-
Call Now
1800-102-2727
Balancing of Redox Reactions - Introduction to Redox Reactions, Need of Balancing a Redox Reaction, Half Reaction Method & Oxidation Number Method
Have you ever seen the manual weighing balance? I think you do! When you go to a shop to buy some groceries like rice, pulses etc they use a weighing balance to calculate the weight.

You must have observed that the shopkeeper put weight on one side of the balance and the goods you want to buy on the other and try to add or remove the goods till the balance becomes equal.
Any guesses, about why we are discussing this and how is it relevant here?
Similar to the weighing balance which helps in balancing the material goods. In the case of redox reactions, the reactions are balanced by equating the number of atoms and charges on both sides of the reaction.
Let’s learn in this article the importance and the methods to balance redox reactions.
Table of Contents
- Introduction to Redox Reactions
- Need for Balancing Redox Reactions
- Methods of Balancing a Redox Reaction
- Half Reaction Method
- Oxidation Number Method
- Practice Problems
- Frequently Asked Questions-FAQs
Introduction to Redox Reaction
Redox reactions are oxidation-reduction chemical reactions in which the reactants are getting oxidised and reduced simultaneously. The term ‘redox' is an abbreviation for reduction oxidation.
In a chemical reaction, the substance being reduced is known as the oxidizing agent, while the substance being oxidized is known as the reducing agent.
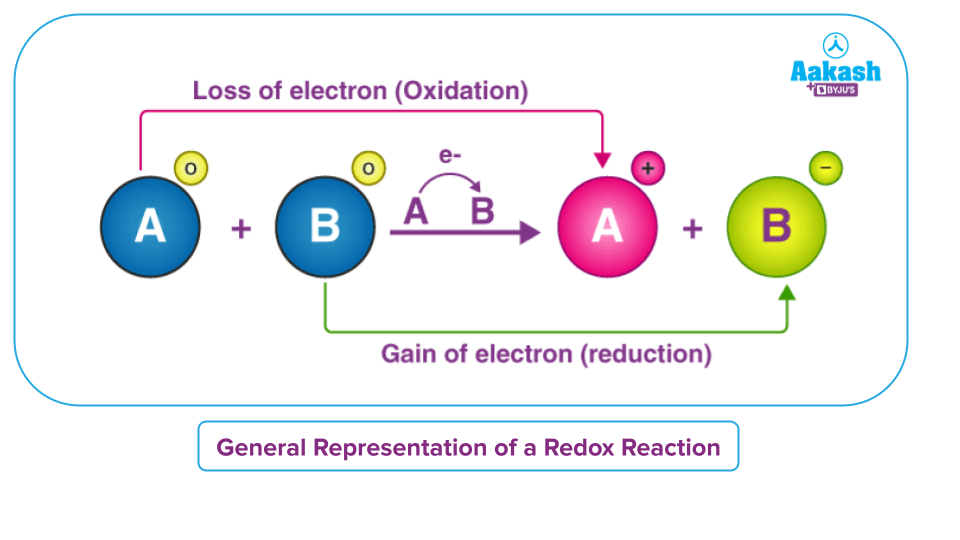
Let us consider an example in which two reactants A and B react to undergo a reaction in which the reactant ‘A’ loses an electron to undergo oxidation and the reactant ‘B’ gains an electron to undergo reduction simultaneously. Since both oxidation and reduction are taking place in the reaction simultaneously, hence it is a redox reaction.
Need for Balancing Redox Reactions
As we know, chemical reactions need to be balanced to obey the law of conservation of the mass. Similarly, in the case of redox reactions, atoms need to be balanced such that the conservation of mass is applied in the redox reaction.
In the case of a redox reaction, the number of atoms as well as the charge needs to be balanced because it needs to satisfy two properties during the course of the reaction:
- Mass conservation: Mass is conserved in a chemical reaction to satisfy the law of conservation of mass which can be done by balancing the number of atoms on the reactant and product side of the reaction.
- Charge conservation: In the case of redox reactions, the exchange of electrons takes place between the element undergoing the oxidation and reduction but the total charge on the reactants and products must be equal.
Methods of Balancing a Redox Reaction
Two methods are used to balance a redox reaction:
- Oxidation number method: This method is based on the change in the oxidation number of the reducing agent and the oxidising agent followed by balancing the elements in which the oxidation state does not change by the normal hit and trial method.
- Half reaction method: Also called ion-electron method. It is based on splitting the redox reaction into two half-reactions. One involves oxidation known as oxidation half-reaction and the other involves reduction known as reduction half-reaction. The half-reactions are balanced separately, merged to obtain the originally given redox reaction, and the elements in which the oxidation state does not change are balanced by a normal hit and trial method.
In writing equations for oxidation-reduction reactions, just as for other reactions, the compositions and formulas must be known for the substances that react and for the products that are formed failing which it is very difficult to balance the redox reaction by either of the method.
Note: Both these methods are in use and the choice of their use rests with the individual using them.
Half Reaction Method
Balancing a redox reaction by half-reaction method in an acidic medium:
Let’s see the rules for balancing a redox reaction by half-reaction method in an acidic medium.
Consider the following reaction, which occurs in an acidic medium.
MnO4-(aq)+SO2(g) Mn2+(aq)+HSO4-(aq)
Step 1: Using the oxidation state of the element present in the reactant and product side, identify the atoms that are oxidised and reduced.
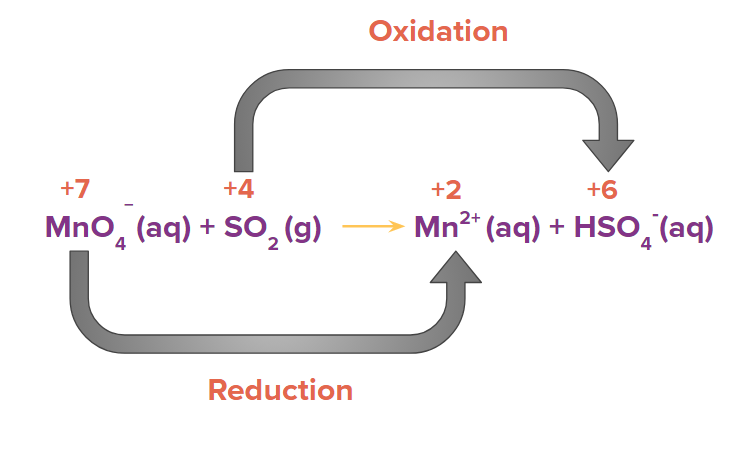
Step 2: Write the two half reactions separately. The reaction in which oxidation takes place is known as oxidation half reaction and the reaction in which reduction takes place is known as reduction half-reaction.
Oxidation half-reaction:

Reduction half-reaction:

Step 3: Equate the number of atoms present in the half-reaction which undergoes oxidation or reduction by multiplying it with suitable factors such that the number of atoms becomes equal. In this case, the atoms are already balanced.
Oxidation half-reaction:

Reduction half-reaction:

Step 4: Multiply the half reactions with a suitable factor such that the number of electrons gained or lost becomes equal.
Oxidation half-reaction:
This reaction is multiplied by a factor of 5.

Reduction half-reaction:
This reaction is multiplied by a factor of 2.

Step 5: Add the half reactions to obtain the original redox reaction.
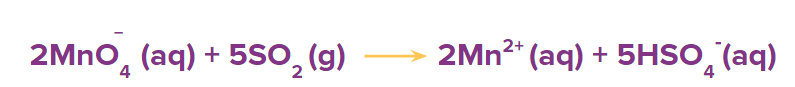
Step 6: Balance the oxygen atoms by adding the H2O molecules at the oxygen deficient side of the reaction.
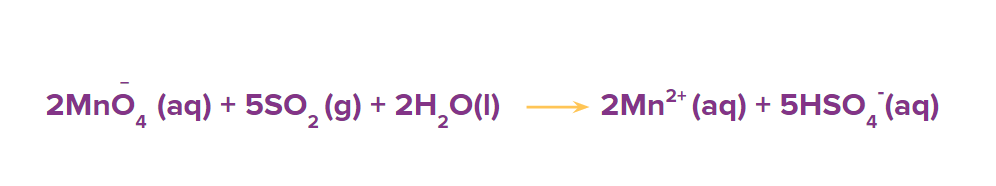
Step 7: To balance the hydrogen atoms, add H+ ions to that side of the reaction which is deficient in the hydrogen atoms.

The above equation is the balanced redox reaction.
Balancing a redox reaction by half-reaction method in a basic medium:
Let’s see the rules for balancing a redox reaction by half reaction method in a basic medium.
Consider the following reaction, which occurs in a basic medium.
Al (s)+NO3-(aq) → Al(OH)4-(aq)+NH3 (g)
Step 1: Using the oxidation state of the element present in the reactant and product side, identify the atoms that are oxidised and reduced.
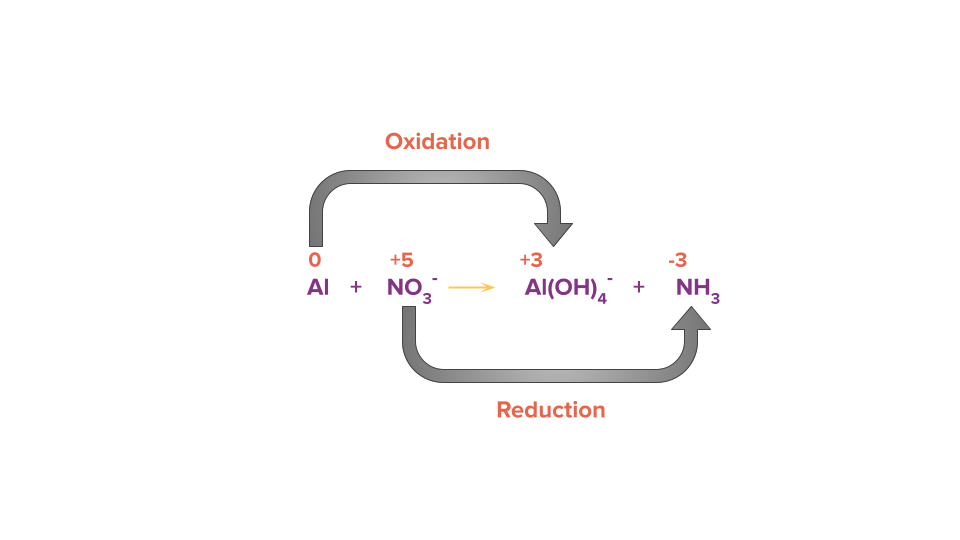
Step 2: Write the two half reactions separately. The reaction in which oxidation takes place is known as oxidation half reaction and the reaction in which reduction takes place is known as reduction half-reaction.
Oxidation half-reaction:

Reduction half-reaction:

Step 3: Equate the number of atoms present in the half-reaction which undergoes oxidation or reduction by multiplying it with suitable factors such that the number of atoms becomes equal. In this case, the atoms are already balanced.
Oxidation half-reaction:

Reduction half-reaction:

Step 4: Multiply the half reactions with a suitable factor such that the number of electrons gained or lost becomes equal.
Oxidation half-reaction:
This reaction is multiplied by a factor of 8.

Reduction half-reaction:
This reaction is multiplied by a factor of 3.

Step 5: Add the half-reactions to obtain the original redox reaction.

Step 6: Balance the oxygen atoms by adding the H2O molecules at the oxygen deficient side of the reaction.

Step 7: To balance the hydrogen atoms, add H+ ions to that side of the reaction which is deficient in the hydrogen atoms.

Step 8: Since the reaction is taking place in the basic medium, add the same number of OH- ions to both sides of the reaction which is equal to the H+ ions that were added in the previous step.

The final reaction will be:

Oxidation Number Method
Balancing a redox reaction by oxidation number method in acidic medium:
Let’s see the rules for balancing a redox reaction by oxidation number method in an acidic medium:
Consider the following reaction, which occurs in an acidic medium.
Cu(s)+NO3-(aq) → Cu2+(aq)+NO (g)
Step 1: Using the oxidation state of the element present in the reactant and product side, identify the atoms that are oxidised and reduced.
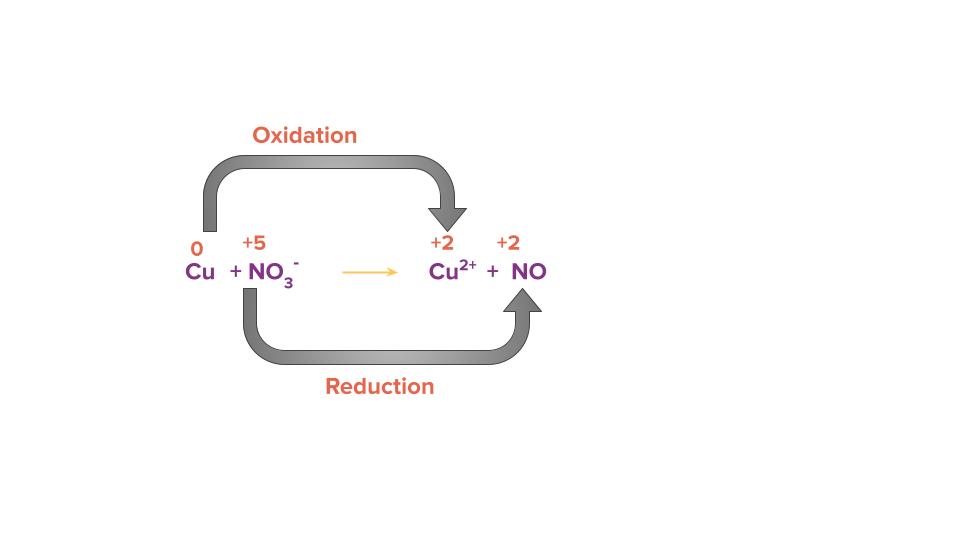
Step 2: Calculate the change in electrons (i.e., loss or gain) in the identified oxidising and reducing species.
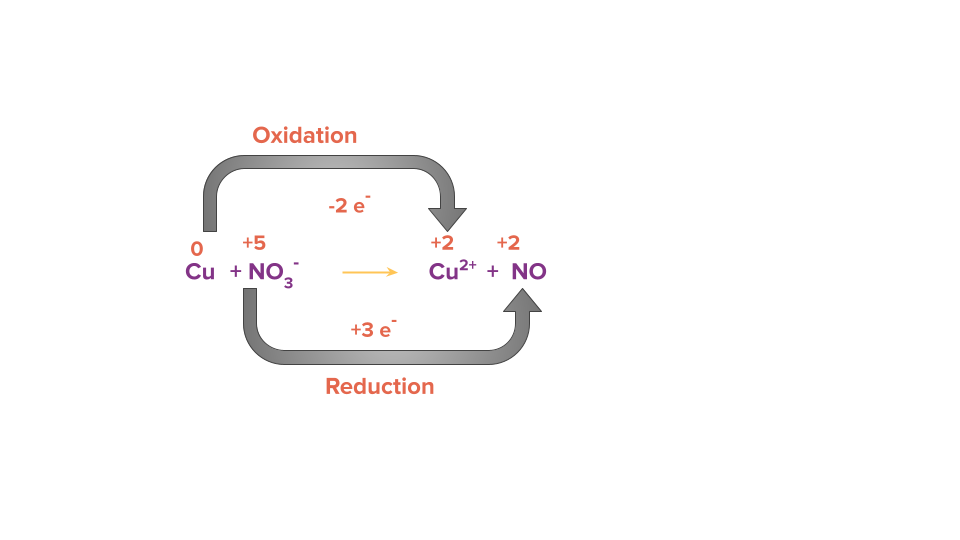
Step 3: Equate the number of electrons gained and lost in the given reaction by multiplying it by a suitable factor.
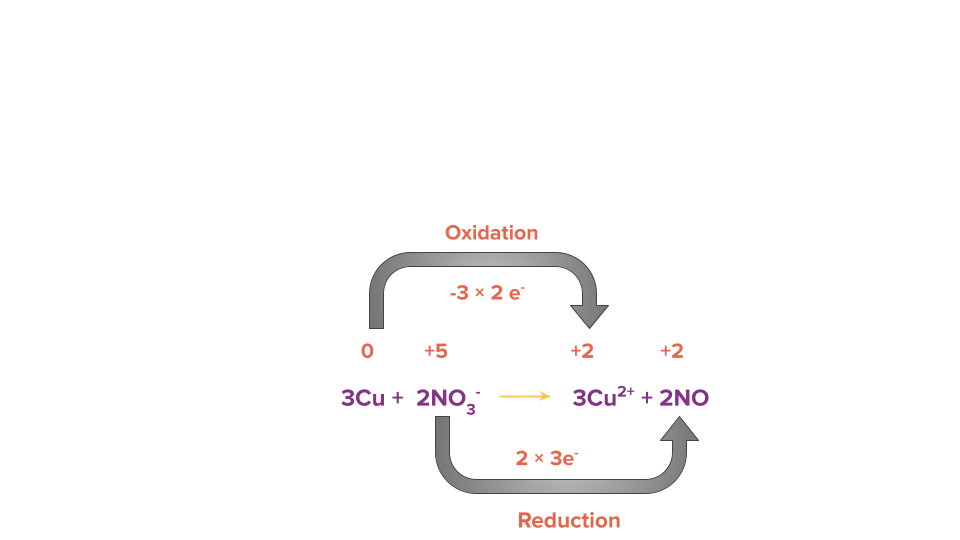
Step 4: Balance all other atoms except H and O by the hit and trial method but in this case all atoms are already balanced.

Step 5: Balance the oxygen atoms by adding the H2O molecules at the oxygen deficient side of the reaction.

Step 6: To balance the hydrogen atoms, add H+ ions to that side of the reaction which is deficient in the hydrogen atoms.

Balancing a redox reaction by oxidation number method in a basic medium:
Let’s see the rules for balancing a redox reaction by oxidation number method in a basic medium.
Consider the following reaction, which occurs in a basic medium.
Cl 2+IO3-IO4-+Cl-
Step 1: Using the oxidation state of the element present in the reactant and product side, identify the atoms that are oxidised and reduced.
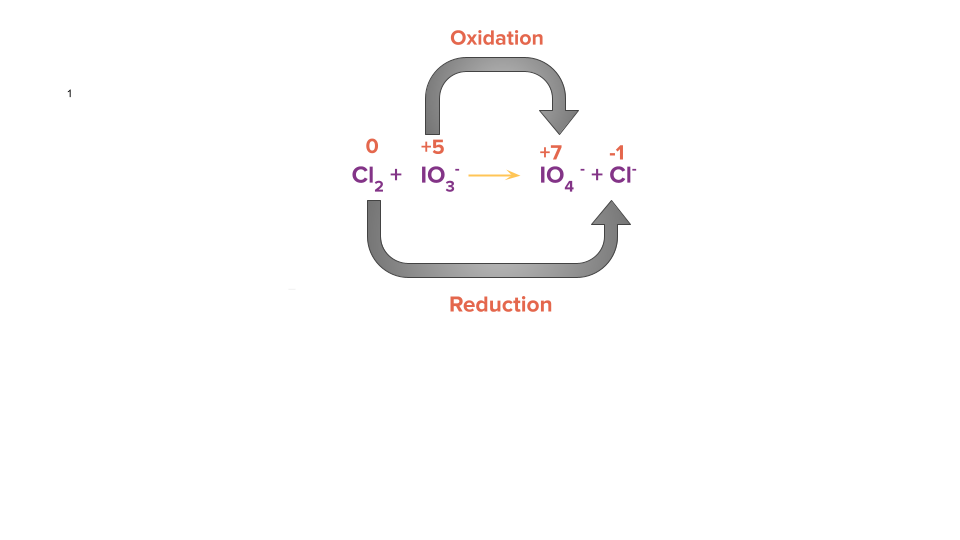
Step 2: Calculate the change in electrons (i.e., loss or gain) in the identified oxidising and reducing species.
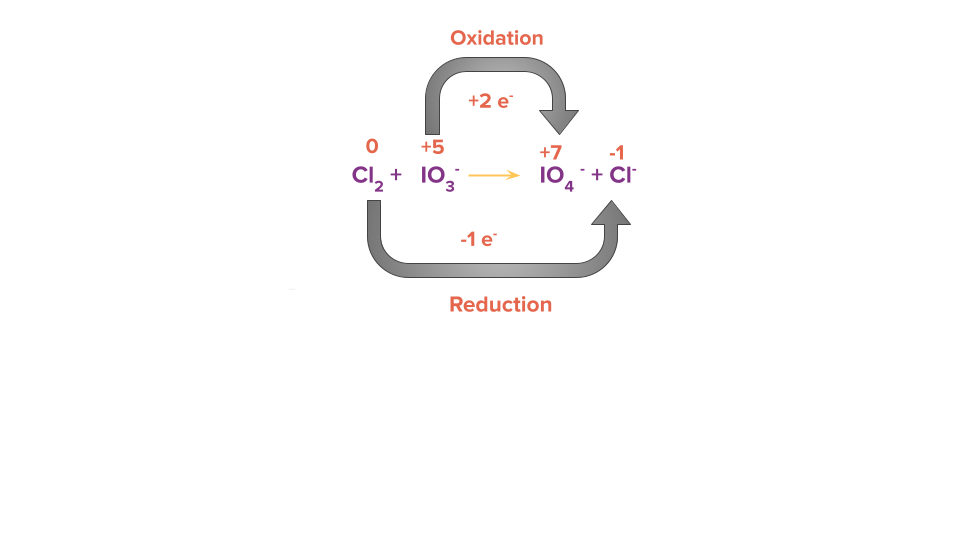
Step 3: Equate the number of electrons gained and lost in the given reaction by multiplying it by a suitable factor.
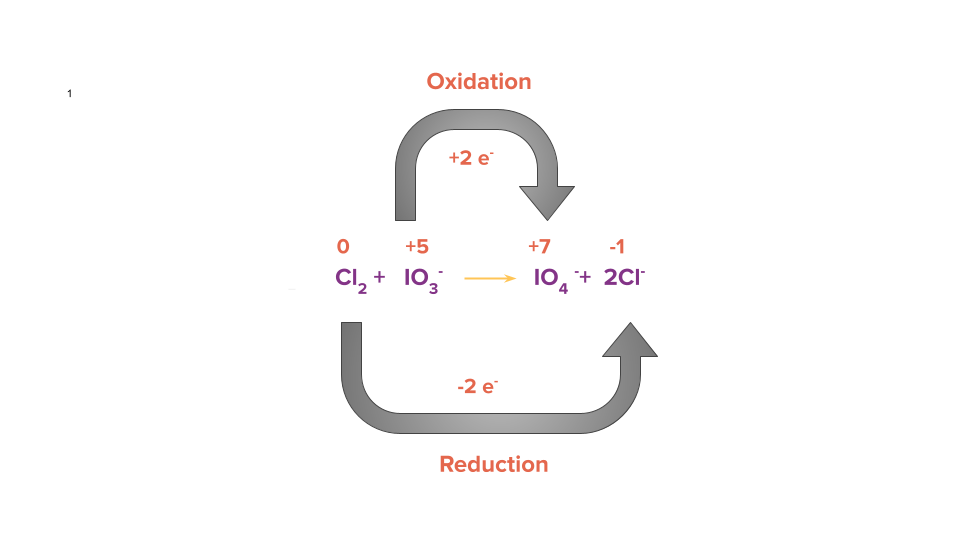
Step 4: Balance all other atoms except H and O by the hit and trial method but in this case all atoms are already balanced.

Step 5: Balance the oxygen atoms by adding the H2O molecules at the oxygen-deficient side of the reaction.

Step 6: To balance the hydrogen atoms, add H+ ions to that side of the reaction which is deficient in the hydrogen atoms.

Step 7: Since the reaction is taking place in the basic medium, add the same number of OH- ions to both sides of the reaction which is equal to the H+ ions that were added in the previous step.

Therefore, the final equation becomes:

Practice Problems
Q. Find the value of a/b in the given reaction. (In acidic medium)
Answer: (A)
Solution:
Step 1:
Oxidation half reaction:
Reduction half reaction:
Step 2:
First balance Cr atom;
Step 3: Balance O atom by adding H2O
Step 4: Now, balance H-atom.
Step 5: Now, balance charge by adding e-.
Step 6: Multiply eq (i) by 6
Step 7: Add equation (ii) and (iii)
Comparing equation (iii) and equation (iv) we get,
a=6, b=7
Therefore ab is equal to (67)
Q. Find coefficients of reaction in basic medium.
Solution:
According to the given question,
Step 1:
Oxidation half reaction:
Oxidation state of manganese changes from (+2 to +4 ).
Reduction half reaction: ClO3-ClO2
Oxidation state of chlorine changes from (+5 to +4).
Step 2: Equate the number of atoms present in the half-reaction which undergoes oxidation or reduction by multiplying it with suitable factors such that the number of atoms becomes equal. In this case, the number of atoms is already balanced.
Oxidation half-reaction:
Reduction half-reaction:
e-+ClO3-ClO2
Step 3: Multiply the half-reactions with a suitable factor such that the number of electrons gained or loss becomes equal.
Oxidation half-reaction:
Reduction half-reaction:
2e-+2ClO3-2ClO2
Step 4: Add the half-reactions to obtain the original redox reaction.
Step 5: Balance the oxygen atoms by adding the H2O molecules at the oxygen-deficient side of the reaction. In this case, oxygen atoms are already balanced.
Step 6: To balance the hydrogen atoms, add H+ ions to that side of the reaction which is deficient in the hydrogen atoms. In this case, there are no hydrogen atoms.
Step 7: Since the reaction is taking place in the basic medium, add the same number of OH- ions to both sides of the reaction which is equal to the H+ ions that were added in the previous step. In this case, there are no (H+) ions.
Therefore, the final reaction will be:
Comparing equations (i) and (ii), we get;
a = 1, b = 2, c = 1
Q 3. Find coefficients of the reaction a H2O2+b I2c HIO3 +d H2O.
- a=3, b=3, c=2, d=4
- a=5, b=3, c=2, d=4
- a=5, b=1, c=2, d=4
- a=5, b=2, c=2, d=4
Answer: (C)
Solution:
Step 1:
Oxidation half reaction: I2 HIO3
Oxidation state of iodine changes from 0 to +5
Reduction half reaction: H2O2 H2O
Oxidation state of oxygen changes from -1 to -2
Step 2:
Balancing reduction half reaction:
H2O2 H2O
Balance H- atoms by adding H+ions.
Balance charge, by adding e-.
Step 3: Balancing oxidation half reaction:
I2 HIO3
Balancing the iodine atoms.
I2 2HIO3
Now, balancing the O - atoms.
I2+ 6H2O 2HIO3
Now, balancing the H atoms.
I2+ 6H2O 2HIO3+10H+
Balancing the charge by adding electrons at the product side.
Step 4: Multiply eq (i) by 5
Step 5: Adding equation (ii) and (iii)
I2+ 6H2O+5H2O2+10H+ 2HIO3+10H++ 10H2O
I2+5H2O2 2HIO3+ 4H2O
The final redox equation is:
5H2O2+ I2 2HIO3+ 4H2O
Comparing above equation with a H2O2+b I2c HIO3 +d H2O
We get;
a=5, b=1, c=2, d=4
So, option C is the correct answer.
Q 4. Which of the following redox reactions is not balanced?
- CaCO3(s)CaO(s)+ CO2(g)
- Na(s)+Cl2(g)NaCl(s)
- NaH(s)Na(s)+H2(g)
- Both B and C
Answer: (D)
Solution:
All decomposition reactions are not the redox reactions.
CaCO3CaO+ CO2
|
Element |
Initial O.S |
Final O.S |
|
Ca |
+2 |
+2 |
|
O |
-2 |
-2 |
|
C |
+4 |
+4 |
No change in the oxidation state of atoms involved in the reaction.
Na(s)+Cl2(g)NaCl(s) and NaH(s)Na(s)+H2(g) are redox reactions but they are not balanced because the number of atoms is not the same in both the reactant and product side.
Frequently Asked Questions-FAQs
Q. How to define oxidation and reduction according to the classical concept?
Answer: According to the classical concept, oxidation is defined as the gain of oxygen to the substance or loss of hydrogen from the substance. Whereas reduction is defined as the gain of hydrogen to the substance or loss of oxygen from the substance.
For example: Let's consider a reaction.
4Na+O2→ 2Na2O
In the above reaction sodium is undergoing oxidation as it gains oxygen to form sodium oxide.
Considering other reactions where oxygen is removed from the compound, the process is known as reduction.
2CaO → 2Ca+O2
Q. What is oxidation half reaction in a redox reaction?
Answer: Redox reaction is the type of reaction which undergoes both oxidation and reduction simultaneously. Oxidation half reaction refers to the type of reaction in which the reactant of the redox reaction undergoes oxidation and this half reaction is written separately from a redox reaction.
Q. How to define oxidation and reduction according to the electronic concept?
Answer: According to the electronic concept of oxidation and reduction the gain of electrons is known as reduction whereas if an element loses electrons it is said to undergo oxidation.
For example:
4Na+O2→ 2Na2O; here in reactants sodium is in 0 oxidation state and in the product, sodium is in +1 oxidation state. So, sodium in this reaction loses 1 electron and gets oxidised.
2CaO → 2Ca+O2; here in reactants calcium is in +2 oxidation state and in products, calcium is in 0 oxidation state. So, calcium in this reaction gains 2 electrons and gets reduced.
Q. What is the oxidation state of an element? How does it help in determining the oxidation and reduction process?
Answer: It is a real or imaginary charge developed on an atom when it goes from its most stable elemental free state to the combined state. When the oxidation state of the element increases it undergoes oxidation and when the decrease in oxidation state takes place it is said to undergo reduction. Therefore oxidation state or oxidation number help in determining whether the element is undergoing oxidation or reduction.


