-
Call Now
1800-102-2727
Addition of HOX (Hypohalous Acid) - Addition of Hypohalous Acid to an Alkene, Alkynes, Practice problems and FAQs
Everyone enjoys cheese, especially homemade cheese. Right????
How do we make cheese at home?

We all know that it is formed by milk coagulation, as studied in surface chemistry. However, chemistry states that the breakdown of milk proteins to produce cheese is accomplished by biocatalysts, which are enzymes derived from biological sources or whole cells that are used to accelerate chemical reactions.
A versatile biocatalyst is a halohydrin derivative. These enzymes can be used in the pharmaceutical, agrochemical, and chemical industries to synthesize important building blocks and precursors.
Let us talk about how they will form!
Table of Contents:
- Addition of Hypohalous acid to alkene–Halohydrin
- Addition of hypohalous acid to alkynes
- Practice Problems
- Frequently Asked Questions
Addition of Hypohalous acid to Alkenes–Halohydrin:
Halohydrins are formed by electrophilic addition of other halogen-containing reagents, such as hypohalous acids, HOX, to an alkene. The halogen is the electrophilic element in each of these reagents. The electronegativity of oxygen is higher than that of all halogens except flourine, O has δ− charge and X has δ+ charge in HOX.
Example 1: Addition of HOBr to ethene to form 2-Bromoethanol

Example 2: Addition of HOBr to propene to form 1-Bromopropan-2-ol
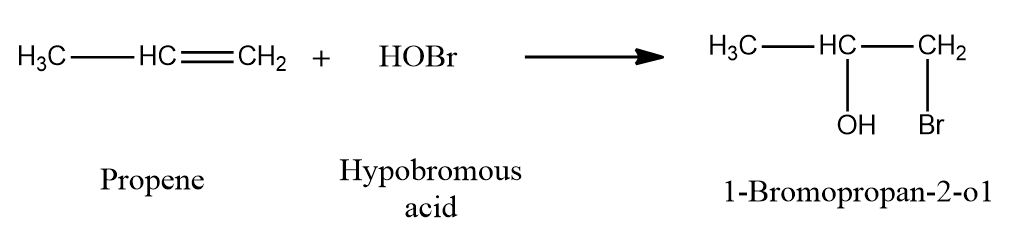
Halohydrins, on the other hand, are not generated by adding a hypohalous acid directly to an alkene; instead, the alkene is reacted with Br2 or Cl2 in the presence of water. Let us discuss in detail through mechanisms.
Mechanism:
Step-1: The first step is the same as when alkenes are halogenated. A cyclic bromonium ion is formed when a halogen, in this case bromine, interacts with an alkene.
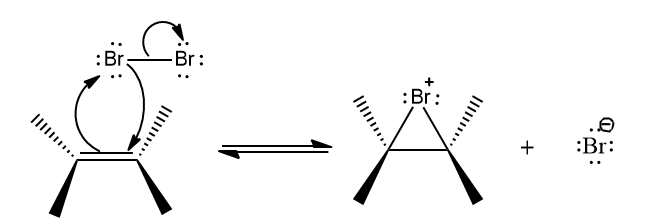
Step-2: The bromine ion is not attacked in step two; instead, water acts as the nucleophile. Because water is more abundant than bromine ion, it is more likely to be available in the proper position for nucleophilic attack. The nucleophile property of water is superior to that of the bromine ion. The more powerful a nucleophile is, the more likely it will attack. The attack of Nucleophile is preferred on more substitute carbon.

Step-3: It is the final acid-base step, in which a proton is lost from oxygen to the solvent (water), resulting in the neutral halohydrin. The result of the addition reaction in this case is a bromohydrin.

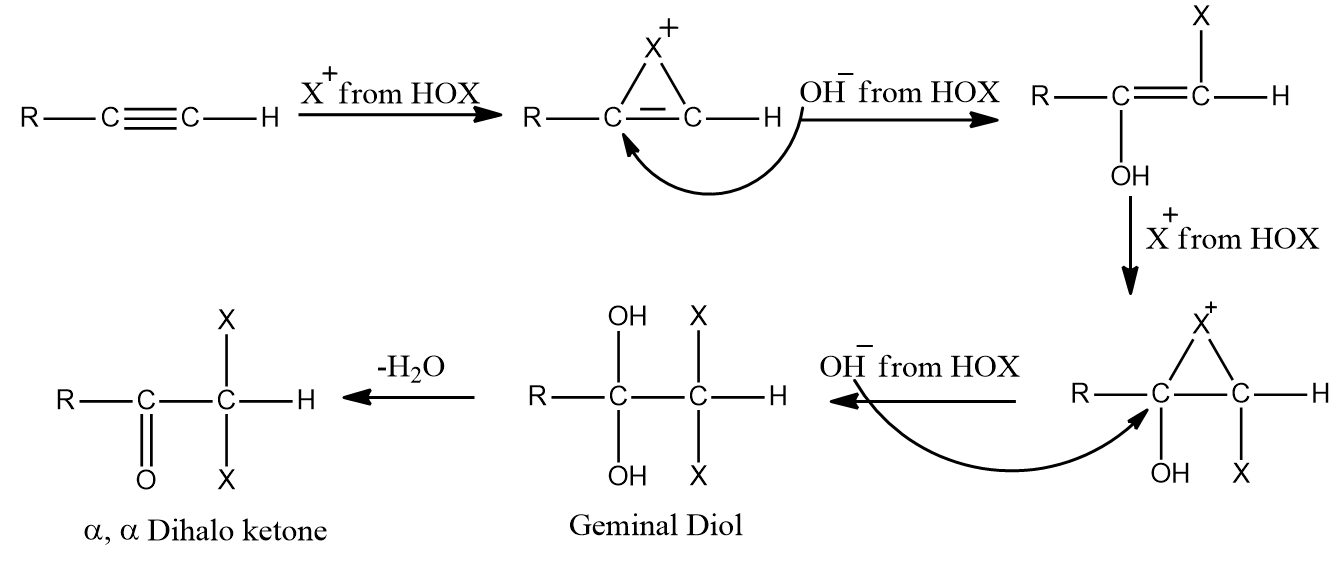
Addition of Hypohalous acid to Alkynes:
Except for fluorine, HOX breaks down into OH- and X+ because oxygen has a higher electronegativity than halogens. Two HOX molecules (HOCl, HOBr, or HOI) are added to alkynes to form geminal diols.
On the removal of H2O, these geminal diols form dihalo ketones (two halogen groups are present at the position with respect to the carbonyl group). The geminal diols are extremely unstable. Dehydration easily converts them to their respective ketones or aldehydes (loss of one water molecule). Generally, the -OH group is added to the carbon atom of an alkyne where the positive charge is more stabilised.
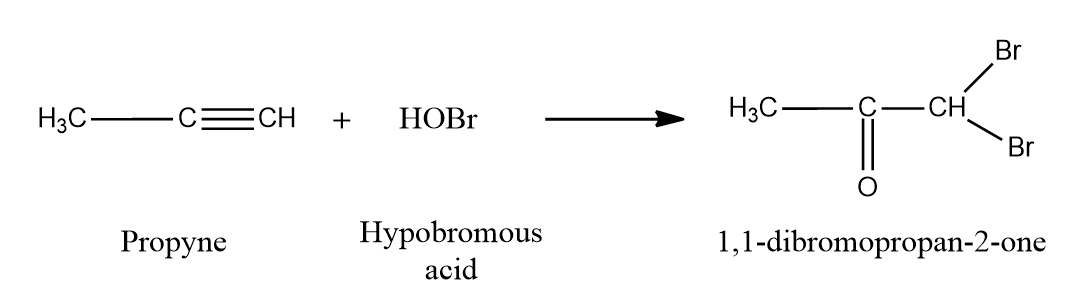
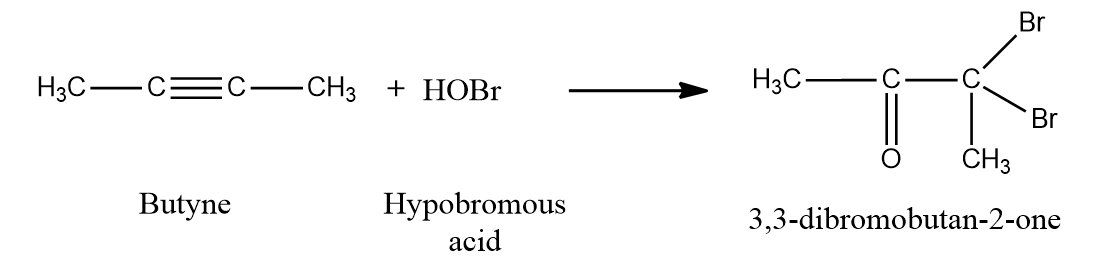
Mechanism:
When HOX is introduced to an alkyne, the electrons shift in such a way that the positive charge is present on the carbon connected to the alkyl (-R) group, which is stabilized by the alkyl group's +I effect and hyperconjugation. The electrons shift in such a way that a positive charge is present on the carbon linked to the -OH group, when the second molecule of HOX is added to the alkene produced previously, as the positive charge is stabilized by the +M effect of the -OH group.
Practice Problems:
Q1. What is the product of the following reaction?
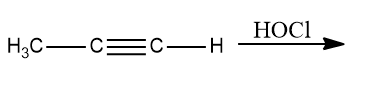
a. 1,1-dichloropropan-2-one
b. 1,2-dichloropropan-2-one
c. 2,3-dichloropropane-2,3-diol
d. 1,2-dichloropropane-1,2-diol
Answer: (A)
Solution: HOCl, with the exception of fluorine, degrades into OH- and Cl+ because oxygen has a higher electronegativity than halogens. To form geminal diols, two HOCl are added to alkynes. Geminal diols are highly unstable.
They are easily converted to their respective ketones or aldehydes by dehydration (loss of one water molecule).
Hence, the correct option is (A).
Q2. What is the product of the following reaction?
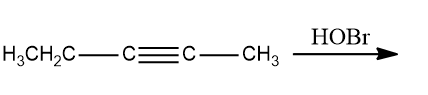
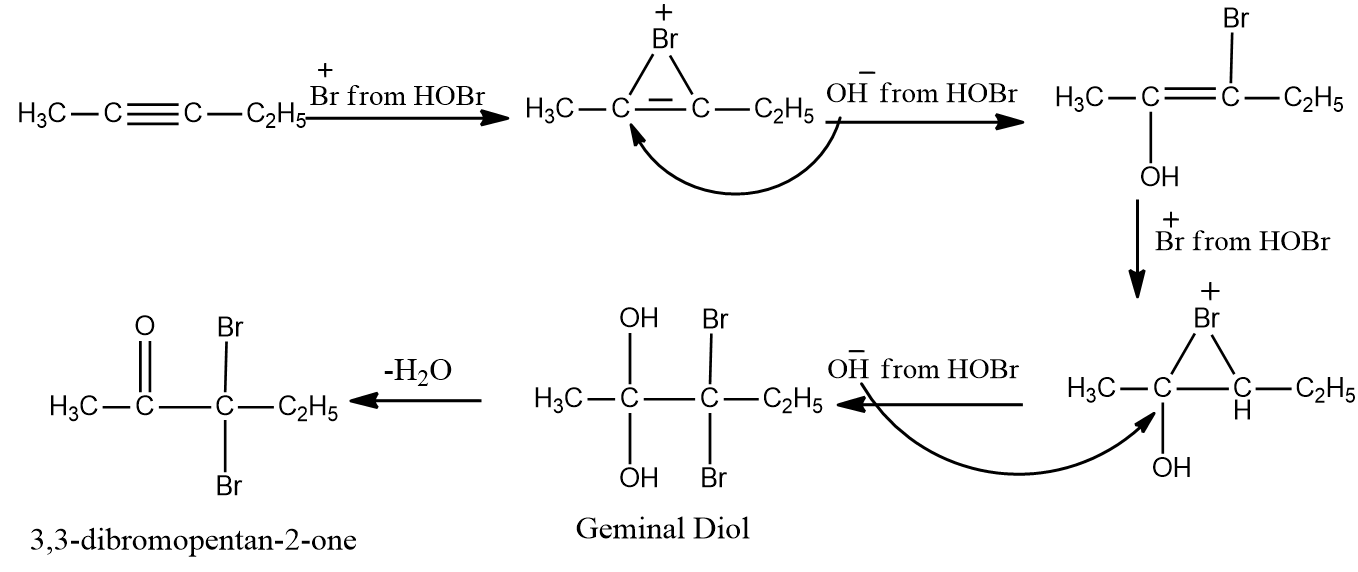
a. 2,3-dibromopentane-2,3-diol
b. 2,2-dibromopentan-3-one
c. 3,3-dibromopentan-2-one
d. 2,2-dibromopentane-1,2-diol
Answer: (C)
Solution:HOBr, with the exception of fluorine, degrades into OH- and Br+ because oxygen has a higher electronegativity than halogens. To form geminal diols, two HOBr are added to alkynes. The addition of HOBr takes place as per the stability of intermediates. Geminal diols are highly unstable.
They are easily converted to their respective ketones or aldehydes by dehydration (loss of one water molecule).
Hence, the correct option is (C).
Q3. What is the product of the following reaction?
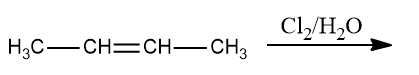
a. Butane-1,2-diol
b. 3-chlorobutan-3-ol
c. 3-chlorobutan-2-ol
d. 2-chlorobutan-2-ol
Answer: (C)
Solution:HOCl, with the exception of fluorine, degrades into OH- and Cl+ because oxygen has a higher electronegativity than halogens. To form geminal diols, two HOCl are added to alkynes. Geminal diols are highly unstable.
They are easily converted to their respective ketones or aldehydes by dehydration (loss of one water molecule).
Hence, the correct option is (C).
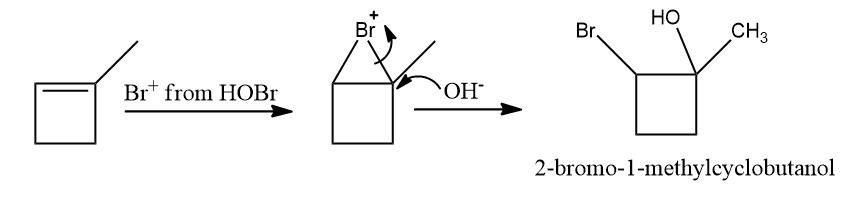
Q4. What is the product of the following reaction?

a. 2-bromo-2-methylcyclobutanol
b. 3-bromo-2-methylcyclobutan-2-ol
c. 1-bromo-1-methylcyclobutanol
d. 2-bromo-1-methylcyclobutanol
Answer: (D)
Solution: HOCl, with the exception of fluorine, degrades into OH- and Cl+ because oxygen has a higher electronegativity than halogens. To form geminal diols, two HOCl are added to alkynes. Geminal diols are highly unstable.
They are easily converted to their respective ketones or aldehydes by dehydration (loss of one water molecule).
Hence, the correct option is (D).
Frequently Asked Questions-FAQs:
1. What is a halohydrin, and what does it do?
Answer: Halohydrin is a functional group in which neighbouring carbon atoms are linked by hydroxyl and halogen groups. Depending on the halogen present, halohydrins are classified as fluorohydrins, chlorohydrins, bromohydrins, or iodohydrins.
2. What purposes do unsaturated hydrocarbons serve?
Answer: In various industrial processes for the creation of insecticides, gasoline, paints, lacquers, and other products, unsaturated hydrocarbons are frequently used. They are additionally utilized in various chemical processes to create new compounds.
3. Is the synthesis of halohydrin anti-Markovnikov?
Answer: The Halohydrin Formation reaction involves breaking a pi bond and replacing it with a halohydrin. Hydrin = OHand Halo = halogen. In water, this reaction produces an anti-addition reaction that follows Markovnikov's rule. As a result, the synthesis of halohydrin is not anti-Markovnikov.
4. What exactly is Halo alcohol?
Answer: A halohydrin, also known as a haloalcohol or -halo alcohol in organic chemistry, is a functional group consisting of a halogen and a hydroxyl linked to adjacent carbon atoms.


