-
Call Now
1800-102-2727
Acids and Bases
Acids and Bases
Acids and bases are substances with unique properties such as opposite extreme pH values, corrosion, different taste, and reaction mechanisms. Commonly found in household items and kitchens, they find wide industrial applications. Multiple famous scientists have contributed their definitions to explain their state of occurrence and reactivity in different natures of solvents.
Table of Contents
- What are acids?
- What are bases?
- Arrhenius Concept
- Bronsted-Lowry Concept
- Lewis Concept
- Practice Problems
- Frequently Asked Questions
What Are Acids?
Acids are substances capable of releasing protons into the solution, thus imparting acidic nature. Holding the ability to corrode metals, they taste sour and neutralise bases by their ability to release hydrogen ions that bind with hydroxyl ions of bases. The acids turn blue litmus red, while red litmus will exhibit the same colour. They also react with metals to release hydrogen gas and have low pH from 0 to 6.
What Are Bases?
Bases are capable of releasing hydroxyl ions or accepting protons. They impart basic nature to the solution. They can corrode concrete and also different metals in the presence of moisture. The bases taste bitter in water, are harsh and have a soapy texture. These are neutralised by acids whose protons fuse with the hydroxyl ions of bases to produce salt and water. The bases turn red litmus blue while blue litmus remains unchanged and has a pH of 7 to 14.
Arrhenius Concept
He introduced the earliest modern acid and base concept in 1884. He defined the acid as a substance capable of releasing hydrogen or hydronium ions in aqueous solutions. While the base, according to him, was capable of releasing hydroxyl ions in an aqueous solution. He failed to explain the acid and base in non-aqueous solutions. Simultaneously, he could not provide information on BF3, NH3 and other similar compounds. The limitation was its applicability to aqueous solutions only and its inability to explain nonmetal and metal oxides' acidic and basic nature.
Bronsted-Lowry Concept
The scientists Bronsted and Lowry individually defined the acids and bases in terms of proton release in acid-base reactions in 1923. They defined an acid as a chemical substance containing hydrogen that donates a proton and a base as a chemical substance containing hydrogen that accepts a proton. It referred to acid as protongenic or proton donors and base as protophilic or proton acceptors.
Conjugate Acid-Base Pairs
The theory led to the identification of conjugate acids and bases. The loss of hydrogen by acid would lead to the formation of a conjugate base, and accepting of the proton by base would result in conjugate acid formation. In the example of the reaction between ammonia and hydrochloric acid, we have HCl as an acid and NH3 as a base. The conjugate acid will be ammonium ion (NH4+), and the conjugate base will be chloride ion (Cl-).
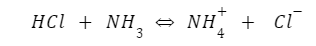
Lewis Concept
At the same time period of 1923, Lewis gave a broader and detailed aspect of acid and base. It dealt with acid and base in terms of electrons. Thus, he defined an acid as capable of accepting electron pairs and a base as capable of donating electron pairs. It covered acids such as incomplete octets of the central atom, simple cations and molecules with vacant-d orbital-like AlCl3, BeCl2, Ag+, SF4. It also covered bases with lone pairs, charges, and coordination complexes like NH3, Cl-, CN. It also provided another perspective for scientists to understand their nature in reactions.
The Lewis concept still posed limitations in an inability to address the magnitude of relative acid and base strength and behaviour of protonic acids such as H2SO4.
Practice Problems
Q1. What is the pH of gastric juice and blood plasma?
A. 1 and 7
B. 2 and 14
C. 14 and 2
D. 7 and 1
Ans. a. 1 and 7
The actual pH range of gastric juice is 1 to 3, and blood plasma is 7.3 to 7.4.
Q2. Which concept could not explain reactions in non-protonic solvents?
A. Lewis Concept
B. Lowry Concept
C. Arrhenius Concept
D. Bronsted-Lowry Concept
Ans. d. Bronsted-Lowry Concept
Bronsted-Lowry Concept explains reactions only in aqueous solutions.
Q3. What will be the conjugate acid in the ammonia and water reaction?
A. Ammonia
B. Water
C. Ammonium ion
D. Hydroxyl ion
Ans. c. Ammonium ion
The conjugate acid will be an ammonium ion, while the conjugate base will be a hydroxyl ion due to its unique capability to accept hydroxyl and hydrogen ions.
Frequently Asked Questions
Q1. What is the product of the acid and base reaction?
Ans.The acids and bases react to form salt and water. For instance, hydrogen chloride and sodium hydroxide react to yield salt, sodium chloride and water.
Q2. What are amphiprotic and aprotic solvents?
Ans. Amphiprotic solvents are both protophilic and protogenic. For example, ammonia and water. Aprotic solvents can not accept or donate protons. For example, carbon tetrachloride and benzene.
Q3. What are monobasic, dibasic and tribasic acids?
Ans. Acids capable of releasing one, two and three protons are monobasic, dibasic and tribasic acids, respectively. For example, hydrochloric acid, phosphorous acid and phosphoric acid.


