does not depend upon the number of hydrogen atoms present, rather it depends only on the number of ionisable or replaceable H+ ions. This can be understood better from the basicity of the oxoacids of phosphorus.
- Hypophosphorous acid (H3PO2):
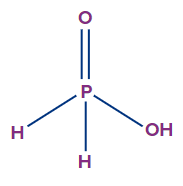
- It is also called as phosphinic acid.
- The oxidation state of phosphorus in hypophosphorous acid is +1, and it is a monobasic acid.
- Though there are three hydrogen atoms present in this acid molecule, the number of ionisable or replaceable H+ ions is one (one O-H bond). So, it is a monobasic acid.
- Orthophosphorous acid (H3PO3):
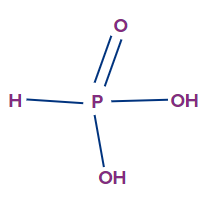
- It is also called as phosphonic acid.
- The oxidation state of phosphorus in orthophosphorous acid is +3, and it is a dibasic acid.
- Though there are three hydrogen atoms present in this acid molecule, the number of ionisable or replaceable H+ ions is two (two O-H bonds). So, it is a dibasic acid.
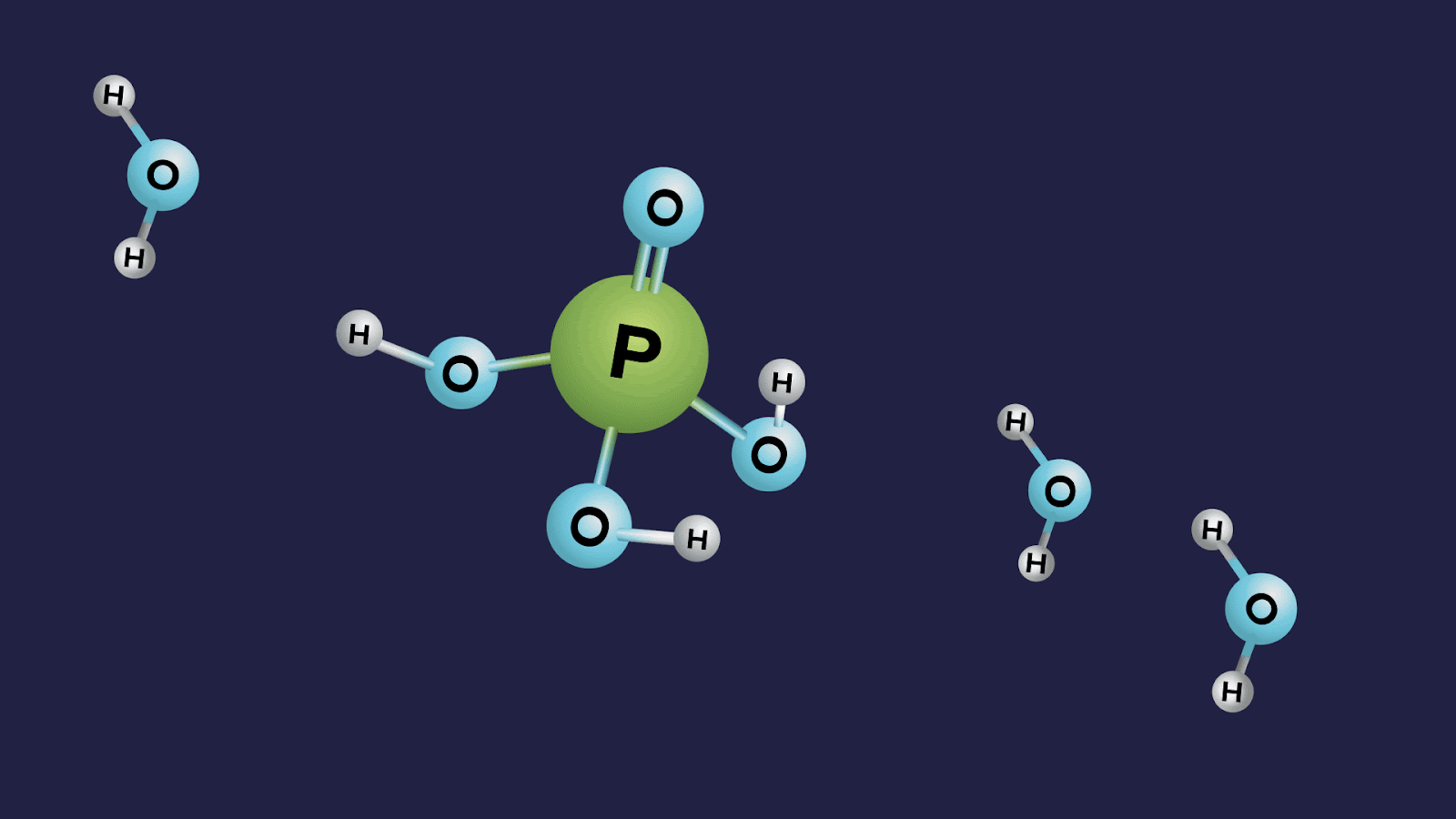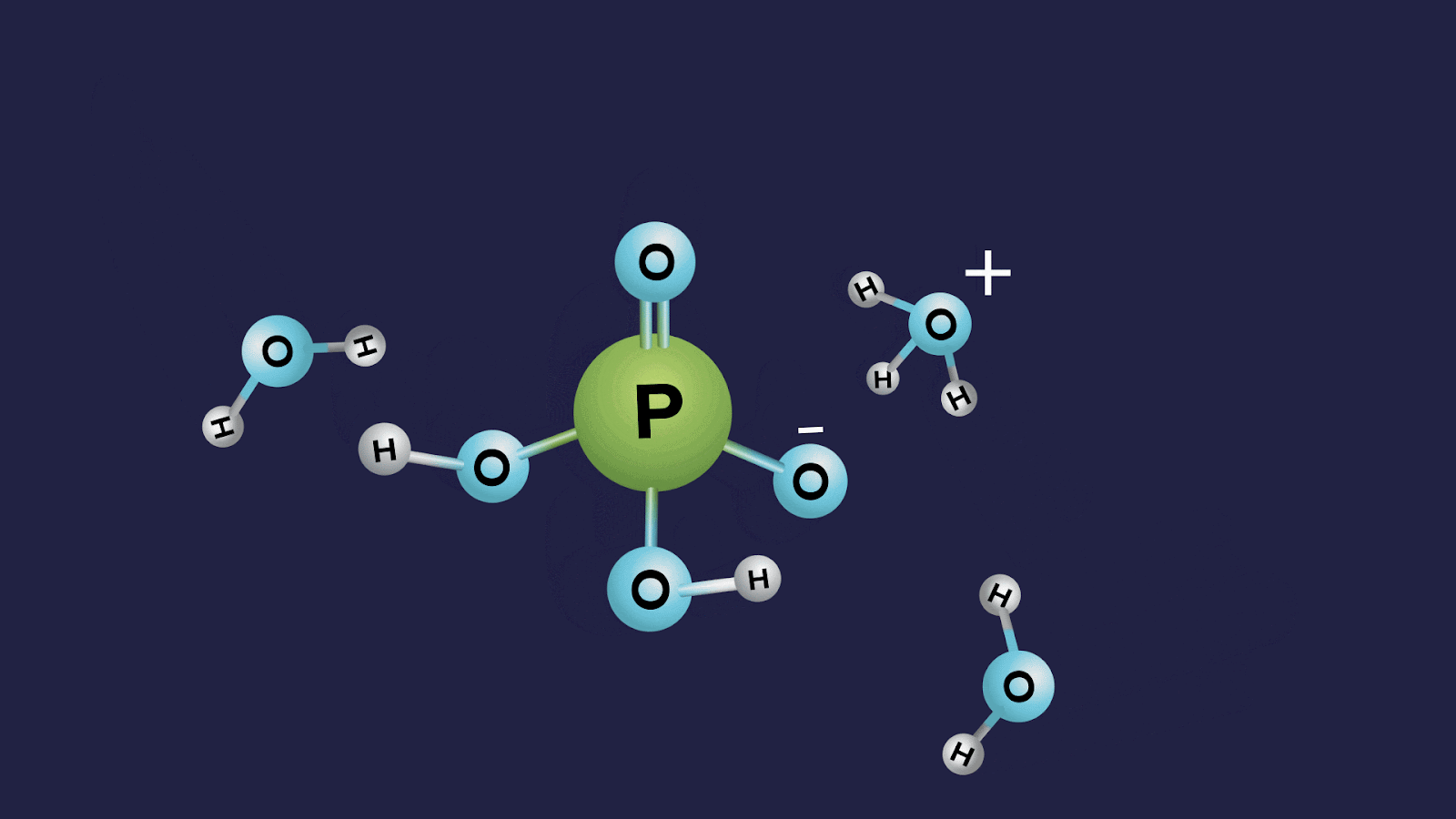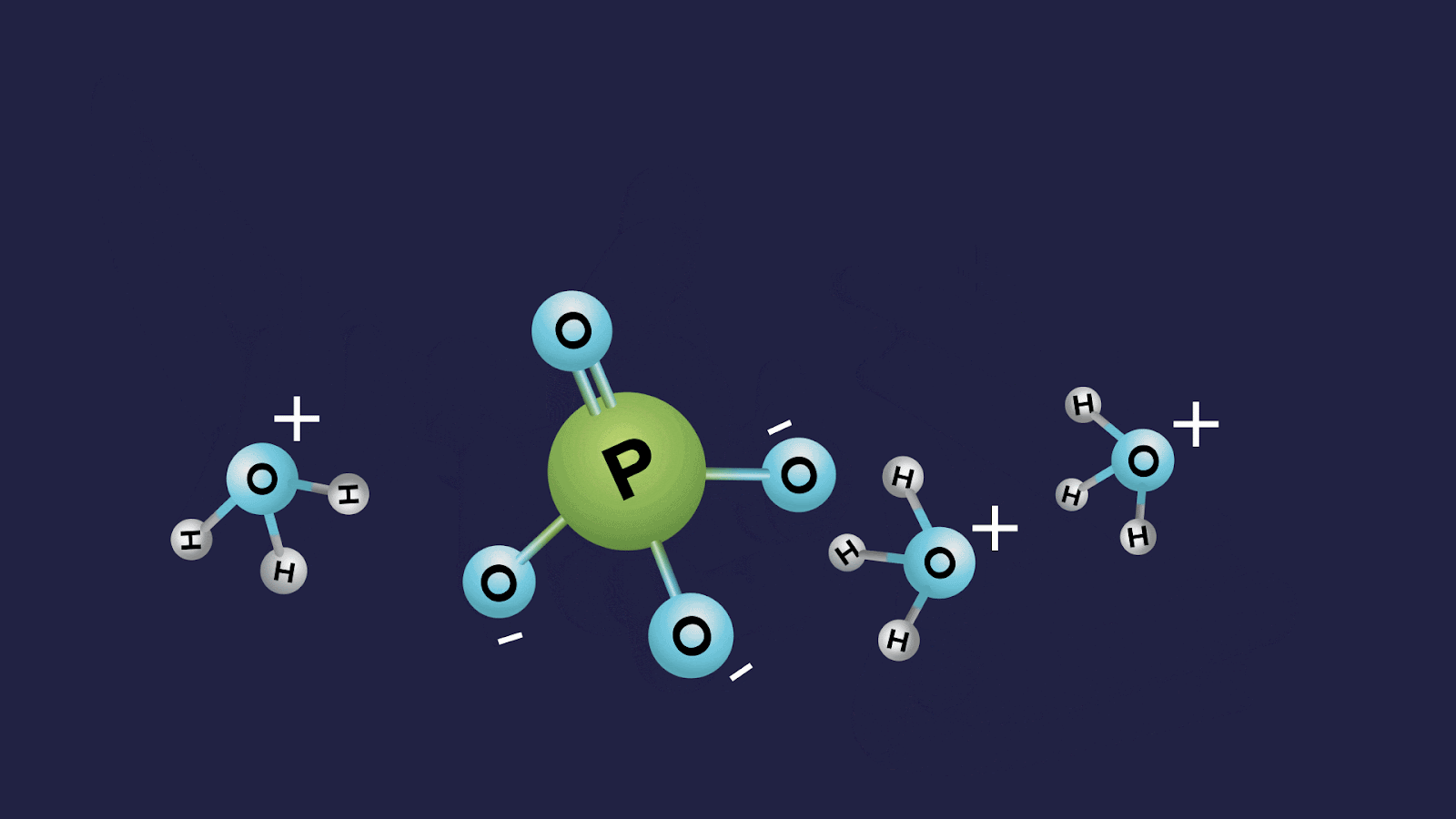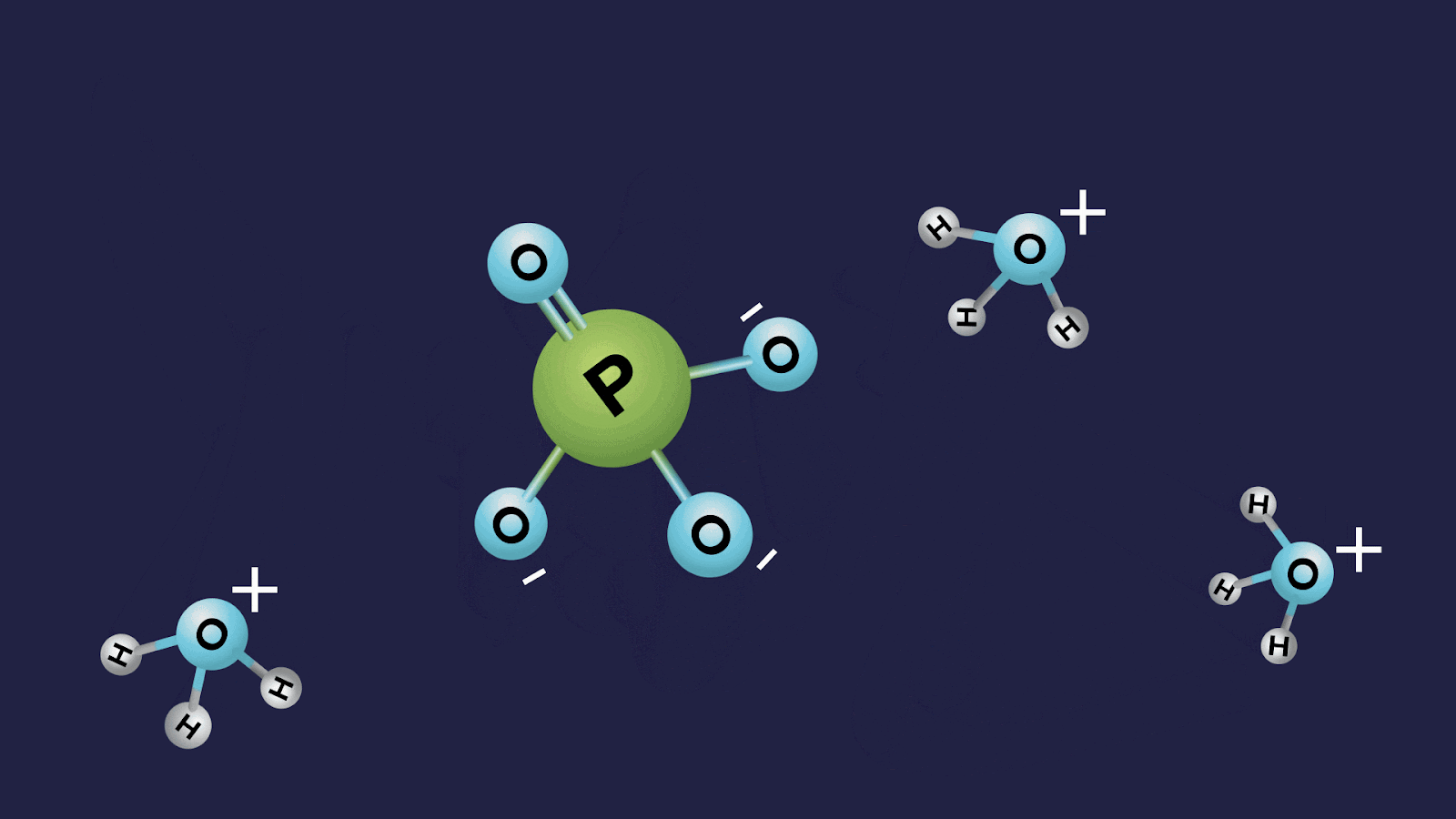
- Orthophosphoric acid (H3PO4):
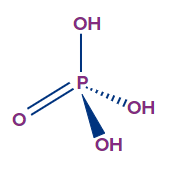
- The oxidation state of phosphorus in orthophosphoric acid is +5, and it is a tribasic acid.
- The number of ionisable or replaceable H+ ions is three (three O-H bonds). So, it is a tribasic acid.
- Peroxomonophosphoric acid (H3PO5):
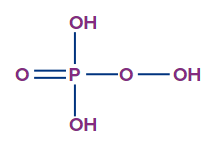
- The oxidation state of phosphorus in peroxomonophosphoric acid is +5, and it is a tribasic acid.
- The number of ionisable or replaceable H+ ions is three (three O-H bonds). So, it is a tribasic acid.
- Metaphosphoric acid (HPO3):
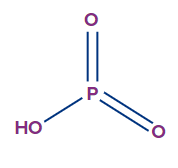
- It exists in the polymeric form.
- The oxidation state of phosphorus in metaphosphoric acid is +5, and it is a monobasic acid.
- The number of ionisable or replaceable H+ ions is one (one O-H bonds). So, it is a monobasic acid.
- Pyrophosphoric acid (H4P2O7):
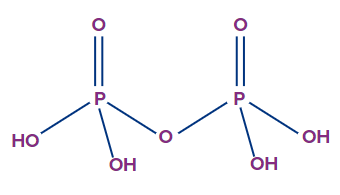
- The oxidation state of phosphorus in pyrophosphoric acid is +5, and it is a tetrabasic acid.
- The number of ionisable or replaceable H+ ions is four (four O-H bonds). So, it is a tetrabasic acid.
- Peroxodiphosphoric acid (H4P2O8):
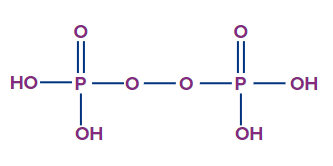
- The oxidation state of phosphorus in peroxodiphosphoric acid is +5, and it is a tetrabasic acid.
- The number of ionisable or replaceable H+ ions is four (four O-H bonds). So, it is a tetrabasic acid.
- Pyrophosphorous acid (H4P2O5):
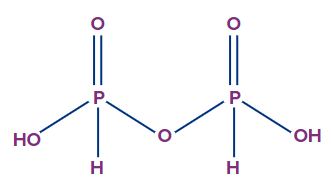
- The oxidation state of phosphorus in pyrophosphorous acid is +3, and it is a dibasic acid.
- The number of ionisable or replaceable H+ ions is two (two O-H bonds). So, it is a dibasic acid.
- Hypophosphoric acid (H4P2O6):
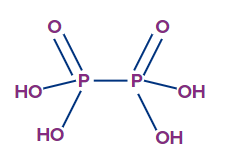
- The oxidation state of phosphorus in hypophosphoric acid is +4, and it is a tetrabasic acid.
- The number of ionisable or replaceable H+ ions is four (four O-H bonds). So, it is a tetrabasic acid.
Practice Problems
1. According to the Arrhenius concept of acids and bases, an acid is a substance that dissociates in water to give __________ ions.
a. H+ (aq)
b. OH- (aq)
c. H+/OH- (aq)
d. None of the above.
Answer: A
Solution: According to Arrhenius’ concept, acids are substances that dissociate in water to given H+(aq) ions, whereases bases are substances taht dissociate in water to given OH- (aq) ions.
So, option A is the correct answer.
2. According to the Bronsted-Lowry concept of acids and bases, a base is a substance that __________ H+ ions.
a. Donates
b. Accepts
c. Donates or accepts
d. None of the above
Answer: B
Solution: According to Bronsted-Lowry's concept of acids and bases, acids are substances that donate H+ ions and bases are the substances that accept H+ ions.
Example:
Base Acid Conjugate Acid (CA) Conjugate Base (CB)
In the reaction given above, H2O acts as a proton donor and NH3 acts as a proton acceptor. So, H2O and NH3 are Bronsted–Lowry acid and base, respectively.
So, option B is the correct answer.
3. Which of the following is a tetrabasic acid?
a. Sulphuric acid
b. Nitric acid
c. Orthophosphoric acid
d. Silicic acid
Answer: D
Solution:
1. Sulphuric acid is a dibasic acid as it has two ionisable H+ ions.

2. Nitric acid is a monobasic acid as it has one ionisable H+ ion.
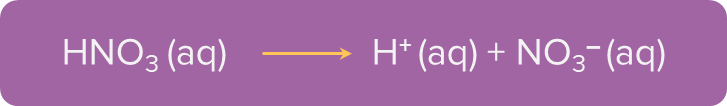
3. Orthophosphoric acid is a tribasic acid as it has three ionisable H+ ions.

4. Silicic acid is a tetrabasic acid as it has four ionisable H+ ions.

So, option D is the correct answer.
4. The basicity of (HPO3)3 is
a. 1
b. 2
c. 3
d. 4
Answer: 3
Solution: The structure of (HPO3)3 is as follows.
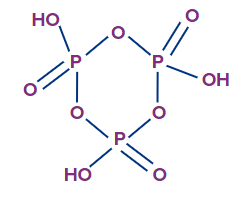
As can be seen from the structure, the number of ionisable H+ ions is 3 (three O-H bonds). So, the basicity of (HPO3)3 is 3.
So, option C is the correct answer.
Frequently Asked Questions – FAQ
1. What is the difference between acidiyt and basicity?
Answer: The terms acidity and basicity are applicable to bases and acids, respectively, and not the other way around. The number of hydroxyl ions that a basic molecule can produce in an aqueous solution is what is known as the acidity of bases. The number of hydronium ions that a substance can produce in an aqueous solution is what is known as the basicity of acids.
2. What is the difference between basicity and basic strength?
Answer: The number of hydrogen ions that can be created by one acid molecule determines the basicity of an acid. On the other hand, a base's basic strength is determined by how quickly it releases OH- ions when dissolved in water.
3. What is the basicity of peroxydisulphuric acid?
Answer: The structure of perxoydisulphuric acid is as follows.
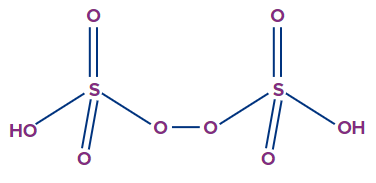
From the structure, it can be observed that 2 ionisable H+ ions are present in the molecule. Therefore, the basicity of peroxydisulphuric acid is 2.
4. What is the basicity of dithionic acid?
Answer: The structure of dithionic acid is as follows.
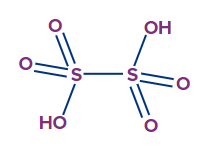
From the structure, it can be observed that 2 ionisable H+ ions are present in the molecule. Therefore, the basicity of dithionic acid is 2.


