-
Call Now
1800-102-2727
Allotropes of Carbon – Allotropy, Crystalline and Amorphous Allotropes of Carbon, Practice Problems and FAQ
Who hasn't heard of Shahrukh Khan, a.k.a. King Khan? The versatile and gifted Indian actor has played a variety of characters in multiple Bollywood films with success. In his films and television productions, Shahrukh Khan has taken on a variety of characters, including Samrat Ashoka in the Asoka film, a hockey coach in Chak-de India, and a soldier in the Doordarshan serial Fauji. By changing his clothes and make-up, he was able to assume the appearances of a coach, Samrat Ashoka, and a soldier.

Isn't it intriguing how well-suited a single man may be to so many characters from such various backgrounds?
The actor that performs in several programmes, sporting varying appearances, garb, and gestures, is the same actor in reality. A single person is actually assuming the roles of many different people. This component and the allotropy phenomenon share a lot of parallels.
This property, which allows an element to have numerous physical forms in a single physical state, is known to be present in a number of the elements in our periodic table. The Greek term "allotropia," which means "changeable," is where the word "allotropy" originates.
Baron Jöns Jakob Berzelius, a Swedish scientist, first proposed the concept of allotropy in 1841. Allotropes are different structural configurations of a chemical element that essentially share the same physical state despite having various atom configurations.
Let's focus on allotropes of carbon in this article.
TABLE OF CONTENTS
- Allotropy
- Crystalline Allotropes of Carbon
- Amorphous Allotropes of Carbon
- Practice Problems
- Frequently Asked Questions –FAQ
Allotropy
Allotropy is the property of some chemical elements that allow them to exist in two or more different forms, called allotropes of the element, while yet existing in the same physical state. Allotropes are unique structural variations of an element where the atoms' bonds are changed.
For example, oxygen has majorly two allotropes namely, oxygen and ozone.
The phenomenon of allotropy is only present in elements, not in compounds. Polymorphism is a phenomenon for compounds, yet, it only applies to solid crystals. Allotropy only refers to different manifestations of an element within the same physical phase (the state of matter, such as a solid, liquid or gas). The distinctions between these states of matter are not sufficient to demonstrate allotropy. Allotropes of chemical elements are also known as the element's polymorphs or phases.
For some elements, allotropes have different chemical formulae or crystal structures in addition to having different physical phases. For example, ozone (O3) and dioxygen (O2), two oxygen allotropes, can both exist as solid, liquid, and gaseous phases. When heated to a liquid state, some elements, like phosphorus, change from their various solid allotropes into identical P4 forms. Other substances, however, do not maintain distinct allotropes throughout a range of physical phases.
Crystalline Allotropes of Carbon
The two most common chemical elements with allotropes are carbon and sulphur. Because of its valency, carbon may form a wide variety of allotropes. Carbon is one of the few elements that can have a variety of allotropic forms due to its capability to have various oxidation states or coordination numbers. Another vital factor is the catenation capacity of carbon. As a result, several carbon allotropes are created. Diamond is the most popular crystalline form of carbon, followed by graphite and fullerenes.
Graphite
The van der Waals forces hold the layers of graphite together, and there is 340 pm distance between each layer. Planar hexagonal rings of C atoms that have undergone sp2 hybridisation make up each layer. With three of its neighbouring carbon atoms, each carbon atom in the hexagonal ring forms three sigma bonds, and the fourth electron forms a pi bond. The ring's carbon-carbon bond has a 141.5 pm length. Because of its ability to slide across other layers, graphite is soft and has lubricating qualities.

Graphite is of two forms: 𝛼 and 𝛽. In the 𝛼 form, the layers are set up in the form of ABAB… pattern, with the third layer being placed just above the first layer. The layers are ordered in ABCABC…. pattern in the 𝛽 form.
Properties
- It has a metallic lustre and is a dark grey material.
- It seems greasy and incredibly soft to the touch.
- The fourth valence electron of each carbon is free to move since only three of each carbon's electrons are required to form hexagonal rings in graphite. Due to this, graphite functions well as a heat and electrical conductor.
- Diluted acids, alkalis, and chlorine do not harm it. It slowly turns into carbon dioxide when oxidised with sulphuric acid and potassium dichromate.
- Graphite is more thermodynamically stable than diamond.
Uses
- It is used to create carbon arcs and electrodes.
- It serves as a lubricant for machinery operating in hot environments.
- It is a component in the production of lead pencils. Clay is combined with powdered graphite before being formed into sticks. Pencils are made from these sticks.
- In atomic reactors, it serves as a moderator.
- In the production of steel, it serves as a reducing agent.
- High-strength composite materials make use of it.
- It is employed in the production of the highly tolerant crucible.
Diamond
Each carbon atom in the crystalline lattice of diamond is sp3 hybridised. The length of the C-C bond, which connects each carbon atom to the other four carbons, is 154 pm. A rigid 3D network of carbon atoms is created by the structure as it stretches into space. Diamond is among the earth's hardest materials because protracted covalent bonding is incredibly hard to break.

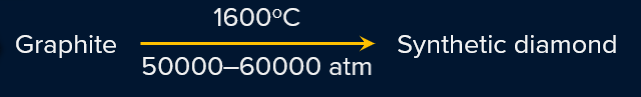
Synthetic diamonds can be prepared from diamonds in the following way.
Properties
- The most dense and pure kind of carbon is diamond. It has a density of 3.51 gcm-3.
- It has the highest melting point (3843 K) of any natural substance known to man and is the toughest.
- In all solvents, it is insoluble.
- It possesses a high refractive index and is translucent.
- It does not conduct electricity well. This is due to the fact that no unpaired electrons remain in the crystal as all of each carbon's valence electrons are utilised in the C-C sigma (σ) bonds.
- Diamond is virtually insensitive to all chemicals, including strong acids, alkalis, and salts chemically.
- Diamond has an ignition temperature that varies from 690 °C (1,274 °F) to 840 °C (1,540 °F) in a pure oxygen environment; smaller crystals often burn more quickly.
- It burns with a light blue flame and warms up from red to white heat before continuing to burn even after the heat source has been turned off.
- In contrast, owing to the dilution of oxygen by nitrogen in the air, combustion will stop as soon as the heat source is withdrawn.
- The size and colour of a diamond determine its worth. Diamonds with less colour are more valuable than those that are bluish-white. The least expensive and least used type of diamond is the black diamond.
Uses
- Instruments like a glass cutter, a marble-cutting saw, and rock drilling equipment is made of diamonds.
- Diamonds' remarkable brilliance makes them ideal for usage in jewellery.
- Sharp-edged diamonds are a tool that eye surgeons generally use.
- Drawing incredibly thin wires of metals like tungsten requires the use of diamond dies.
Fullerenes
The only pure form of carbon is found in fullerenes, which are crystalline carbon allotropes with a smooth structure free of "dangling" bonds. The fullerene is a big, spherical molecule with the chemical formula C2n, where n ≥ 30. A sooty substance is created when Cn small molecule condensation causes graphite to be heated in an electric arc in the inert atmosphere, such as He or Ar, practically producing fullerene.
The sooty material that results primarily consists of C60, with traces of other fullerenes and a minor amount of C70. By extracting the fullerenes using benzene or toluene, proceeded by chromatography using alumina, the C60 and C70 fullerenes can be completely extracted from the fullerenes soot.
Fullerenes are cage-like molecules; the Buckminsterfullerene (C60) molecule has the shape of a soccer ball. A carbon atom is located at each of the 60 vertices of the saucer-shaped fullerene molecule. There are 12 five-membered rings and 20 six-membered rings in it.
Five-membered rings are fused solely to six-membered rings, while six-membered rings are fused to both other six-membered rings and five-membered rings. The carbon-carbon bond lengths between its single and double bonds, which are both present, are 142 pm and 138.3 pm, respectively.

Properties
- The structure and behaviour of fullerene change when the temperature changes. At a higher temperature, the fullerene changes into the C70 form.
- The structure of fullerene changes as pressure varies.
- Ionisation enthalpy for fullerene is 7.61 electron volts.
- The electron affinity of fullerene ranges from 2.6 to 2.8 electron volts.
- Fullerene (C60) mimics an electrophile in chemical reactions.
- The fullerene molecule can act as an electron acceptor. It is easily capable of taking three or more electrons. This means that it might have the ability to function as an oxidising agent.
- Fullerenes are doped with alkali or alkaline earth metals to produce superconductivity.
- Ferromagnetism is a characteristic of fullerene.
- They are highly soluble in organic solvents
Uses
- Buckminsterfullerene is used as catalysts, lubricants, and drug delivery systems.
- Some varieties of fullerene are also employed as gas absorbents.
- Fullerene-based conductors are used.
- It is capable of absorbing gases.
- Fullerene-based lubricants are used.
- Various kinds of fullerenes are used in the production of goods connected to cosmetics.
- Carbon nanotubes are made of graphene sheets.
- In various methods, fullerenes are used in biological applications.
Carbon Nanotubes
Cylindrical fullerenes are what make up carbon nanotubes. These carbon tubes are typically only a few nanometers broad, but their lengths can range from a few micrometres to several millimetres. They might be open-ended or frequently have closed endings. Additionally, there are instances where the tube narrows before cutting off. Their unusual molecular structure confers remarkable macroscopic properties, such as high tensile strength, high electrical conductivity, high ductility, high heat conductivity, and relative chemical inactivity (as it is cylindrical and "planar," meaning it lacks easily displaceable "exposed" atoms). Since a nanotube's diameter is only a few nanometers, or about 50,000 times smaller than the width of a human hair, and its length can reach several centimetres, the name "nanotube" refers to its size.
Jackets that are bulletproof are made with carbon nanotubes. The bodies of spacecraft and aircraft can be constructed from carbon nanotubes. Due to their semiconducting characteristics, carbon nanotubes can be used to create high-performance nanoscaled thin-film transistors that can replace silicon-based transistors.
Amorphous Allotropes of Carbon
The term "amorphous carbon" refers to carbon that lacks any crystalline structure. There is some short-range order, as with other glassy materials, but no long-range pattern of atomic positions. Although completely amorphous carbon can be created, the majority of amorphous carbon actually contains small crystals of carbon that resemble graphite or even diamond.
Amorphous carbon is the colloquial term for coal, soot, and carbon black. However, they are byproducts of pyrolysis, which under typical circumstances do not result in real amorphous carbon. Pyrolysis is the process of breaking down a substance through the action of heat.
Some important amorphous allotropic forms of carbon are coal, coke, wood charcoal, animal charcoal, sugar charcoal, lampblack and gas carbon.
Recommended Videos
Structure and Properties of Graphite | CHEMISTRY | JEE | Concept of the Day | Kartikey Sir
Practice Problems
1. Which of the following best describes fullerene’s chemical formula?
a. C20
b. C06
c. C1
d. C60
Answer: D
Solution: The only pure form of carbon comes in the crystalline form of fullerene. It is a C60 molecule that is utilised to create new polymers, pharmaceuticals, light batteries, and small ball bearings.

So, option D is the correct answer.
2. The number of five-membered rings in the C60 fullerene structure is
a. 43
b. 20
c. 12
d. 6
Answer: C
Solution: 12 five-membered rings and 20 six-membered rings make up the C60 molecules known as fullerenes. Six-membered rings are linked to both the five-membered and six-membered rings, while the five-membered rings are linked to only the six-membered rings.

So, option C is the correct answer.
3. Which of the following is an amorphous carbon allotrope?
a. Fullerenes
b. Diamond
c. Graphite
d. Lampblack
Answer: D
Solution: Printing ink, black paint, varnish, and carbon paper are all made from lampblack, an amorphous form of carbon that is created by burning vegetable oils in a small amount of air. Lampblack is distinct from the crystalline allotropes of carbon, such as diamond, fullerenes, and graphite.
So, option D is the correct answer.
- Among the following which one is referred as black lead?
- Charcoal
- Diamond
- Graphite
- Fullerenes
Answer: C
Solution: Another crystalline allotropic carbon structure is graphite, which has hexagonal plates, a dark grey colour, and each carbon in graphite is sp2 hybridised. It conducts electricity because it has free electrons. It is known as black lead and works great as a lubricant.
So, option C is the correct answer.
Frequently Asked Questions – FAQ
1. Which form of carbon is used as a reducing agent?
Answer: Coal is destructively distilled to produce coke, which contains between 80 and 95 percent carbon. It is a firm, dark greyish-black solid. It is primarily employed in metallurgical processes as a fuel and as a reducing agent.
2. Which carbon allotrope has the highest hardness?
Answer: The hardest allotrope of carbon is diamond. A stiff 3-D network of carbon atoms with directional covalent bonds results from the structure's extension into space. Diamond is the earth's hardest material because protracted covalent bonding is incredibly difficult to break.
3. What is carbon's purest form?
Answer: Since fullerene lacks diamond-like edges or surface bonds that draw in other atoms, as do graphite and diamond, it is the purest form of carbon. The fullerene is a big, spherical molecule with the chemical formula C2n, where n ≥ 30. A sooty substance is created when Cn small molecule condensation causes graphite to be heated in an electric arc in the inert atmosphere, such as He or Ar, practically producing fullerene.
4. Which allotrope of carbon is created when vegetable matter carbonises?
Answer: Almost everywhere in the world, there are vast amounts of coal. It is thought that this coal was created by the slow carbonization of ancient plant matter that was buried in the earth's crust. It comes in a variety of forms and is an impure form of carbon. The carbon content of these is 60 % for peat, 70 % for lignite, 78% for bituminous coal, 83% for semi bituminous coal, and 70% for anthracite (90 Carbon). It is widely used as a fuel in furnaces, boilers, and engines. Additionally, it is employed in the production of synthetic gasoline and fuel gases.
5. What is glassy carbon?
Answer: A form of non-graphitizing carbon known as "glassy carbon" or "vitreous carbon" is frequently used as an electrode material in electrochemistry as well as for high-temperature crucibles and as a part of some prosthetic devices.


