-
Call Now
1800-102-2727
Associated Colloids: Definition of Colloid, Types, Associated Colloid, Micelle, Characteristics, Practice Problems & Frequently Asked Questions
Smoke particles are colloidal carbon particles with an electrical charge that are present in the atmosphere.
Do you know how these smoke particles (colloidal particles) are removed from the air?

The Cottrell precipitator, which is based on the electrophoresis concept, is used to precipitate smoke particles. A chamber consisting of successive metal plates connected to a high-potential source by a metal wire can permit the passage of smoke. When charged smoke particles are brought to an electrode with an opposing charge, the heated air escapes through the vent. This process also gets rid of dust particles. Therefore, in significant industrial locations, smoke nuisance can be avoided.
We will learn more about colloids and their features on this topic page, with a focus on associated colloids.
Table of Contents
- Definition of Colloid
- Types of Colloid
- Associated Colloid
- Micelle
- Characteristics of Associated Colloids
- Practice Problems
- Frequently Asked Questions(FAQs)
Definition of Colloid:
A colloid is a form of combination existing in between completely soluble and insoluble mixture. It is a suspension that neither settles down nor can be filtered of suspended matter by normal filtration. In contrast to a solution, where the entire combination is homogeneous, the particles in a colloid are a heterogeneous mixture The suspended particles in terms of size, are again between those of somewhere between a solution and a suspension. A colloid's dispersed particles are typical 1-1000 nm, whereas solute particles are 0.01-1 nm and the suspension's particles are greater than 1000 nm.
Colloids are heterogeneous in nature. Colloidal solutions are known to exhibit the Tyndall Effect, a phenomenon where light rays falling on colloids are scattered as a consequence of the interactions between the light and the colloidal particles.
Usually, the components are spread equally throughout a colloidal mixture. In these combinations, the component that is scattered is known as the dispersed phase, and the component through which it is dispersed is known as the continuous phase/dispersing medium.
Types of colloids
Colloids are classified into many types based on the interested property.
Considering the physical state of the dispersed phase and dispersion medium, nine types of combinations can be made.
Colloids are categorised also on the basis of the formation of the colloid particles into three types as-
1. Multimolecular colloids
2. Macromolecular colloids
3. Associated colloids
Multimolecular colloids:
Multimolecular colloids, also known as micromolecular colloids, are generated when a large number of atoms or small molecules with a diameter of less than 1 nm of a substance interact in a dispersion medium to form aggregates of the size in the colloidal range. Atoms or molecules that make up the dispersible particles of multimolecular colloids that are bound together by van der Waals forces. Multimolecular colloids are generally lyophobic in nature.
Example: A sol or colloidal suspension of gold nanoparticles in a dispersed media, typically water, is known as colloidal gold. Depending on how the particles have aggregated and the size of the particles, the colloid is typically either a deep red colour or a blue/purple colour. Bredig's arc technique is used to create gold sol.
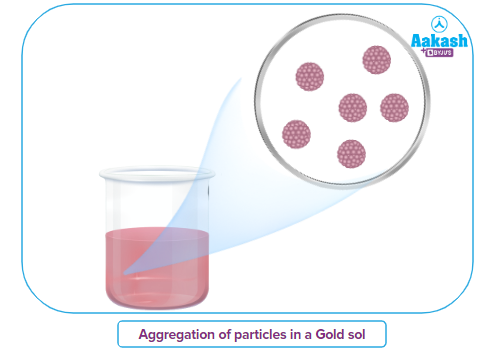
Macromolecular colloids:
The dispersed phase particles in these kinds of colloids are already big enough to be of colloidal dimension. These molecules are classified as macromolecules because of their extremely high molecular mass. Macromolecular colloids are the colloidal solutions that are created when such molecules are dispersed in an appropriate dispersion medium. Mostly macromolecular colloids make up lyophilic sols.
Example: Examples are natural macromolecules like nucleic acids, starch, proteins, gelatin, cellulose, and gelatin. On the other side, manmade macromolecular colloids include synthetic polymers like synthetic rubber, polyethene etc.

Associated colloids
Sodium palmitate was observed to exhibit high electrical conductivity above a certain concentration. This prompted a theory of association of the palmitate ions to a bigger ion called a micelle.
Associated colloids are ionic compounds, which act as strong electrolytes at low concentrations and become colloidal sols at large concentrations. The charged particles of the electrolytes, aggregate and exhibit colloidal behaviour when concentrated. These associated particles are known as micelles. They are sometimes referred to as associated colloids. Naturally, these colloids are micelles and are charged species. Micelle formation occurs above a characteristic temperature and concentration. Diluting these colloids allows them to be reverted back to their original electrolyte solution. Associated colloids have good electrical conductivity
The temperature above which micelle formation can happen is known as the Kraft temperature (Tk) and the concentration above which micelle formation occurs is known as the Critical Micelle Concentration (CMC).
Examples include surface-active substances like soaps.
Soap (sodium stearate) is a salt of a log chain fatty acid and sodium hydroxide base. In water, it ionises as
At low concentrations the hydrophobic long alkyl chain of the anion get attached to dust particles and brought into water by the water-soluble carboxylate ion.

Micelle:
These anions associate in concentrated solutions, resulting in the formation of a colloidal-sized aggregate.
Micelles are small, spherical formations made of a few hundred molecules. They unite to form a larger aggregate that is in the colloidal range.
Example: An extended chain alkyl group is represented by R in the formula for soap, which is written asRCOONa. Soap ionises to produce the RCOO- ion when it is dissolved in water. The RCOO- ion contains two parts-long hydrocarbon chain R (tail), and a polar carboxylic acid anion group -COO-.
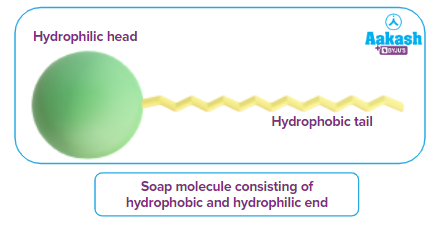
While the COO- group, being polar and hydrophilic, is water-loving, the hydrocarbon tail R is non-polar and hydrophobic. Hydrophobic tails try to avoid contact with water. As a result, the soap molecules come together to form spherical bigger molecules such that, all the tails are pointing to the centre of the sphere. The carboxylic groups are positioned at the periphery of the sphere. This sphere formed from several molecules is called a micelle.
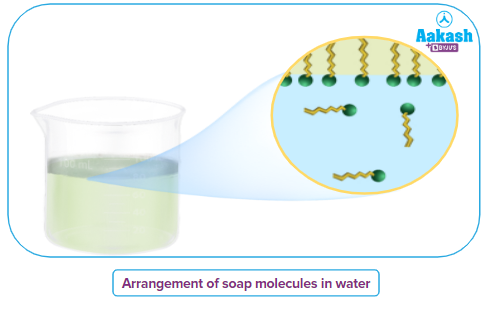
A soap micelle is thus a negatively charged colloid particle with negatively charged COO- groups organised spherically at the surface. The hydrocarbon chains, on the other hand, tend towards the centre.
The COO- groups on the micelle's surface are surrounded by Na+ ions, which tend to draw the micelle into the bulk of the solution. A micelle can have up to 100 molecules or more. The critical micelle concentration, or CMC, is the lowest concentration of soap at which micelles form. Micelles form only above a certain temperature known as the Kraft temperature (Tk). Kraft temperature increases with increasing length of the hydrocarbon chain increase.
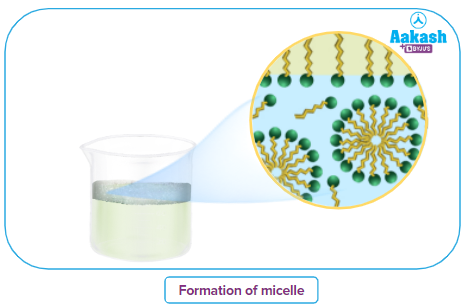
Stearate ions combine with dirt particles to form micelles when the concentration is high, the CMC is exceeded, and the kraft temperature is exceeded. It has been shown that as the system temperature increases, the CMC of each ionic surfactant decreases at first, and then increases.
Characteristics of Associated Colloids(micelles)
Associated colloids and micelle formation occur, above a minimum concentration of electrolyte known as critical micelle concentration. (CMC).
CMC -
- decreases with the length of the carbon chain
- Increases with the lyophobic nature of the hydrocarbon chain
- Increases with increasing temperature
- Do not form below the Kraft temperature.
- Smaller for simpler ionic compounds
After formation,
- surface tension of the solution remains constant
- Molar conductivity decrease and
- Turbidity of the solution increases.
Practice Problems:
Q1. The micelle formation occurs only above:
(A) Kraft Temperature (Tk)
(B) Critical micelle concentration (CMC)
(C) Both A and B
(D) None of the above
Answer: (C)
Solution: Micelles are aggregations of surfactant molecules spread in a liquid colloid. They are created through the self-assembly of amphiphilic molecules. The structures have a hydrophilic/polar (head) portion and a hydrophobic/nonpolar section (tail).
Micelle formation occurs only above a certain temperature known as the Kraft temperature (Tk) and a certain concentration known as the critical micelle concentration (CMC).
Q2. Among the following, which is not an example of an associated colloid?
(A) Sodium stearate
(B) Gum
(C) Detergent
(D) Potassium stearate
Answer: (B)
Solution: Detergents, such as sodium and potassium stearate, and soaps are examples of associated colloids, whereas gum is an example of a lyophilic colloidal substance.
Q.3. Among the following, which is the characteristic of a multimolecular colloid?
(A) A large number of molecules come together to form a colloidal-sized particle.
(B) An example of a multi-molecular colloid is the starch solution.
(C) The majority of multi-molecular colloids are lyophilic in nature.
(D) A particle of colloidal size is created when a large number of atoms aggregate together.
Answer: (D)
Solution: Multimolecular colloids, also known as micromolecular colloids, are generated when a large number of atoms or small molecules with a diameter of less than 1 nm of a substance interact in a dispersion medium to form aggregates with the size in the colloidal range. Multimolecular colloids are generally lyophobic in nature and Starch solution is a lyophilic colloidal example, not a multi-molecular colloidal example.
Q4. Among the following, which is an example of an associated colloid?
(A) Protein + water
(B) Soap + Water
(C) Both A and B
(D) none of the above
Answer: (B)
Solution: Protein + Water is a colloidal solution. A synthetic composition known as a colloid is one in which one substance is evenly distributed throughout another. In contrast to an answer, where the substance's particles are completely dissolved inside, the scattered substance's particles are only suspended in the mixture.
An example of an associated colloid is the colloidal mixture of soap and water. Associated colloids are microscopic heterogeneous structures in which the micelles, which make up the colloidally scattered stage, are formed by the agglomeration of atoms or other minute fragments of the substance that have been broken down in the scattering medium.
Frequently Asked Questions(FAQs):
Q1. What is the effect of CMC on washing clothes?
Answer: CMC tells us the amount of detergents to be used for laundering. The soap or surfactants reduces the surface tension of water to improve the wettability of cloth fabric. The surface tension though it decreases with increasing concentration, it becomes almost constant after the CMC is reached. So the minimum amount of surfactants to be used is to have minimum CMC. But about 3 to 4 times the minimum micelle concentration is necessary for complete cleaning of the cloth. Lesser than that will result in incomplete cleaning while an excess amount will be a waste.
Q2. Will micelles also occur in other solvents, such as ethanol?
Answer: No, When soap is added to water, micelles form. Micelles form around molecules of gasoline, grease, and filth. Soaps in the form of micelles remove filth that accumulates in the centre of the micelle.
Micelle formation does not occur in ethanol. This is due to the fact that the hydrocarbon chain of soap is soluble in alcohol. As a result, no micelle forms. Micelle production occurs when soap is introduced to water but not when ethanol is added.
Q3. Why is the role of micelles in human digestion important?
Answer: Since vitamins are not digested by humans and the majority of vitamins are fat-soluble, micelles serve the same purpose in the process of digestion in the body as they do in our daily lives by removing dirt from the medium. Micelles, therefore, aid in removing them from the body throughout this process.
The human body's digestive system needs micellar solution because it is crucial in the elimination of complex fats and fat-soluble vitamins that are indigestible to humans.
Q4. Define Lyophilic and Lyophobic colloids.
Answer: Lyophilic colloid: The term lyophilic refers to a liquid-loving. Lyophilic colloids are a type of colloidal solution where the dispersed phase particles have a strong affinity for the dispersion medium. These also are known by the name intrinsic colloids. These have a stable, reversible character. It is therefore difficult to precipitate.
Examples of lyophilic colloids are starch, gum and albumin.
Lyophobic colloids: Lyophobic refers to liquid-hating. Lyophobic colloids are colloidal solutions in which the dispersed phase particles have no attraction for the dispersion medium. They are inherently irreversible. These also are known by the name extrinsic colloids. By adding a small amount of electrolyte and stirring it, it readily precipitates or coagulates. As a result, they are unstable.
Examples of lyophobic colloids are platinum sol and gold sol.


