-
Call Now
1800-102-2727
Addition Polymerisation – Definition, Mechanism of Chain Growth Polymerisation in Polyethylene, Difference between HDP and LDP
We must be familiar with products like plastic bags, plastic bottles, trays, garbage bags etc. in our day-to-day life. But, do you know how these types of plastic products are made?
These products are the polymers of ethene molecules which combine with each other under suitable conditions to form polyethylene. This process of combining a large number of simple molecules is known as polymerisation. The ethene molecules are converted into polymers by a process called addition polymerisation.

Let’s get to know more about addition polymerisation, its types, mechanism and the difference between high-density polyethylene (HDPE) and low-density polyethylene (LDPE) on this concept page.
TABLE OF CONTENTS
- Addition Polymerisation – Definition
- Addition Polymerisation – Types
- Mechanism of Chain Growth in Polyethylene
- Difference Between HDPE and LDPE
- Cationic Addition Polymerisation
- Anionic Addition Polymerisation
- Examples of Addition Polymerisation
- Practice Problems
- Frequently Asked Questions – FAQ
Addition Polymerisation – Definition
The molecules of the same or different monomers add on one another in addition polymerisation, resulting in the production of a macromolecule/polymer. The molecular formula of the repeating unit of the polymer is the same as the parent monomers. Addition polymerisation happens most commonly in compounds with multiple bonds. As this type of polymerisation results in an increase in the chain length, it is also called chain growth polymerisation.

Addition Polymerisation – Types
Based on the type of initiators, addition polymerisation can be classified into
- Free-radical addition polymerisation
- Cationic addition polymerisation
- Anionic addition polymerisation
Mechanism of Chain Growth in Polyethylene
Polyethylene is one of the most important polymers that can be synthesised by a free radical polymerisation process. Free-radical polymerisation process consists of three steps, namely
- Chain initiation
- Chain propagation
- Chain termination
Chain Initiation: In free-radical polymerisation, the initiator molecule is a free-radical. The initiator is a molecule that initiates the polymerisation process. Tert-butyl peroxide and benzoyl peroxide are the common free-radical initiators. Under mild conditions, these initiators decompose into tert-butoxide and benzoyl peroxide radicals, respectively. These reactive intermediates react with the monomer to form a larger free-radical molecule.
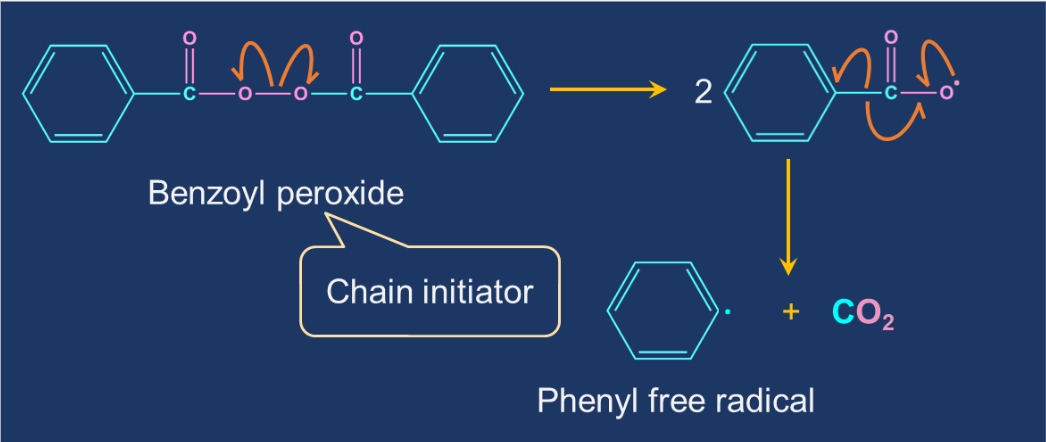
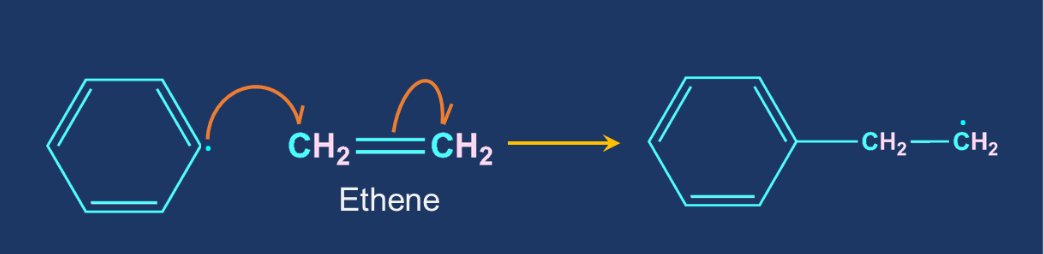
Chain Propagation: The repeated addition of monomers to the reactive intermediates result in the formation of the products.
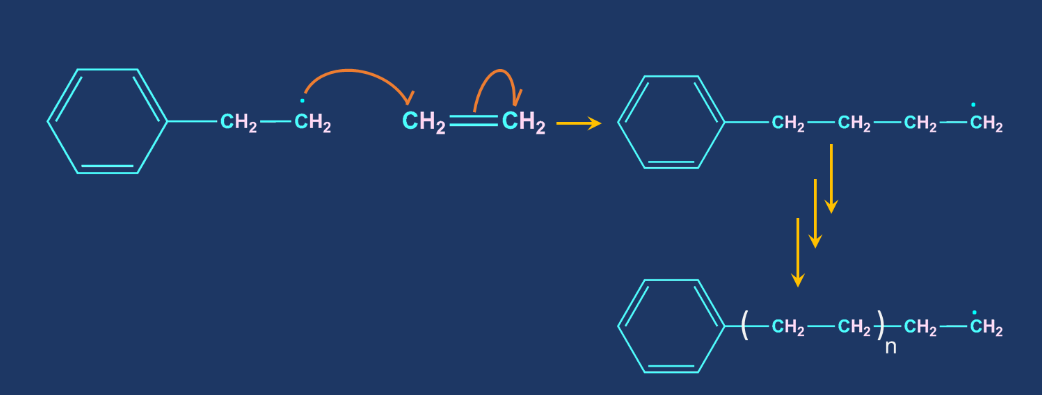
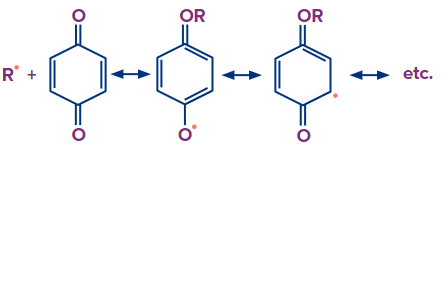
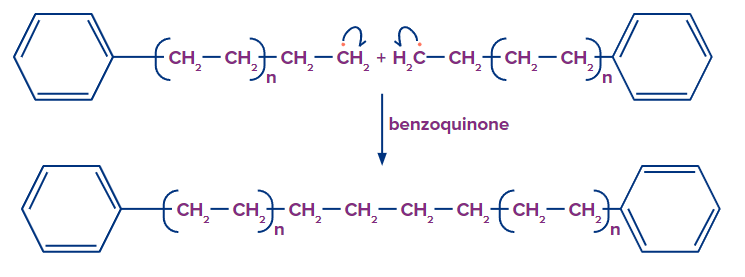
Chain Termination: Chain termination occurs when one radical combines with another radical. It takes place in the presence of a benzoquinone inhibitor. A reactive intermediate (R*) is formed in the free radical polymerisation, which helps in the chain propagation step. Benzoquinone captures this reactive intermediate and transforms it into a non-reactive radical. The chain propagation step comes to an end if this reactive intermediate is not present.
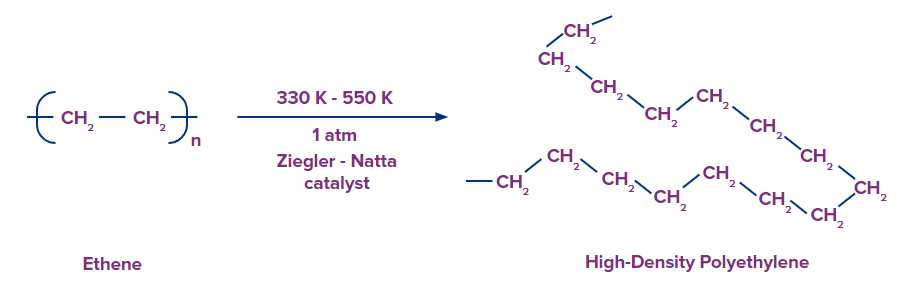
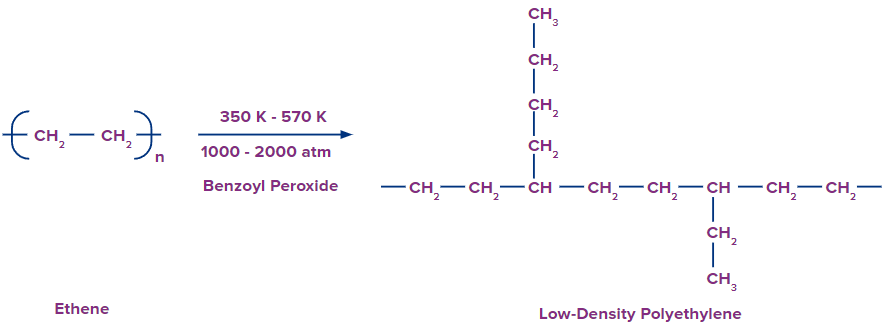
Difference Between HDPE and LDPE Polymer
|
High-Density polyethylene (HDP) |
Low-Density polyethylene (LDP) |
|
|
Polymerisation reaction |
|
|
|
Catalyst used |
Ziegler-Natta catalyst {(C2H5)3Al and TiCl4} is used for the reaction. |
Benzoyl peroxide (C6H5C(=O)O-)2)catalyst is used for the reaction. |
|
Temperature and pressure for reaction |
Reaction takes place at a temperature range of 330 K to 550 K and pressure range of 1 atm to 3 atm. |
Reaction takes place at a temperature range of 350 K to 570 K and high-pressure range of 1000 atm to 2000 atm. |
|
Properties |
They are chemically inert, tougher and harder than LDPE and poor conductors of electricity. |
They are chemically inert, flexible and poor conductors of electricity. |
|
Examples of product formed |
It is used in making toys, ice trays, fishing nets, ropes etc |
It is used for the production of garbage bags, pipes and fittings, lamination, squeeze bottles etc. |
Cationic Addition Polymerisation
The monomers having electron-donating groups favour cationic mechanisms of addition polymerisation. The electron-donating groups stabilise the cations formed. Isobutylene undergoes a cationic mechanism as it has two methyl groups. Cationic addition polymerisation consists of three steps, namely
- Chain initiation
- Chain propagation
- Chain termination
Chain initiation: In the cationic mechanism of addition polymerisation, the initiator is cationic in nature. In this step, the acid attacks the double bond of the monomer, resulting in a cationic species.
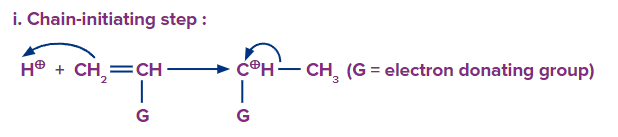
Chain propagation: A bigger cation is formed by the repeated addition of the monomer molecules.
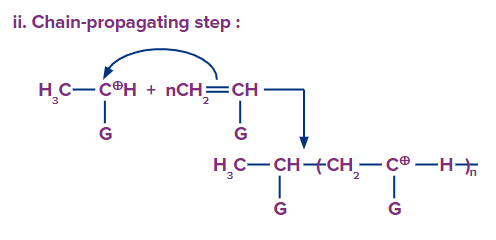
Chain termination: Termination occurs by the loss of a proton.
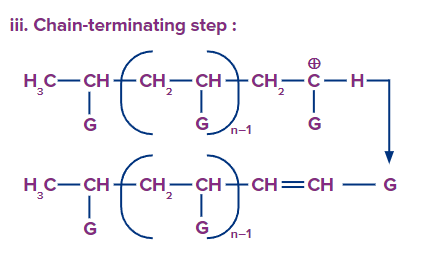
Anionic Addition Polymerisation
The monomers having electron-withdrawing groups favour anionic addition polymerisation. The electron-withdrawing group stabilises the intermediate anion formed. Anionic addition polymerisation consists of three steps, namely
- Chain initiation
- Chain propagation
- Chain termination
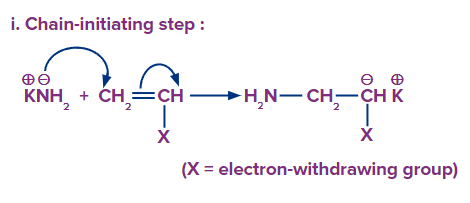
Chain initiation: In the anionic mechanism of addition polymerisation, the initiator is anionic in nature. In this step, the base attacks the double bond of the monomer, resulting in the formation of an anionic species. n-Butyl lithium and potassium amide are a few examples of anionic initiators.
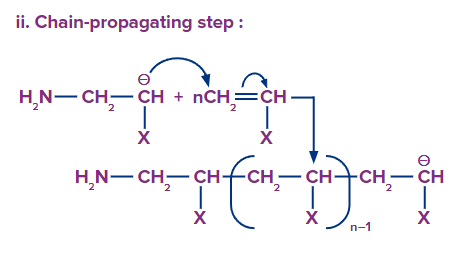
Chain propagation: The anion formed in the first steps adds to the double bond of another monomer and the process continues.
Chain termination: The chain is terminated by the addition of acid.
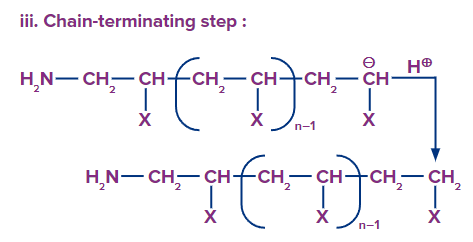
Examples of Addition Polymerisation
Low-Density Polyethylene
It is produced when ethane is polymerised under specific circumstances of high pressure (1000–2000 atm) and high temperature (350–570 K) with O2 or a peroxide initiator.
Characteristics of LDPE:
- Chemically inert
- Tough
- Flexible
- Poor conductor of electricity
Uses of LDPE: In electric wires, pipes, bottles, wrappers, toys, etc,.
High-Density Polyethylene
It is obtained by the addition polymerisation of ethene in a hydrocarbon solvent in the presence of Ziegler-Natta catalyst (TiCl4+(C2H5)3Al).
Characteristics of HDPE:
- High density
- Chemically inert
- Tough and hard
- Better conductor of electricity when compared to LDPE
Uses of HDPE: In bottles, pipes, plates, buckets, dustbins, etc,.
Teflon
It is manufactured by heating tetrafluoroethene with any free radical or persulphate catalyst at high pressure.
Characteristics of Teflon:
- Chemically inert
- Resistant to attack by corrosive agents
Uses of Teflon: In oil seats, gaskets, as a surface coating agent in non-stick cookware, etc,.
Polyacrylonitrile
It is obtained by the addition polymerisation of acrylonitrile in the presence of a peroxide catalyst. It is used in acrylic fibres.
Recommended Video
Polymers Class 11 & 12 Chemistry in One Shot (Formula and Practice Questions) | NEET 2022 Chemistry
Practice Problems
1. The monomer that has the greatest ability to undergo cationic addition polymerisation is __________ .
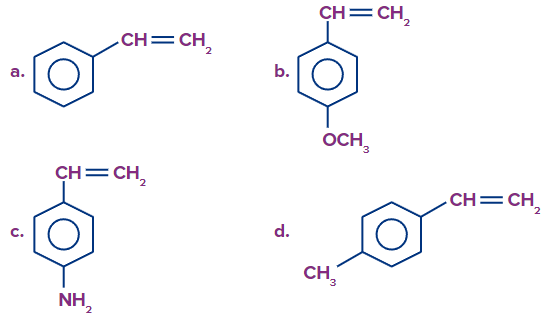
Answer: C
Solution: The monomers containing strong electron-donating groups undergo cationic addition polymerisation. This is because the electron-donating group stabilises the intermediate cation formed in the chain propagation step.
Among the given compounds, compound (C) has a stronger electron-donating group i.e. -NH2 (+M effect) than the electron-donating groups given in other options. Therefore, compound (C) has a greater ability to undergo cationic addition polymerisation.
So, option C is the correct answer.
2. The monomer that has the greatest ability to undergo anionic addition polymerisation is __________ .
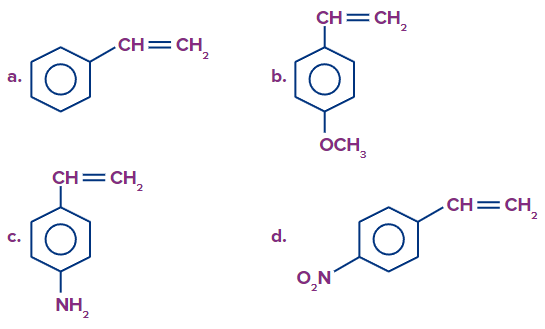
Answer: D
Solution: The monomers containing strong electron-withdrawing groups undergo anionic addition polymerisation. This is because the electron-withdrawing group stabilises the intermediate anion formed in the chain propagation step.
Among the given compounds, compound (D) has the stronger electron-withdrawing group i.e. -NO2 (-M effect) than the electron-withdrawing groups given in other options. Therefore, compound (D) has a greater ability to undergo anionic addition polymerisation.
So, option D is the correct answer.
3. Free radical polymerisation requires a free radical initiator. The most commonly used free radical initiator is __________ .
- Ph-CO-O-O-CO-Ph, benzoyl peroxide
- (CH3)3C-O-O-C(CH3)3, tert-butyl peroxide
- C6H5-N=N(C6H5)→O, azoxybenzene
- CH2N2, diazomethane
Answer: A
Solution: In free-radical polymerisation, the initiator molecule is a free-radical. Initiator is a molecule that initiates the polymerisation process. Benzoyl peroxide is the most common free-radical initiator.
For example, let us consider the initiation step in polyethylene formation:
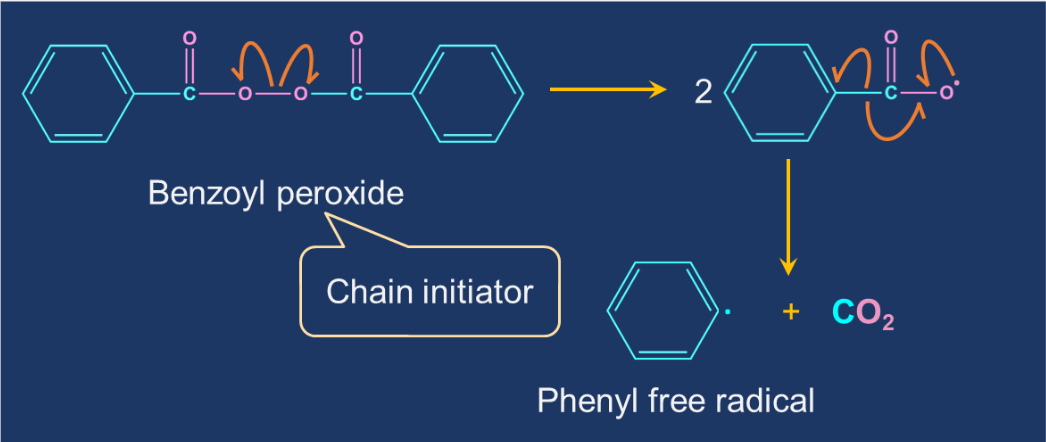
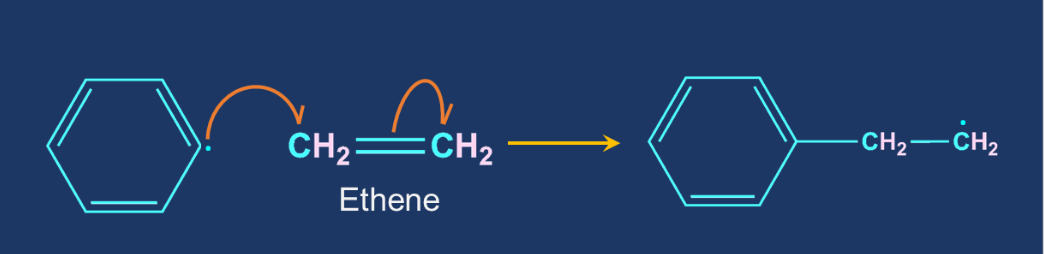
So, option A is the correct answer.
4. Which of the following species can best serve as an initiator for radical polymerisation?
- ROH
- ROR
- ROOR
- RCOOR
Answer: C
Solution: In free-radical polymerisation, the initiator molecule is a free-radical. Initiator is a molecule that initiates the polymerisation process. ROOR is the common representation of the alkyl peroxide group. R represents an alkyl group present in the given compound.
So, option C is the correct answer.
Frequently Asked Questions – FAQ
1. What is the role of benzoquinone in the chain terminating step of the free radical mechanism of chain growth polymer?
Answer: Chain termination occurs when one radical combines with another radical. It takes place in the presence of a benzoquinone inhibitor. A reactive intermediate (R*) is formed in the free radical polymerisation, which helps in the chain propagation step. Benzoquinone captures this reactive intermediate and transforms it into a non-reactive radical. The chain propagation step comes to an end if this reactive intermediate is not present.

2. Why is polyethylene harmful to the environment?
Answer: Polyethylene is harmful to the environment as most of the polyethylene products are non-biodegradable in nature. They cannot be degraded into simpler units by microorganisms or enzymes and results in pollution and climate change.
3. How can we differentiate polyethylene and plastics?
Answer: Plastic can be considered as a broad category in which polyethylene is one example of it. Other examples of plastics include bakelite, melamine etc. Therefore, it can be said all polyethylene products are plastics but vice-versa is not true.
4. What are the other examples of molecules undergoing addition polymerisation?
Answer: In case of addition polymerisation, monomers combine together in the presence catalyst and at a suitable temperature and pressure to form a compound of large molecular mass. Few examples of addition polymer includes- Buna-S, Buna-N, Teflon, polyethylene, polyacrylonitrile (PAN).