-
Call Now
1800-102-2727
Atomic Number - Introduction, Examples, Practice Problems & FAQs
A person's unique identity is represented by their Aadhar card number, PAN number, passport number, driving license number etc. Similarly, what, in your opinion, is an element's most distinct identity?
For a long time, the concept of atomic mass dominated research; many attempts to build the periodic table based on atomic mass were made, but with the discovery of x-rays, electrons, and nuclei, Henry Mosley bombarded several metals with a beam of electrons in an experiment. The x-ray spectrum produced by each element is determined by the position of hydrogen in the list of elements leading up to uranium. He established a link between wavelength and the number of atoms in an element. He predicted the atomic numbers of a few missing elements. As a result, the atomic number, not the atomic mass, determines the element's identity.
“Moseley’s law was one of the greatest advances yet made in natural philosophy.” - de Broglie

Table of Contents
- General representation of an element
- Atomic number
- Discovery of atomic number
- Practice problems
- Frequently asked questions-FAQs
What is an Atomic Number?
The atomic number of an element is the number of protons present in the nucleus. An atomic number is used to differentiate one element from another.
Carbon has 6 protons in its nucleus and its atomic number is 6.
Oxygen has 8 protons in its nucleus and its atomic number is 8.
General Representation of an Element
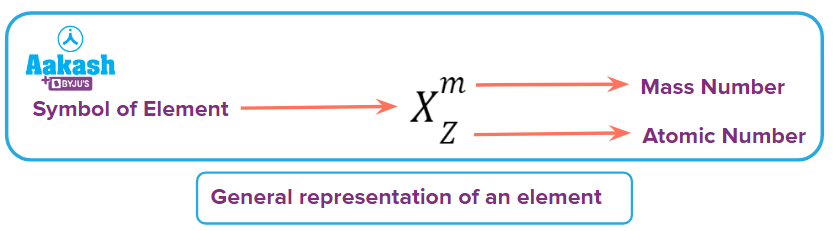
For example:
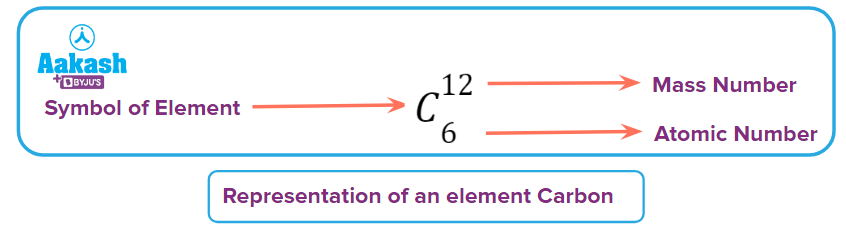
A chemical element is defined by the number of protons in its nucleus (its "atomic number," or Z). An element is defined entirely by its atomic number (Z), atoms of the same chemical element might have different numbers of neutrons. Isotopes are subspecies of chemical elements with the same Z value but different mass number (m). As number of neutrons in the isotopes would be different.
Discovery of Atomic Number
Including Dalton’s atomic theory to Rutherford atomic model, Dobernier’s triad to Mendeleev’s periodic tables all are based on the notion of atomic mass.
Henry Mosley bombarded different metals with a beam of electrons. The frequency of the produced x-ray from the different elements was plotted with their corresponding atomic number. A straight line is obtained between the square root of the frequency ( of produced x-ray and atomic number (Z).
Mathematical form,
Where, v= frequency of emitted x - ray
Z= atomic number of the element
a & b are constants.

Mosley confirmed that the atomic number is the number of positive charges in the atomic nucleus. He also discovered some unknown elements, with atomic numbers 43, 61, and 75. After the contribution of Mosley, the entire vacancy of the periodic table was filled.
Practice Problems
Q 1. Classification of elements in modern periodic table is based on the:
A. Atomic mass
B. Atomic number
C. Number of protons
D. All of these
Answer: (B)
Solution: Elements in the modern periodic table are arranged on the basis of their increasing atomic number. Arranged from Hydrogen (1) tor Ognesson (118).
Q 2. The total number of protons present in one molecule of CO2 is:
A. 22
B. 40
C. 44
D. 60
Answer: (A)
Solution:
Number of protons in one atom of carbon = 6
Number of protons in one atom of oxygen = 8
So, total number of protons in one molecule of CO2 = = 22
The correct answer is option (A).
Q 3. The number of electrons in dispositive copper ion is:
A. 64
B. 67
C. 31
D. 27
Answer: (D)
Solution:
The atomic number of Cu is 29. In a neutral atom, the number of protons = number of electrons.
Cu2+ ioncontains 29 protons but will have 27 electrons (29 - 2 = 27) as there is two units of positive charge on copper atom. So, the correct answer is option (D).
Q 4. The total number of protons present in one mole of NH3 molecules is:
[NA refers to Avogadro's number]
Answer: (B)
Solution:
Number of protons per atom of
Number of protons per atom of
Number of protons per molecule of NH3=
Number of protons in one mole of NH3 molecules
So, the correct answer is option (B).
Frequently Asked Questions-FAQs
Q1. Who was the assassin who killed Henry Moseley?
Answer: At the battle of Suvla Bay, he was hit in the head by a Turkish sniper (in Turkey). At the age of 27, he died, leaving the world of one of the most promising experimental physicists.
Q2. Why can an atomic number never be a fraction?
Answer: Atomic number is the number of protons present in an atom. The number of protons can never be in fraction. The mass number of any atom is also a whole number.
Q3. How many atomic numbers are discovered till date?
Answer: The last element of our modern periodic table is Uuo having atomic number 118. It is also called Oganesson (this element was discovered by a nuclear physicist Yuri Oganessian).
Q 4. What are isotopes and isobars, and what are some examples?
Answer: Isotopes are elements that share the same atomic number but have a different mass number. Isobars, on the other hand, are atoms with the same mass number but a different atomic number. Protium (one proton and no neutrons), deuterium (one proton and one neutron), and tritium (one proton and two neutrons) are the examples of hydrogen isotope. Because Argon, Potassium, and Calcium have the same mass numbers but distinct atomic numbers, they are isobars.


