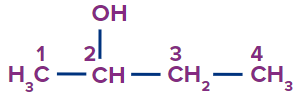-
Call Now
1800-102-2727
Alcohol: Introduction, Classification, Nomenclature, Uses, Practice Problems & FAQs
Every time we enter a doctor's office to receive a shot, we observe that the cotton used to clean the injection site and stop bleeding thereafter is typically soaked in a solution. The solution is mostly ethyl alcohol.
Lower alcohols like ethyl, propyl and isopropyl alcohols have antibacterial and antifungal effects. In situations where soap and water are not easily available, it is used as a disinfectant. Hence they are widely used for sterilising hands, instruments, and laboratories Alcohol is used to disinfect the operating room after surgery to prevent infections. Alcohol was once used on wounds as a disinfectant, antiseptic, and snake bite remedy.
Alcohols are also used as solvent anti-freeze agents.
Table of Contents:
- Introduction of Alcohol
- Classification of Alcohol
- Nomenclature of Alcohol
- Uses of Alcohol
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Alcohol:
Alcohols belong to a specific class of chemical molecules in which the hydroxyl group (-OH) replaces the hydrogen atom of an aliphatic carbon. As a result, an alcohol molecule is made up of two portions, one of which has an alkyl group and the other of which contains a hydroxyl group. The notation for it is R-OH, where R stands for the alkyl group.
Alcohols have a general formula with alkanes, but their functional component, known as the hydroxyl group, has the chemical formula -OH. The most popular type of alcohol, ethanol, is utilised in alcoholic beverages, gasoline, biological specimen preservation, paints and medications, as well as fuel.
The basic formula for the homologous series of alcohols is CnH2n+1OH where n=1,2,3,4.....
Classification of Alcohol:
A) Alcohol classification according to -OH groups:
Depending on how many hydroxyl groups are linked to the carbon atoms, alcohols are divided into four groups: monohydric alcohol, dihydric alcohol, trihydric alcohol, and polyhydric alcohol.
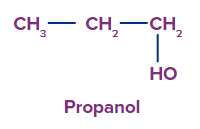
1. Monohydric alcohol: Alcohol with just one hydroxyl group is said to be monohydric alcohol.
Examples include propanol, ethyl alcohol, and methyl alcohol.
The type of hybridization of the carbon atom that the hydroxyl group is connected to helps to further categorise monohydric alcohols.
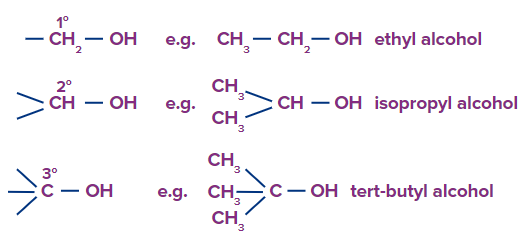
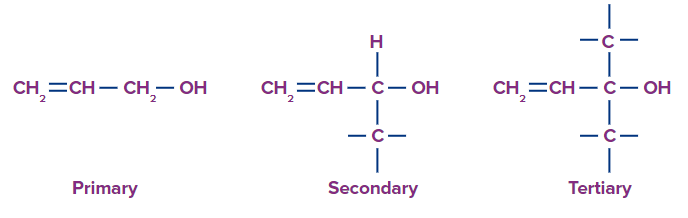
(a) Alcohols containing C(sp3)-OH bond: These alcohols have an alkyl group with an sp3 hybridised carbon atoms linked to the hydroxyl group ( -OH) . R-OH is used to represent these alcohols. They can also be divided into primary, secondary, and tertiary alcohols, depending on which type of carbon atoms the -OH group is connected to.
(b) Allylic alcohols: These alcohols have an allylic carbon, or an sp3-hybridised carbon atom, adjacent to the carbon-carbon double bond. Primary, secondary, and tertiary alcohols can all be found in allylic alcohol.
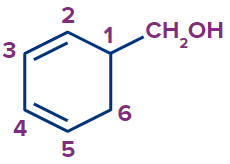
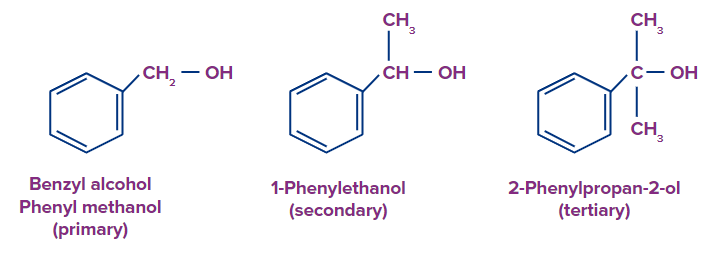
(c) Benzylic alcohol: These alcohols have an aromatic ring and a sp3 hybridised carbon atom adjacent to the hydroxyl group ( -OH). The side chain joined to an aromatic ring contains the hydroxyl group
(-OH). Primary, secondary, and tertiary alcohols can all be forms of benzylic alcohol.
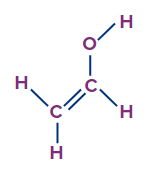
(d) Vinylic alcohols: These monohydric alcohols have a hydroxyl group ( -OH) group that is joined to a vinylic carbon or sp2 hybridised carbon atom. These compounds are also referred to as vinylic alcohols.
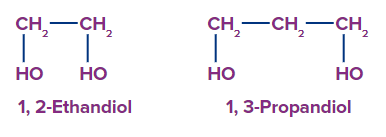
2. Dihydric Alcohol: A category of alcohol with two hydroxyl groups is called dihydric alcohol.
For example: 1,2–Ethanol, 1,3 propandiol.
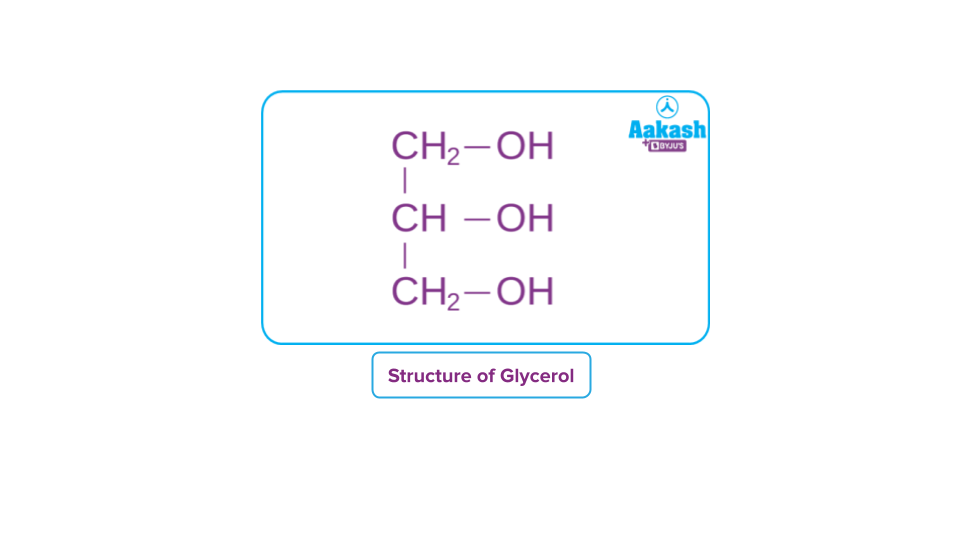
3. Trihydric Alcohol: Three hydroxyl groups are found in trihydric alcohol.
For Example: 1,2,3-Propantriol and 1,2,3-Butanetriol.
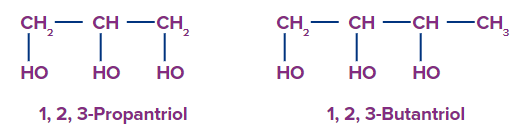
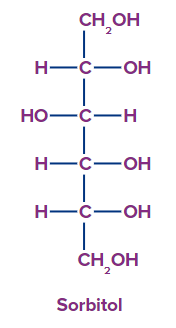
4. Polyhydric Alcohol: Polyhydric alcohol is described as alcohol that has four or more hydroxyl groups.
For example, Sorbitol.
B) Classification of alcohol based on the type of carbon atom bonded to the -OH group:
According to how many carbon atoms are directly linked to the carbon bonded with the hydroxyl group
( -OH) alcohols can be divided into three categories: primary alcohol, secondary alcohol, and tertiary alcohol.
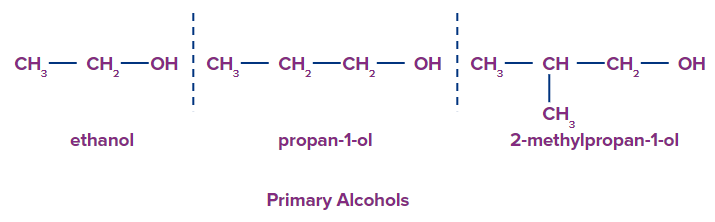
1. Primary Alcohol: In primary alcohols, only one more carbon atom is attached to the carbon bonded with the hydroxyl group. Primary alcohols having two methyl groups at the end are named with the prefix ‘iso’ and the methyl carbon is also included in the word root. The 2-methyl propanol is commonly named isobutanol.
For example, Ethanol, Propan-1-ol, 2-methyl propan-1-ol etc.
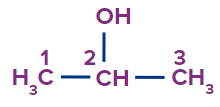
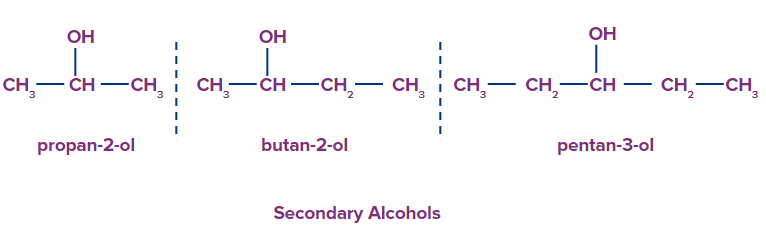
2. Secondary Alcohols: Secondary alcohols include two carbon atoms attached to the carbon bonded with the hydroxyl group.
For example, Propan-2-ol, Bhutan-2-ol, Pentan-2-ol etc
3. Tertiary Alcohols: Tertiary alcohols include three carbon atoms attached to the carbon bonded with the hydroxyl group,
For example, 2-Methyl propan-2-ol, 2-Methyl butan-2-ol etc.

Nomenclature of Alcohols:
A functional group is an atom or collection of atoms that determine the characteristics and particular chemical reactions of an organic molecule.
After the primary suffix in the IUPAC name of the compound, a secondary suffix is added to designate compounds that contain functional groups in order to specify the type of functional group present in the organic compound.
The basic skeleton of the IUPAC naming system is
Secondary prefix + Primary prefix + Root word + Primary suffix + Secondary suffix
Secondary prefix - specifies the substitutes that are affixed to the parent chain.
Primary prefix - specifies the parent chain's nature (cyclo, bicyclo, spiro)
Root Word - specifies how many carbon atoms are in the parent chain.
Primary Suffix - specifies the saturation of the parent chain
Secondary Suffix - specifies the functional group
Alcohols are named by adding a -ol to the end of the parent alkane name, according to the International Union of Pure and Applied Chemistry (IUPAC). The following are some fundamental IUPAC guidelines for naming alcohols:
1. The parent molecule, an alkane with the same number of carbon atoms, is chosen to be the longest continuous chain (LCC) of carbon atoms containing the -OH group. The chain is numbered starting from the end that is near the -OH group.
2. The name of the parent hydrocarbon is prefixed with the number indicating the position of the -OHgroup, and the -e ending of the parent alkane is changed to the suffix -ol. (The carbon atom carrying the -OH group in cyclic alcohols is denoted C1, but the 1 is not used in the name.) As with alkanes, substitutes are given names and numbers.
3. Suffixes like -diol and -triol are used when a molecule contains multiple -OH groups (polyhydroxy alcohols). In these situations, the parent alkane's -e ending is kept.
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
Propan-2-ol |
- |
- |
prop |
ane |
-ol |
|
2 |
Butan-2-ol |
- |
- |
but |
ane |
-ol |
|
3 |
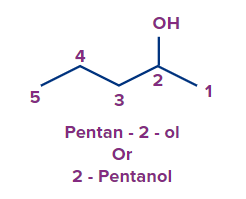
Pentan-2-ol |
- |
- |
Pent |
ane |
-ol |
|
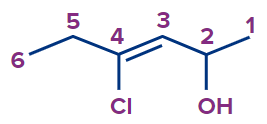
4 |
4-Chlorohex-3-en-2-ol |
4-chloro |
- |
hex |
3-ene |
-ol |
|
5 |
Cyclohexa-2,4-dienyl methanol |
hexa-2,4-dienyl |
cyclo |
meth |
ane |
-ol |
|
6 |
Propan-1,2,3-Triol |
- |
- |
Prop |
ane |
-1,2,3 Triol |
Uses of Alcohols:
- Over 2 billion people consume ethanol (also known as ethyl alcohol) every day. Yeast, sugars, and starch are fermented to create this kind of alcohol. Despite the fact that humans have been consuming it for generations, excessive consumption is harmful to human health. Only a small amount of ethanol can be metabolised by the human liver. Additionally, it affects cognitive and motor abilities and depresses the central nervous system (CNS).
- In order to prevent excessive alcohol intake, ethanol and methanol are frequently combined. Denaturing ethanol is the name given to this procedure. Since the resulting mixture, also known as methylated spirit, is dangerous, ethanol should not be consumed.
- In order to dissolve numerous organic molecules that are immiscible with water, ethanol is employed as a solvent. It is a component of paints, cosmetics, detergents, inks, and scents.
- Due to its ability to inhibit or stop the growth of microorganisms, ethanol possesses antibacterial characteristics. The microorganisms' protein structures are changed, which makes them incapable of carrying out their intended functions. When the bonds holding the protein together to maintain its specific form are disrupted, a process known as denaturation takes place, which results in a protein-losing its structure. Because of this, ethanol is a component of hand sanitisers and hand wipes.
- Biofuels are being looked at more and more as potential replacement fuels for the future because fossil fuels are non-renewable and contribute to global warming.
- Because ethanol burns completely, it serves as a clean fuel. As a result, it does not significantly increase pollutants. It produces water and carbon dioxide while it burns with a clear, smokeless flame.
- Propanol is employed as a solvent, which is one of its most popular applications. Other solvents, including coolant, lacquer formulations, detergents, colour solutions, window cleaning, and more are produced using it. The most prevalent form of propanol used in the printing industry is isopropanol or isopropyl alcohol, particularly for flexographic ink.
- Butanol is a potent, environmentally friendly solvent that may be used to clean and polish items. Additionally, butanol can be used as fuel. Numerous foods and beverages contain n-butanol, which is also used to flavour food artificially.
Practice Problems:
Q1. What is the following compound's IUPAC name?
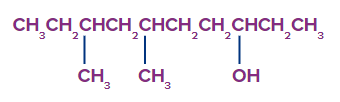
a. 5-hydroxy-2,3-dimethyldecane
b. 1-ethyl4,6-dimethyloctanol
c. 3-hydroxy-6,8-dimethyl decane
d. 6,8-dimethyldecan-3-ol
Answer: Option D.
Solution: The longest carbon chain (LCC) is a derivative of decane with ten carbon atoms, and the third carbon atom's hydroxyl groups (-OH) turns the combination into a 3-decanol.
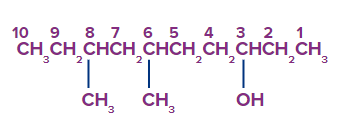
Beginning with the end nearest the OH group, the carbon atoms are numbered. The two methyls (CH3) groups are then fixed in place at positions six and eight. It is known as 6,8-dimethyl-3-decanol
(not 3,5-dimethyl-8-decanol).
Q2. What is the following compound's IUPAC name?
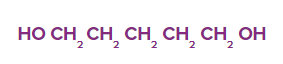
Solution: The longest carbon chain (LCC) is a pentane derivative due to its five carbon atoms. Because of the presence of two hydroxyl groups (-OH) groups on the first and fifth carbon atoms, the chemical is classified as a diol and is known as 1,5-pentanediol.
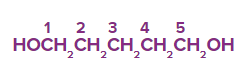
Q3. What is the following compound's IUPAC name?
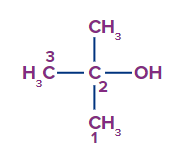
a. 2,2-dimethyl ethanol
b. Tertiary butanol
c. 2-methylpropan-2-ol
d. Both B and C
Solution: Option D
The given compound is alcohol.
Word root: The parent chain is of three carbon atoms. So, prop will be the word root.
Prefix: 2-methyl
Primary suffix: ane
Secondary suffix: -ol.
Therefore, the compound IUPAC name is 2-Methylpropan-2-ol or 2-Methyl 2-propanol and its common name is tert-butanol.
Q.4. What is the following compound's IUPAC name?
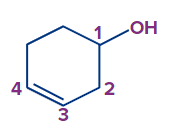
Solution: The given compound is alcohol.
Word root: The parent chain is of six carbon atoms. So, hex will be the word root.
Prefix: Cyclo
Primary suffix: 3-ene.
Secondary suffix: -ol.
Hence, the compound's IUPAC name is 3-cyclohexen-1-ol
Frequently Asked Questions(FAQs):
Q1. What is the test to distinguish between primary alcohol, secondary alcohol and tertiary alcohol?
Answer: The Lucas Test is the method used to discriminate between primary, secondary, and tertiary alcohols. When the reactivity of alcohol is examined in this test using the Lucas reagent, each type of alcohol yields distinct results. A strong hydrochloric acid solution of anhydrous zinc chloride is known as a Lucas reagent.
If the unknown sample contains primary alcohol, the solution won't become turbid at room temperature after adding the Lucas reagent. However, if the solution is heated for 30 to 45 minutes, the solution can occasionally become cloudy.
If the unknown sample contains secondary alcohol, the solution will become cloudy after 3 to 5 minutes at room temperature following the addition of the Lucas reagent.
If tertiary alcohol is present in a sample, applying the Lucas reagent at room temperature will quickly cause the solution to get cloudy.
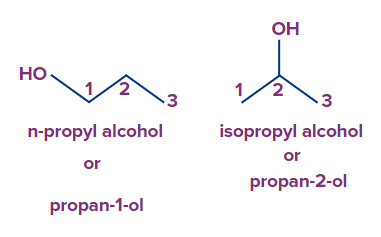
Q2. Why do n-propyl alcohol and isopropyl alcohol position isomers?
Answer: Position isomers are constitutional isomers with different locations of the functional group but the same carbon skeleton and functional groups. In n-propyl alcohol, the first carbon atom has the -OH group attached to it. In isopropyl alcohol, the second carbon atom contains the -OH group.
Q3. Why do tertiary alcohols react with chemicals more quickly than primary and secondary alcohols?
Answer: Because there are more alkyl groups in tertiary alcohols, the inductive (+I) effect is stronger. As a result, the charge density surrounding the oxygen atom and the carbon atom increases. As a result, the C-O connection can be broken more easily. The result is the formation of a tertiary carbocation, which is more stable than secondary carbocation and primary carbocation.
Q4. Why is methanol consumption harmful to humans?
Answer: Although consuming methanol is a rare event, its negative consequences on the body cannot be ignored. Despite not being hazardous in and of itself, methanol turns into formic acid when taken. This formic acid can cause severe metabolic acidosis because of its high toxicity. The optic nerve can be harmed by 10 mL of methanol at low concentrations, which can lead to blindness that lasts a lifetime. Hand sanitisers are made using ethanol since it is considerably safer than methanol.