-
Call Now
1800-102-2727
Acidity of Alcohols and Phenols: Acidity and Order of Acidity of Alcohols, Acidity of Phenol and Substituted Phenol, Practice Problems & FAQs
Alcohol provides us with comfort in many different ways.
Although most of us may identify with the film Hangover, alcohol offers medical benefits when handled properly.

Today, we're examining alcohol to see which beverage crosses the threshold into medicine.
Due to its high antioxidant content, dark beer functions as a beverage that prevents ageing.
Beer is flavoured with plants like ginger all over the world. It helps with rheumatoid arthritis and morning sickness.
Alcohol was frequently used as an antibacterial and anaesthetic for treating snake bites. Before operations, doctors used to provide whiskey shots to their patients.
We have stocked up on hand sanitizers and wipes since the Sars epidemic, but alcohol used to be the antiseptic back then.
Table Of Contents:
- Introduction of Acidity
- Acidity of Alcohols
- Acidity of Alcohol in Gas Phase
- Order of Acidity of Alcohol
- Acidity of Phenol
- Acidity of Substituted Phenol
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Acidity:
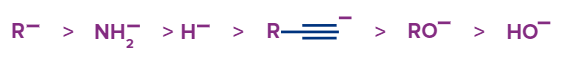
Alcohols are chemical substances that have an alkyl-substituted saturated carbon atom bonded to at least one hydroxyl functional group (-OH). Alcohol's hydroxyl group is polar because oxygen atoms have a higher electronegativity (3.44) than hydrogen atoms have (2.2). Alcohols give a proton (H+) and produce an alkoxide ion (R-O-) in the presence of a strong base due to this difference in electronegativity. They are hence slightly acidic in nature.
Before studying about the acidity of alcohol, we should take the Bronsted-Lowry concept of acid and base into account. According to this, bases are the exclusive protons acceptors and acids are the exclusive protons donors. From this alcohols are Bronsted acids. Alcohols are acidic because of the polarity of the (O-H) bond. The O-H bond is weakened as a result of the shared pair of electrons moving closer to the O O atom. This causes a proton to be released from the alcohol molecule, making them acidic.
Acidity of Alcohol:
Like water, alcohols are a weak base and acidic in nature. The hydrogen becomes partially positive as a result of the polarisation of the O-H bond. Alcohol is additionally both basic and nucleophilic owing to the electron pairs on the oxygen atom. Hydroxide, a weak leaving group, is changed into water, a strong one, during protonation of an alcohol. The following diagram illustrates the two acid-base equilibria for ethanol.

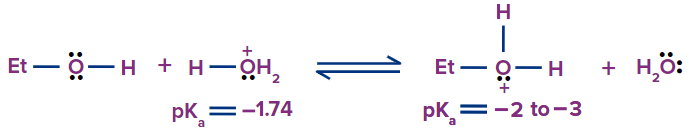
Only methanol (pKa = 15.5) is a somewhat stronger acid than water (pKa = 15.7). Tert-butanol (pKa = 18.0) and ethanol, among others, are weaker acids. On the other hand, terminal alkynes, hydrogen, ammonia, and alkanes are all far weaker acids than all alcohols.
Acidity of Alcohol in Gas Phase:
We observed that the destabilisation of alkoxide ions in the presence of the solvent causes the acidity of alcohol to decrease as the alkyl substituent increases. But in the gaseous phase, this is entirely the opposite. Alcohols that have more alkyl substitutions than those that have fewer substitutions are more acidic. This is because the anion's polarizability, or the conjugate base, predominates in the gas phase, which lacks a solvent. The electrons get easier to travel in the electric field and become more polarizable as we continue to add larger and larger alkyl groups. The anion becomes more stable in the gas phase and more acidic as a result of this polarizability.
In gas phase,
In Solvent phase,
Order of Acidity of Alcohol:
Alcohol's acidity is determined by the potency of the conjugate base it produces,i.e. alkoxide ions. It implies that the acid will be weaker the stronger the alcohol's conjugate base.
It means that the acid will become weaker as the conjugate base of the alcohol becomes stronger. A weaker conjugate base results from a more stable alkoxide ion, and as a consequence, the alcohol will be more acidic because the more stable alkoxide ion will more readily release H+ ion.
In the case of simple alkyl alcohols, primary alcohols (1o) are far more acidic than secondary alcohols (2o), which are more acidic than tertiary alcohols (3o).
In order to understand and analyse the stability of the alkoxide ion, we will focus on two aspects:
Steric factors:
When there are more substituted alkyl groups, the alkoxide ion is bulkier, making it more challenging for the solvent to stabilise the alkoxide. Alkoxide ion will be more reactive as a conjugate base with a higher basic strength as a result of its reduced stability. The acid is weak because of its conjugate base.
Electronic Factors:
As the quantity of electron-donating groups attached to the carbon with a hydroxyl group increases, the electron density on the O atom increases, making the alkoxide less stable, more reactive, and a powerful conjugate base. More substitutions result in reduced acidity in alcohols.
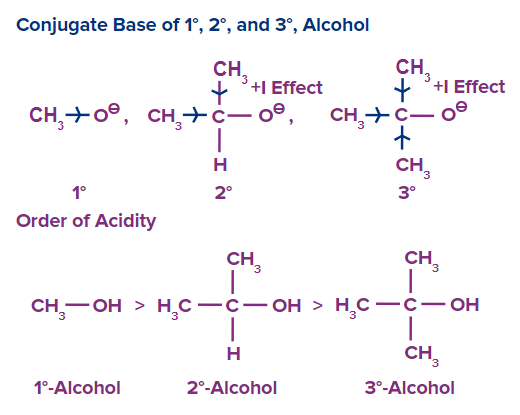
Primary alkoxide ions are the most stable when considering both electronic and steric variables, while tertiary alkoxide ions are the least stable. Because of this, primary alcohols have the highest acidity levels, whereas tertiary alcohols have the lowest acidity levels.
Thus, the descending sequence of alcohol acidity is as follows:
Primary alcohol (1o) >Secondary alcohol (2o) >Tertiary alcohol (3o)
Acidity of Phenol:
Because phenol is such a weak acid, the equilibrium is located far to the left. Because the generated phenoxide ion is somewhat stabilised, phenol can lose a hydrogen ion. Around the ring, the oxygen atom's negative charge is distributed.The much more stable the ion, the more likely its formation. The delocalized electrons on the benzene ring overlap one of the lone pairs on the oxygen atom.
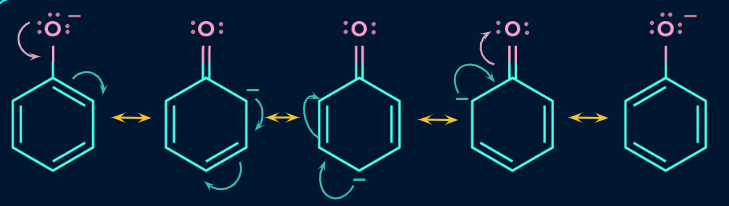
A delocalization that affects the ring and the oxygen atom simultaneously results from this overlap. As a result, the oxygen no longer carries the complete negative charge, which is now dispersed throughout the entire ion.
The ion is more stable than it would be if the entire charge was concentrated on the oxygen by distributing. The delocalized electrons will be directed to oxygen because it is the element in the ion with the greatest electronegative charge. This indicates that there will still be a significant amount of charge surrounding the oxygen, which will tend to draw the hydrogen ion back. Phenol is therefore merely a very weak acid.
Acidity of Substituted Phenol:
Phenol becomes more acidic when substituted with electron-withdrawing groups like the Nitro and Chloro group because these groups cause the formation of a stable phenoxide ion by the delocalization of the negative charge and inductive effects, or the ( effect. Because the action destabilised the phenoxide ion, a phenol substituted with an electron-donating group, such as a methyl or methoxy group, becomes less acidic. Following is a list of the para-substituted phenols of EWG and EDG in descending order of acidity:
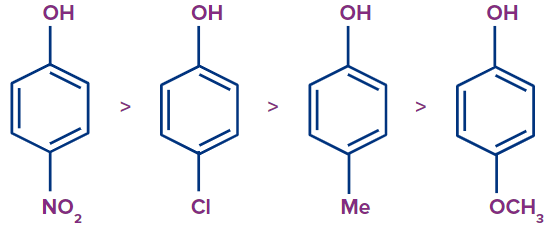
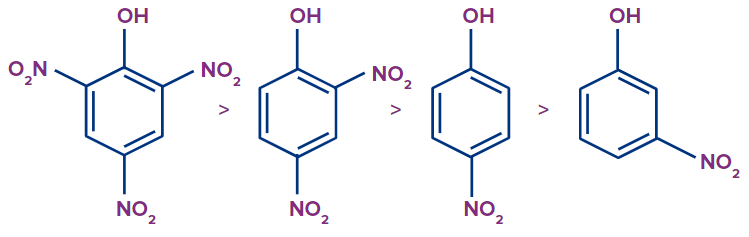
The majority of the negative charge is shown to be concentrated in the ortho and para locations of the phenoxide ion resonating structure. The conclusion that follows is that phenol becomes more acidic when electron-withdrawing groups are positioned in the ortho and para locations. The following list is arranged in decreasing order for ortho, para, and meta substituted nitro-phenol.
Practice Problems:
Q.1 Which alcohol is least acidic?
(A) CH3OH
(B) (CH3)2CHOH
(C) (CH3)3COH
(D) none of the above
Answer: (C)
Solution: Primary alkoxide ions are most stable when considering both electronic and steric variables, while tertiary alkoxide ions are the least stable. Because of this, primary alcohols have the highest acidity levels, whereas tertiary alcohols have the lowest acidity levels.
Thus, the descending sequence of alcohol acidity is as follows:
(1o) Primary alcohol >(2o) Secondary alcohol > (3o) Tertiary alcohol
Q.2 Among the following, which is least acidic?
(A) o-fluorophenol
(B) o-chlorophenol
(C) o-bromophenol
(D) o-iodophenol
Answer: (A)
Solution: o-chlorophenol > o-bromophenol > o-iodophenol > o-fluorophenol
This is so because fluorine in o-fluorophenol forms an H-bond with the -OH group's H atom due to fluorine's strong electronegative properties. The acidity of this -OH is therefore at its lowest.
Since the electronegativity in the other three cases is not as great as it is for F, we should now search for the halogens' -I effect.
In terms of electronegativity, Cl >Br>I is followed, and the -I effect follows the same order.
Q.3 Among the following, the correct order of acidic strength in water of the given substituted phenols:
(A) P-nitrophenol > m-nitrophenol > Phenol > Methylphenol
(B) m-nitrophenol > P-nitrophenol > Phenol > Methylphenol
(C) P-nitrophenol > m-nitrophenol > Methylphenol > Phenol
(D) m-nitrophenol > P-nitrophenol > Methylphenol > Phenol
Answer: (A)
Solution: When electron withdrawing groups are present, the acidity of phenols rises, whereas when electron releasing groups are present, the acidity of phenols lowers.
The para-nitrophenol is more acidic than the meta-nitrophenol because the presence of the -NO2 group at the para position stabilises the phenoxide ion more than when it is present at the meta position.
Thus the correct order of acidity is
P-nitrophenol > m-nitrophenol > Phenol > Methylphenol
Q.4. Among the following, which is more acidic?
(A) Picric acid
(B) Benzoic acid
(C) Both A and B
(D) None of these
Answer: (A)
Solution: 2,4,6- Trinitro phenol (Picric acid) is more acidic than benzoic acid because it contains three -NO2 groups, which are electron withdrawing groups. When an electron withdrawing group is present on a ring, the acidity of the ring is increased; when an electron donating group is present, the acidity of the ring is decreased.
Frequently Asked Questions-FAQs:
Q1. Compared to cyclohexanol, phenol is more acidic. How?
Answer: Whether the conjugate base anion can be stabilised by resonance has a significant impact on a molecule's acidity. For instance, phenol is a stronger acid than cyclohexanol because its conjugate base is substantially more stable than cyclohexanol's conjugate base. This anion's capacity to delocalize the negative charge throughout the ring through resonance, thereby stabilising it, accounts for the increased stability of the phenol conjugate base. Cyclohexanol's conjugate base is less stable because it lacks resonance structures to keep the charge in place.
Q2. Why doesn't phenol react with NaHCO3 (Sodium Bicarbonate)?
Answer: Phenol is acidic by nature. It is, however, a weak acid. Electrons are drawn away from the -OH group by the presence of -OH on phenol and electron conjugation in benzene rings. A weak base is sodium bicarbonate. It accepts electrons readily from stronger acids such as carbonic acid, but lacks the strength to remove the proton from phenol. As a result of the mild acidic character of phenol, NaHCO3 does not react with it.
Q3. When compared to phenol, why are carboxylic acids more acidic?
Answer: An acid is a chemical that rapidly releases H+. The strength of an acid is determined by its capacity to quickly give away H+. The greater the acid's strength, the easier it is to release H+.
Carboxylic acids are more powerful acids than similar alcohols and even phenols because they lose their proton to form a stable conjugate base, the carboxylate ion, which is more resonance stabilised than the alkoxide or phenoxide ion. Carboxylic acid is more acidic than phenol because it has an electron-withdrawing carbonyl group in addition to stable carboxylate ions, whereas phenol is stabilised by resonance structures of phenoxide ions. Additionally, phenol just contains carbon and lacks the two oxygen atoms that share the negative charge in carboxylic acid. Regular alcohol, on the other hand, is a weaker acid because it lacks resonance stabilisation after losing a proton. As a result, these ions are more stable in the following order: carboxylate ion > phenoxide ion > alkoxide ion.
Q4. Explain the Basicity of alcohol?
Answer: Alcohols have conjugate base alkoxide ions that are more basic than hydroxide ions because they are less acidic than water. Alcohol can be transformed into metal alkoxide by using strong bases that react with alcohol violently yet under control, such as sodium/potassium hydride or sodium/potassium metal. When the alkyl substitution is bulky, the steric effect prevents the alkoxide ion from being solvated completely, which leads to reduced stabilisation. Inductive effects also contribute to destabilisation. The equilibrium is thereby tilted in favour of alcohol.
Alcohols can serve as bases by taking in protons from potent acids. It should be noted that conjugate bases of substances whose pKa are higher than an alcohol will deprotonate that alcohol.