-
Call Now
1800-102-2727
Applications of Adsorption- Adsorption, Factors Affecting Adsorption of a Gas on Solid, Applications of Adsorption, Practice Problems and FAQs
Have you ever noticed opaque glass window panes of your house or car in the early morning or during rainy days? It will have a layer of water droplets hanging on the glass, especially during cold weather conditions.
But, if it is water, why does it stick to the glass instead of flowing down? Do you know which phenomenon is responsible for it?
The phenomenon responsible for the misty window is known as adsorption. Adsorption was first used by a German physicist named Heinrich Kayser in 1881. Adsorption is frequently described as a surface phenomenon in which particles adhere to the top layer of material. It typically involves molecules, atoms, or even ions of dissolved gas, liquid, or solid attached to the surface. So, next time when you write your name in the fog that has accumulated on your windowpane, thank the wonderful phenomenon of adsorption.

Let’s have a tour in this article to learn more about the applications of adsorption.
Table of Content
- Introduction to Adsorption
- Factors Affecting Adsorption of a Gas on Solid
- Applications of Adsorption
- Practice Problems
- Frequently Asked Questions
Introduction to Adsorption
Adsorption is the process of a surface having higher concentration than its bulk of a foreign material. Adsorption is a surface process compared to absorption which is bulk process.
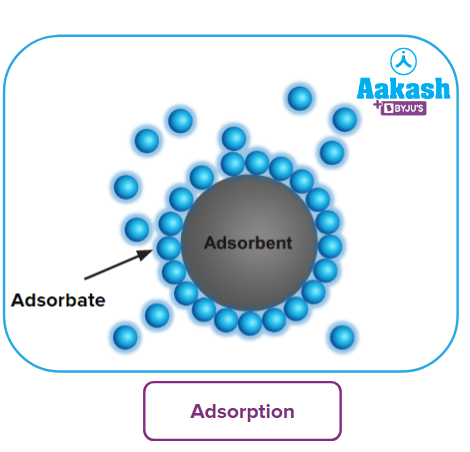
It is the accumulation of other substances at the surface of another solid or liquid substance rather than in its bulk. The molecular species or substance that concentrate or accumulate at the surface is known as adsorbate. The substance that provides the surface for adsorbate is the Adsorbent. For instance, charcoal, silica gel, and alumina are the commonly employed adsorbents
Adsorption is a surface phenomenon, therefore, the amount of adsorbate attached to the surface of the adsorbent increases with an increase in the surface area of the adsorbent.
Factors Affecting Adsorption of a Gas on Solid
There are different factors which affect the adsorption of a gas on a solid surface which include:
- Nature of Adsorbate: Adsorption quantity is directly proportional to the ease of liquefaction of the gas which in turn depends on the critical temperature of the gas. The greater the value of attractive force between the molecules (van der Waals constant-a), the easier it is for gas to liquefy, and the higher the critical temperature.
- Nature of Adsorbent: Adsorption is directly proportional to the surface area of the solid. Due to this finely divided adsorbents are more efficient than standard adsorbents.
For example, The adsorption of gasses such as O2, N2, H2 on finely divided transition
metals Ni and Co is common.
- Temperature Influence
Adsorbent (s) + Adsorbate (g) ⇌ [Adsorbent---Adsorbate] + ΔH
Adsorption is an exothermic reaction, whereas desorption is endothermic. Increasing the temperature, according to Le Chatelier's principle, promotes desorption.
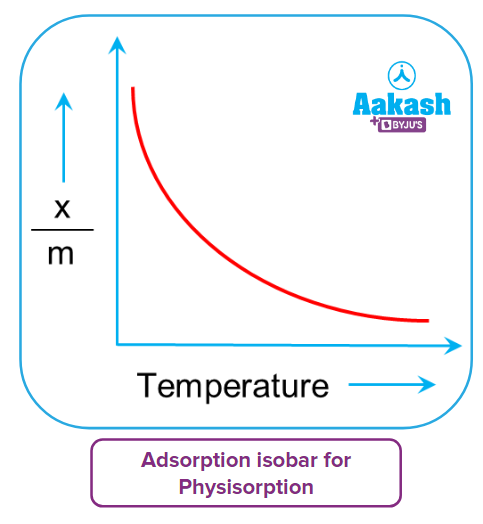
Adsorption isobar curves express the temperature-dependent variation in the amount of gas adsorbed by the adsorbent. It is a plot of (xm) versus temperature at constant pressure. In this equation, x denotes the mass of gas adsorbed, m the mass of the adsorbent, and (xm) the amount of gas adsorbed per unit mass of adsorbents.
The physisorption and chemisorption isobars are plotted separately.
As the temperature rises, physical adsorption decreases.
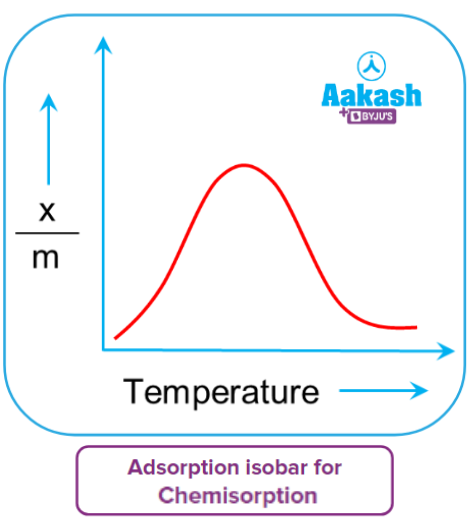
Chemical adsorption involves chemical bonding between the adsorbent and adsorbate. An increase in temperature initially increases the number of such bond formations and hence chemisorption. The heat supplied acts as activation energy, causing the initial increase in chemisorption, and as more and more adsorbate molecules gain energy and possess energy greater than activation energy, the initial increase in chemisorption is reversed. As a result, adsorption increases as temperature rises. Chemical adsorption rises initially to form a peak and then falls gradually.
- Effect of Pressure
The pressure affects the extent of a gas's adsorption(xm). An adsorption isotherm is a curve that expresses the variation in the amount of gas adsorbed by the adsorbent with pressure at a constant temperature. An adsorption isotherm is a curve that shows the variation of quantity of the adsorbent per unit mass to the pressure of the adsorbent gas measured at constant temperature conditions..
The extent of adsorption((xm) is assisted by pressure. It reaches a maximum value equal to the pressure Ps also known as saturation pressure.
Example:
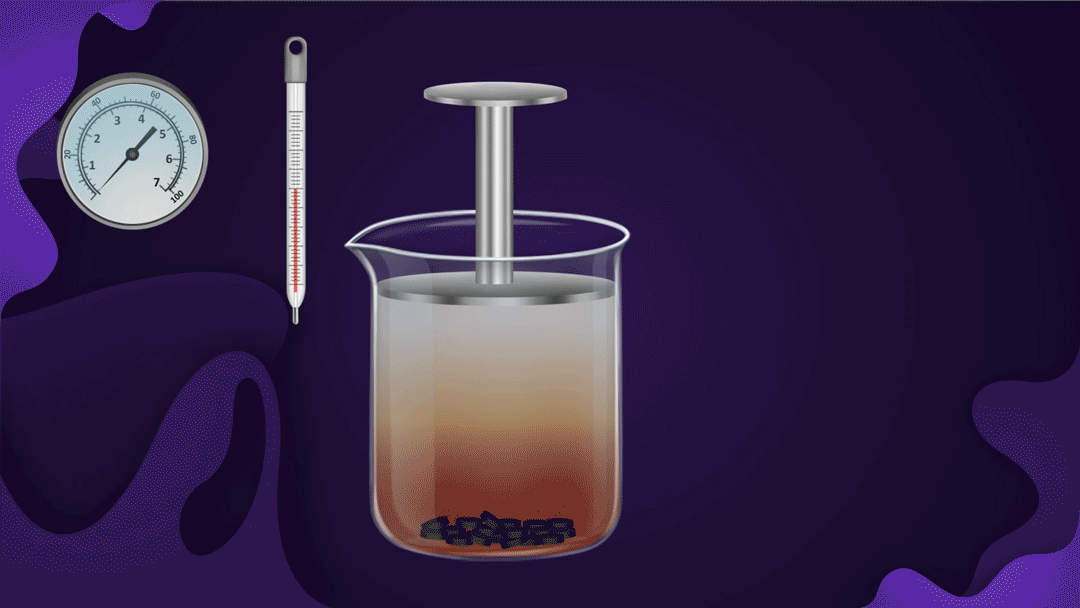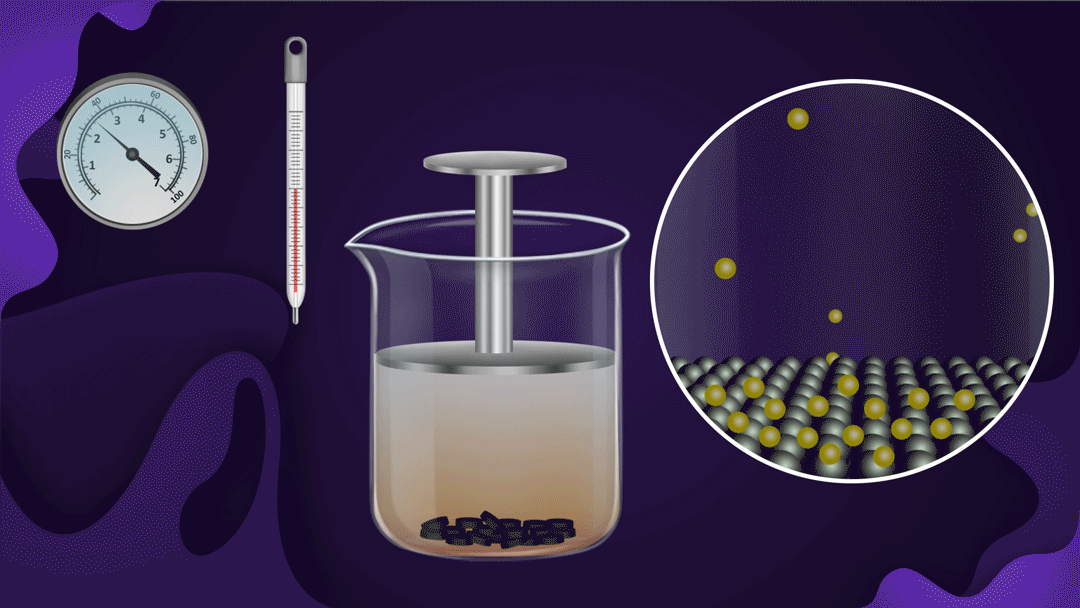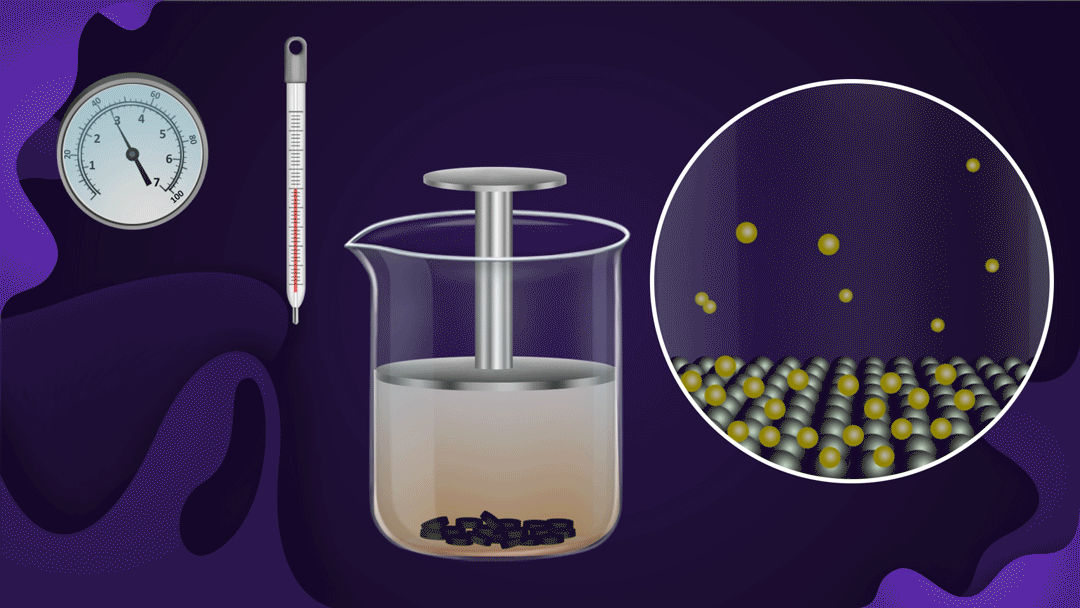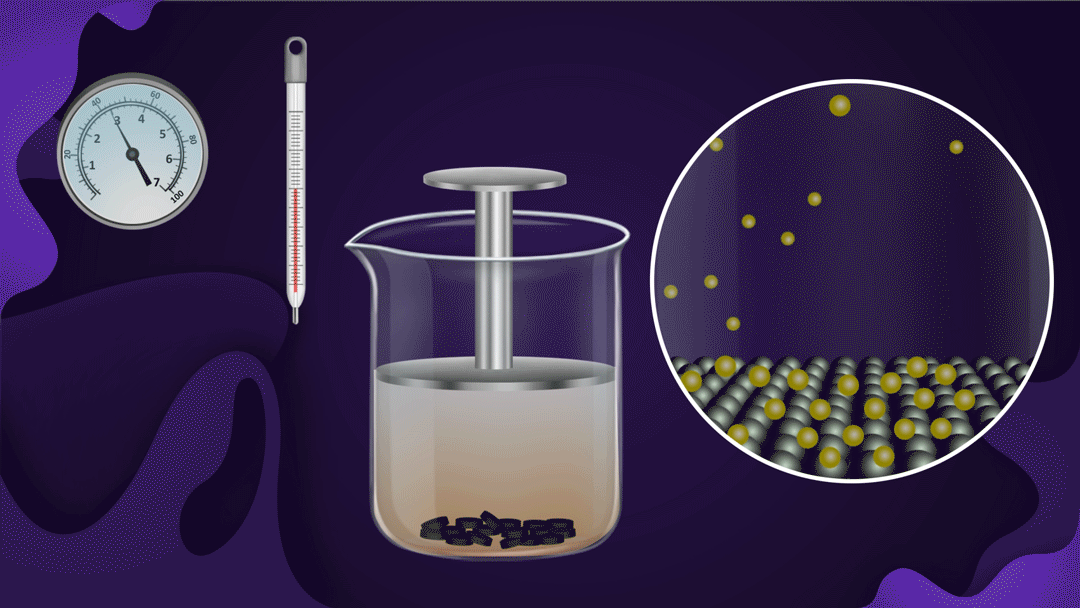
Consider the beaker with a piston.
The beaker's bottom is lined with charcoal, and it's filled with NO2 gas. When a constant temperature piston is pressed, the gas pressure rises, increasing the adsorption of NO2 a gas on the charcoal. As the piston moves inwards, the colour intensity of the NO2 gas decreases, indicating increased adsorption.
Two isotherms provide empirical quantifications for the effect of pressure on adsorption.
Freundlich Adsorption Isotherm
Note: The above isotherms are applied only to physical adsorption.
Applications of Adsorption:
i) Air Pollution Masks: Thanks to covid, people have learned to freely use face masks. But, masks have been in use for a long time to protect people from air pollutants, whether it is viruses, microorganisms, dust, pollen grains, etc.
Miners used masks with active charcoal to adsorb toxic gases and fine dust particles prevalent in mines to protect themselves from lung diseases. These are made of silica gel or activated charcoal powder, and when dust or smoke passes through them, the particles become adsorbent on the surface.
Most people are allergic to pollen grains and other fine particles of plant sources. The development of the disease of Asthma and other respiratory diseases is attributed to pollen grains. Masks with special adsorbent materials with sensitivity to toxic gasses, and pollen grains, have been developed. The use of nano-sized silver and other particles is being developed.
ii) Noble Gas Separation Using Dewar's Flask: The air contains about 1 % of noble gases. The noble gases are subjected to electric arks to remove nitrogen as nitrogen oxides. The nitrogen oxides, oxygen and other gases are removed from the air by cooling in a Dewar’s flask containing activated charcoal. The different noble gases like Helium, Krypton Argon etc., are separated using their difference in adsorption rates on activated charcoal at different low temperatures.
iii) Water Purification:
Municipal water treatment: Sediments, suspensions and colloids can be removed from water easily using alum. A small amount of alum is added to the water and leftover time, undisturbed. The contaminants get adsorbed on alum and are removed by simple filtration methods
Mineral free water preparation: The hardness of water is caused by the presence of calcium and magnesium ions in water. Both of these ions as well as any other cations present are removed in an ion exchange resin-containing column so as to give mineral-free water.
iv) Moisture and Humidity Removal: Moisture-free atmosphere is needed for some of the measurements. Moisture in the air is removed by adsorbing water molecules with silica gel. Might have seen a small packet(containing silica gel) in single pan balances to provide a moisture-free atmosphere.
v) Adsorption Chromatography: Chromatography is a technology that is used in the identification of compounds and separation of components of mixtures in trace or large amounts. The technique is based on the adsorption phenomenon.
Adsorption chromatography works on the difference in the adsorption between two different adsorbates of the adsorbent. For example, if an adsorbent is placed between a solid adsorbate and a moving adsorbate like a liquid or a gas, the difference in adsorption binding of components will make some components to adsorb strongly and cling to the sold adsorbate leaving the less bonded adsorbent to get carried away by the moving adsorbate. Thus the components of a mixture can be separated.
The solid adsorbate is called a stationary phase while the moving adsorbate is called as mobile phase.
It is used for separating pigments, hormones, amino acid and antibodies..
vi) Metallurgy: Froth floatation is a simple but useful method of removing gangue particles present in the sulphide ores of metals like iron and copper. This metallurgical separation process relays on the selective adsorption of sulphide ore particles on oils added to water. The froth formed by the oil with water removes the sulphide ore particles selectively leaving behind the gangue impurity particles. This concentrated ore is processed by roasting and other processes to purify the metal.
vii) Creation of Vaccum
Adsorbates like activated carbon in connection with an already evacuated vessel is used to produce ultra vaccum
Practice Problems
Q1. Which of the following option is correct with respect to adsorption?
a. Adsorption is a surface phenomenon
b. Adsorption first increases with an increase in temperature and then decreases in case physical adsorption process
c. Freundlich Adsorption Isotherm and Langmuir adsorption isotherm explain the relation of adsorption with temperature
d. Amount of adsorbate attached to the surface of the adsorbent decreases with an increase in the surface area of the adsorbent.
Answer: (A)
Solution: Adsorption is a surface phenomenon in which particles adhere to the surface of a material. Adsorption is the process by which atoms, molecules, or ions of dissolved gas, liquid, or solid attach to the surface of a material.
In the case of Chemical adsorption extent of adsorption rises initially with temperature to form a peak, and then falls gradually.
Freundlich Adsorption Isotherm and Langmuir adsorption isotherm explains the relationship of adsorption with pressure.
The amount of adsorbate attached to the surface of the adsorbent increases with an increase in the surface area of the adsorbent.
Therefore, option(A) is correct.
Q2. Select the factor that affects the adsorption of gasses on a solid surface.
a. Surface area of the adsorbent
b. Temperature
c. Nature of adsorbent
d. All of these
Answer: (D)
Solution: Adsorption is a surface phenomenon in which particles adhere to the surface of a material. Adsorption is the process by which atoms, molecules, or ions of dissolved gas, liquid, or solid attach to the surface of a material. There are some factors which affect the adsorption of gas in a solid which include - the surface area of the adsorbent, Temperature, pressure, nature of adsorbent and nature of adsorbent. Therefore option (D) is correct
Q3. Which of the following phenomena is observed in the ion exchange raisin method of removing the hardness of water?
a. Absorption
b. Adsorption
c. Solubility
d. Precipitation
Answer: (B)
Solution: Calcium and magnesium ions are adsorbed on the surface of ion exchange resin in this method of removing water hardness. Therefore option (B) is correct.
Q4.Which among the given option represent the application of adsorption?
a. It is used in the removal of the hardness of water
b. Adsorption is used in the adsorption of froth in the froth flotation process
c. Adsorption is used in the removal of moisture with the help of silica gel
d. All of these
Answer: (D)
Solution: Adsorption is a surface phenomenon in which particles adhere to the surface of a material. Adsorption is the process by which atoms, molecules, or ions of dissolved gas, liquid, or solid attach to the surface of a material. Some important applications of adsorption include:
- It is used in the removal of hardness of water
- Adsorption is used in the adsorption of froth in the froth flotation process
- Adsorption is used in the removal of moisture with the help of silica gel
Therefore option (D) is correct.
Frequently Asked Question-FAQs
Q1. What is the difference between adsorption and absorption?
Answer: Adsorption is the process by which matter particles deposit on the surface of a material. It is a surface phenomenon. Absorption is the process by which molecules are absorbed into the mass of matter (solid, liquid or gas)
Q2. What are physisorption and chemisorption?
Answer: The accumulation of gas on the surface of a solid occurs due to weak van der Waals forces in physisorption. The accumulation of gas on the surface of a solid occurs in physisorption due to the presence of weak van der Waals forces. It usually happens at low temperatures and is multilayer adsorption.
Chemical bonds hold the adsorbate's gas molecules or atoms to the solid surface of the adsorbent in chemical adsorption or chemisorption. Chemical bonds can be either covalent or ionic. It usually happens at high temperatures and is called monolayer adsorption. Because chemisorption requires high activation energy, it is also known as activated adsorption.
Q3. What is the effect of adsorption on the degree of randomness?
Answer: Adsorption reduces the degree of randomness of the particles because the adsorbate particles are bound to the surface, limiting their freedom. As a result, entropy decreases, i.e. ΔS < 0

Q4. What is the difference between adsorbate and adsorbent?
Answer: The molecular species or substances that concentrate or accumulate at the adsorbent's surface are known as adsorbate material. Adsorbate molecules are adsorbed on the surface of the material. Adsorbate adsorbs on the surface of a substance and is known as adsorbent.


