-
Call Now
1800-102-2727
Atomic Radii - Factors Affecting Atomic Radii, Various Types of Atomic Radii, Practice Problems and FAQ
Do you know where the beauty of a jungle lies?
In the diversity and vividness of its inhabitants!
We get to see animals of various sizes, shapes and characters. Some are herbivorous whereas some are carnivorous. Some animals are too small to fall easy prey to predators like tigers, whereas some, like elephants, owing to their large size tend to escape easily from the fiery carnivores.
In the wild, size matters!

So is the case with elements. Their chemical and physical properties depend on how their atoms interact, which in turn is closely related to their respective atomic size.
As a result, atomic size is a crucial component of an element's atom that must be thoroughly examined in order to comprehend the various properties of elements.
Atomic Radii is defined as the distance between the centre of the nucleus and the outermost electron of an atom. It corresponds to half the distance between adjacent atoms of the same element in a molecule.
TABLE OF CONTENTS
- Complications Involved in Finding the Size of an Atom
- Factors Affecting Atomic Radii
- How to Estimate Atomic Radii
- Various Types of Atomic Radii
- Covalent Radii
- Metallic Radii
- Van der Waals Radii
- Trends Across Periodic Table
- Practice Problems
- Frequently Asked Questions - FAQ
Complications Involved in Finding the Size of an Atom
Finding the size of an atom is indeed a complicated process because of the following facts:
- The size of atoms (10-10m in radius) is very small.
- There is no sharp boundary, where the outermost electron exists. Since the electron cloud surrounding the atom does not have a sharp boundary, the determination of the atomic size cannot be precise.
- Hence, we estimate atomic radii in combined states only.
Factors affecting Atomic Radii
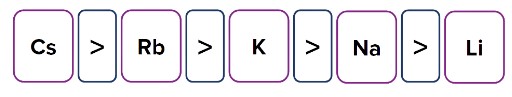
- Number of shells
Atomic radius is directly proportional to the number of shells.
Example:
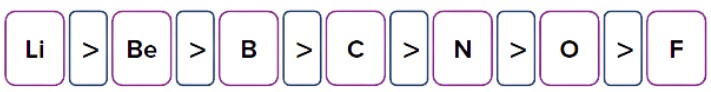
- Effective Nuclear Charge
Atomic radius is inversely proportional to effective nuclear charge due to an increase in the attractive force on the outermost electrons.
Example:
- Screening Effect
In multi-electron atoms, the outermost electrons are shielded or screened from the nucleus by the inner electrons. This is called the shielding or screening effect.
The order of shielding is s>p>d>f.
As a result, the outermost electrons do not feel the full impact/pull of the nucleus. The actual charge felt by electrons is termed an effective nuclear charge. Hence, with an increase in shielding effect, atomic radius increases.
- Number of Bonds
The covalent radius depends on the number of bonds. With the increase in several covalent bonds, atomic radius decreases. Hence, atomic radius is inversely proportional to the number of bonds.
How to estimate the atomic radius
One possible way to find the atomic radius is to use the internuclear distance between the bonded identical atoms. Also, by X-ray diffraction or other spectroscopic techniques, it can be determined.

Various Types of Atomic Radii
Covalent Radii
- Covalent radius is one-half of the distance between the centres of two nuclei (of like atoms) bonded by a single covalent bond.
- Covalent radius is generally used for non-metals.
- Unit: Picometre (pm) or Angstrom (Å)

Metallic Radii
- Metallic radii correspond to half the internuclear distance separating the metal cores in the metallic crystal having metallic nature.
- Example: The distance between two adjacent copper atoms in solid copper is 256 pm. Hence, the metallic radii of copper is assigned a value of 128 pm.
- We use the term atomic radius to refer to both covalent and metallic radius depending upon whether the element is a non-metal or a metal.
Difference between covalent and metallic radii
|
Covalent radius |
Metallic radius |
|
Half of the distance between the centres of two nuclei (of identical atoms) bonded by a single covalent bond. |
Half the internuclear distance separating the metal cores in the metallic crystal. |
|
Overlapping of two half-filled atomic orbitals. |
No overlapping despite the atoms being in contact. |
|
Smallest of all radii. |
Greater than covalent radii. |
Van der Waals Radii
- For noble gases, it is half the distance between the nuclei of two non-bonded nearest neighbouring atoms of the same element in its solid state.

- For non-metals, van der Waals radii is half the distance between the nuclei of two non-bonded nearest neighbouring atoms of two adjacent molecules of the same element in a solid state.

- van der Waals radius does not apply to metals. Its magnitude depends upon the packing of the atom when the element is in the solid state.
Trends across Periodic Table
In the periodic table, trends are seen either from top to bottom (down the group) or from left to
right (across the period).
Down the group,
- Atomic radii in a particular group increases from top to bottom.
- Down the group, the principal quantum number (n) increases and the valence electrons are farther from the nucleus.
- The inner energy levels are fully filled with electrons, which provide a shielding effect to the outer electrons from the pull of the nucleus. Thus, the size increases.

Across the period,
- The atomic radii generally decreases across a period.
- It is because, within the period, the outer electrons are in the same valence shell and the effective nuclear charge increases.
- As the atomic number increases, the attraction of electrons to the nucleus increases. Therefore, the size decreases.
Practice Problems
Q 1. Among lithium, beryllium, and boron, which will have the largest atomic radius?
a. Lithium
b. Beryllium
c. Boron
d. All are equal
Answer: A)
Lithium has the largest atomic radius of 167 pm among the given elements. The atomic radii of beryllium and boron are respectively 112 pm and 87 pm. This is in accordance with the fact that atomic size decreases on moving from left to right across a period.
Q 2. Among sodium, magnesium, and aluminium which will have the largest atomic radius?
a. Magnesium
b. Sodium
c. Aluminium
d. All are equal
Answer: B)
Generally, the atomic radius decreases from left to right across a period. Sodium lies on the leftmost side of group 3. So it has the largest radii in its corresponding period.
Q 3. Among phosphorus, antimony, and bismuth, which will have the largest atomic radius?
a. Phosphorus
b. Antimony
c. Bismuth
d. All are equal
Answer: C)
Atomic size increases on going down the group due to the addition of shells. Hence, bismuth will have the largest size among the given elements.
Q 4. Which element has the smallest atomic radius?
a. Aluminium
b. Gallium
c. Thallium
d. Boron
Answer: D)
The size of atoms increases down the group. So, out of these elements of Group 13, boron will have the smallest size as it is the first element in its group.
Frequently Asked Questions - FAQ
Q 1.What is the order of covalent, metallic and van der Waals radii?
Answer: For the same species, Van der Waals radius > Metallic radius > Covalent radius`
Q 2. Atomic size decreases along a period, but the size of group 18 elements is observed to be the largest in the period. Why?
Answer: The size of group 18 elements is observed to be the largest in the period because the size is determined in terms of van der Waals radius (exists in a monatomic state) that is generally larger than the covalent (or metallic) radius. In fact, the radii of noble gases should be compared not with the covalent radii but with the van der Waals radii of the other elements. Inert gases are monatomic. So, the covalent radii and metallic radii will not be applicable for them. Therefore, for inert gases, van der Waals radius is used to determine size.
Q 3. Why is the van der Waals radius always greater than the covalent radius?
Answer: The van der Waals forces of attraction are weak. Therefore, the internuclear distance in the case of atoms held by van der Waals forces is much larger than those between covalently bonded atoms. Since a covalent bond is formed by the overlap of two half-filled atomic orbitals, a part of the electron cloud becomes common. Therefore, covalent radii are always smaller than the van der Waal radius.
Q 4. Are the atomic size and atomic radius the same?
Answer: The distance between an atom’s nucleus and its outermost shell is measured in atomic size. The atomic radius is defined as the shortest distance between the nuclei of an atom and the atom’s outermost shell in basic chemistry.
Related Topics
|
Ionic Radii |
Electronegativity |
|
Factors affecting electron gain enthalpy |
Successive Ionization Enthalpy |
|
Ionization Enthalpy |
Electron Gain Enthalpy |




