-
Call Now
1800-102-2727
Acidity and pH- Acidity, pH and pOH of acids & Base, Practice Problems and FAQs
You have heard about many acids. hydrochloric, sulphuric acetic, citric acid and so on. You may say that all acids release H+ ions in water and base releases OH- ions in water. You also know that each of them is different - we can use vinegar (acetic acid) in food but not hydrochloric acid (HCl).
Similarly you might have seen panwala using a white paste of lime( a base) but not sodium hydroxide.
But can you answer he following question that may be necessary to explain the above facts?
What is the difference between HCl and vinegar? Is it the concentration of ions released?
If yes, how do we quantify the ionic concentrations in them and relate it to understand their behavior?
Is there any relation between H+ and OH- ions in aqueous solution?
You will see answer for all these and many more questions here. Come let us read further.

Table of Content
- pH and pOH
- Relationship between pH, pOH and Kw
- pH of a weak acid
- pH of weak base
- Practice Problems
- Frequently Asked Questions FAQ
pH and pOH
Any acid releases protons in water. Dissociation of weak acid CH3COOH can be written as :
CH3COOH(aq) ⇌ CH3COO-(aq) + H+(aq).
Similarly any base releases hydroxide ions in water. Dissociation of weak base NH4OH can be written as :
NH4OH(aq) ⇌ NH4+(aq) + OH-(aq).
Sørensen invented this scale used to determine the concentration of H+ & OH-ions.
pH is defined as a negative logarithm of the free H+ions, i.e.,
pH= -log[H+]
Similarly, pOH is defined as a negative logarithm of the free OH-ions, i.e.,
pOH= -log[OH-]
Neutral water is partially ionized into protons and hydroxide ions.
H2O(l) ⇌ H+(aq) + OH- (aq)
At 25oC, the concentration of both ions is = [H+] = [OH-] = 10-7
Relationship between pH, pOH and Kw
For the ionization equilibrium of water,
H2O(l) ⇌ H+(aq) + OH- (aq)
Using law of Mass action
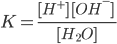
Kw= [H+] [OH-]= 10-14)
Kw is the ionization constant or ionic product of water..
Kw = [H+][OH-]
Applying (-log)both side
-logKw = -log[H+][OH-]
-log 10-14 = -log[H+]+(-log[OH-])
-(-14)log10 = pH + pOH
14 = pH + pOH
For pure water,
[H+] = [OH-]
-log[H+]=-log[OH-]
pH = pOH
14 = pH + pH
14 = 2pH
7 = pH = pOH
Hence, the pH of the neutral solution at 25oC is 7.
Note: Ionization changes with temperature and so will be the concentrations of hydrogen and hydroxide ions. Higher at higher temperatures and lower at lower temperatures. Accordingly, pH of the neutral water will be less than 7 at higher temperature and more than 7 at lower temperatures. Because of this Kw also will be changing with temperatures.
For acidic solution;
[H+]>[OH-]…………………………(1)
-log[H+]<-log[OH-]
pH<pOH
Multiply both sides by [H+] in (1)
[H+]2>[H+][OH-]
[H+]2>Kw
-lo[H+]2<-logKw
2pH < pKw
2pH < 14 (pKw = 14)
pH < 7
Hence, pH of the acidic solution at 25oC is less than 7 if we add an excess of [H+] ions in the solution
![Addition of excess [H+] ions](https://dcx0p3on5z8dw.cloudfront.net/Aakash/s3fs-public/inline-images/image128.gif?OU36FK8ZCWhzBLlMtTltuuj6Et2PIi4N)
Addition of excess [H+] ions
Similarly, For basic solution;
[H+] < [OH-]
-log[H+] >(-log[OH-]
pH > pOH
pH > 7
Hence, pH of the basic solution at 25oC is more than 7 if we add an excess of [OH-]ions in solution
![Addition of excess [OH-] ions](https://dcx0p3on5z8dw.cloudfront.net/Aakash/s3fs-public/inline-images/image126.gif?kHg0eJgIVJZVFukLbNvmyV9f_0FN0UB0)
Addition of excess [OH-] ions
The pH scale runs from 0 to 14, with 7 representing neutrality. Acidity is indicated by a pH less than 7, while a pH greater than 7 indicates a base.
Ionization of weak acid
Consider a weak acid HA (CH3COOH)
HA(aq) + H2O(l) ⇌ H3+O(aq) + A-(aq)
The equilibrium constant can be written as:




pH of weak base

Relationship between pKa and pKb
KaKb = Kw
-logKa + (-logKb) = -logKw
pKa + pKb = pKw = 14
Related Video:
Practice Problems
Q1. What will be the concentration of hydrogen ions in M in a solution of pOH = 12?
Answer. Using a relation; pKw = pH+pOH
14 = pH + 12
2 = pH
2 = -log[H+]
10-2M= [H+]
Q2. The given statement [H+]=-yx10z M then pH = x logy-z is correct or not?
Answer. [H+]=-yx10z M
-log[H+]=-log(-yx10z) M
pH = -(-logyx)+(-log10z)
pH = x logy+(-z log10)
pH = x log y-z
Hence, the given statement is correct.
Q3. If pKb = 4.74, calculate the value of pH of 0.01 M NH4OH solution.
Answer. ypOH=12pKb-logC
14-pH=124.74-log0.01
pH=14-124.74+2
pH=10.63
Q4. Which of the following is the strongest acid?
- Aniline (pKa=4.63)
- Benzoic acid (pKa=4.20)
- Fluoroacetic acid (pKa=2.63)
- Formic Acid (pKa=3.75)
Answer : (C)
Less is the value of pKa, greater is the strength of the acid. The least pKa is in option C, hence fluoro acetic acid is the strongest acid among all.
Q5. Calculate the pH of a solution having a concentration of [H+]=10-8 M
Answer. The concentration of [H+]=10-8 M is given. As the solution is diluted, the concentration of [H+] from water also plays an important role.
At 25oC, [H+] from water =10-7 M
Total concentration of [H+]=10-7 M +10-8 M = 1.110-7 M
pH=-log [H+]=-log(1.110-7 )=-log1.1-log10-7=-0.041+7=6.95
pH=6.95
Frequently Asked Questions
Question 1. What is the pH scale in chemistry?
Answer: The pH scale is normally between 0 and 14. Aqueous solutions at 250C with a pH of less than 7 are acidic and alkaline solutions are those pH greater than 7.
Question 2. Is it necessary to have a pH of 7 for a neutral solution at all temperatures?
Answer. If a solution is neutral, it does not necessarily mean it is pH 7. The equilibrium of the reaction of water dissociating into hydrogen and hydroxide ions is affected by temperature. Because the process is endothermic, heat is absorbed by the forward reaction. When the temperature of the water is raised, the forward reaction is favoured, and more hydrogen and hydroxide ions are produced, lowering the pH.
Question 3. What is the full form of pH?
Answer. pH stands for Hydrogen potential.
Question 4. Is it possible to have a pH less than 0 or more than 14?
Answer. For very concentrated strong acids, the pH value can be less than 0 and for very concentrated strong bases, it can be larger than 14. Concentrated hydrochloric acid, for example, has a pH of roughly -1, while sodium hydroxide solution has a pH of up to 15.
Related topics
|
Anionic Hydrolysis |
Hydrolysis of a salt weak acid and weak base |
|
Hydrolysis of salts- the salt of strong acid and weak base |
Acids, Bases and Salts |
|
Hydrolysis of salts - the salt of a weak acid and strong base |
Theory of Indicators |


