-
Call Now
1800-102-2727
Process of Recombinant DNA Technology: Downstream Processing, its Steps, Clinical Trials and Quality Control
Diabetics is a common disease which is found among adults. But, do you know how many such diabetic people are present just in India? Approximately 70 million people are diabetic in India!!
And India is not the only country on the map. This is really a big number. Right? So you can imagine the amount of insulin needed by the world.
Insulin is a protein which can be produced through the recombinant DNA technology in the laboratories. But do you think the product formed in the lab is enough for the people in the whole world? So what can be the solution?
Another major concern is that the cost per mg for the product would be very high if it is produced on a smaller scale. Hence, large scale production is a better alternative for producing any recombinant product. Let us see how large scale production of recombinant products is achieved.
Table of Contents:
Large Scale Production in rDNA Technology
If any protein-encoding gene is expressed in a host of a different species, it is called a Recombinant protein. Commercial production of insulin occurs by cloning the human insulin gene into the genome of a bacteria, Escherichia coli. Thus, insulin can be considered as a Recombinant protein. This is done because the bacteria multiplies rapidly, making millions of copies of the human insulin gene and simultaneously expressing ample quantities of protein in the culture media.
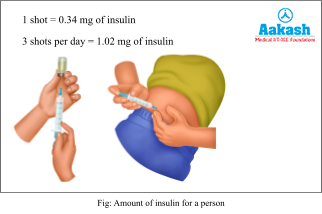
In a 1 litre flask, it yields only around 4000 mg of insulin. Each diabetic person injects 0.34 mg of insulin per meal. So in 3 meals he injects around 1 mg of insulin. The product formed in the lab is not enough for the treatment of 70 million people in India. Even if we run multiple labs and try to meet the needs, the process is very tedious. It would require trained manpower and continuous monitoring to maintain proper conditions. Hence the large-scale production of recombinant products is suggested.
Bioprocessing is the process of obtaining a foreign gene product from a living source such as a genetically engineered living organism, be it plant, animal or microbe. It is a large scale process. This can be divided into 2 major steps: Upstream processing and Downstream processing. These steps are not just specific to large scale productions.

If we are producing the product in the lab the same upstream and downstream steps will be followed but at a smaller scale. The cells possessing the cloned genes of interest may be cultured in the laboratory on a smaller scale. The desired proteins can be extracted from these cultures and then purified by using different separation techniques.
The major biosynthetic stage in the production of a recombinant product is the upstream processing. In this stage, the product is formed on a large scale with the help of huge bioreactors. But before reaching the market the product has to go through another process called downstream processing.
Let's discuss more about the downstream processing.
Downstream processing
Downstream processing involves recovery and purification of the formed product before it can be ready for marketing as a finished product. It helps in the removal of the undesirable elements from the reaction mixture. The undesirable elements are formed either by the cell or are present in the culture media from the reaction mixture.
Steps of Downstream processing
Four major steps are included in the process.

Separation of Cells
The cells need to be separated from the medium before extracting the product. It can be based on two factors. One is the density (heavier things settle and lighter medium remains on the top) or the size of the microbe. So the separation of the cells takes place through centrifugation or filtration. During centrifugation the particles are separated according to their density. Filtration can separate the cells from solution just like in a strainer.

Disintegration of Cells
The product that is formed at the end of the process will be processed based on what type of product it is. It can either be intracellular or extracellular. Intracellular products are those where the product is produced inside the organism and extracellular products are produced and moved or secreted outside the organism.

If the product is an intracellular one, we need the step of disintegration of cells to break or damage the cell and release the desired product. Extracellular products do not need this step.
Cells can be disrupted by many methods like mechanical (heat shock, ultrasonication), chemical treatments (alkalis, detergents) and enzyme action (lysosomes).

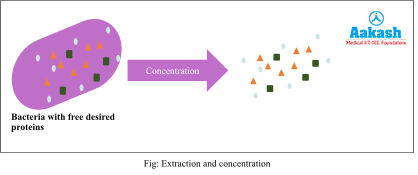
Extraction and Concentration
Even after releasing the product from the cell, the proteins remain mixed with cytoplasm which is mainly water. The product obtained is in very minor quantity in the solution which mainly consists of water. To remove the water from the product, this step of extraction and concentration is carried out. The procedure chosen for this will depend on the nature of the desired product, cost factor and the quality and quantity of product retained from the previous step.
Purification (Chromatography)
The cells produce a number of proteins other than the desired protein. So this step aims at obtaining the product in a highly purified state. Most common method used is chromatography.

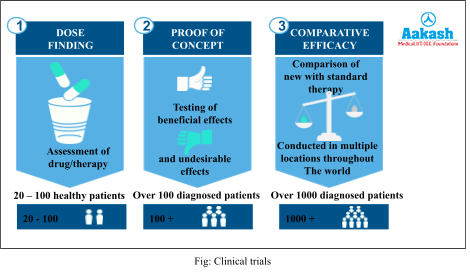
Clinical Trials
After downstream processing, the final step involves clinical trials and quality control. Suitable preservatives are added to the product and the formulation is made to undergo thorough clinical trials as in the case of drugs in order to assess its efficacy, side effects, dosage etc. Clinical trials are done only before the product is released in the market for the first time.
Quality Control
Quality control is the step to maintain the identity and purity of the formed product. Each product needs to undergo strict quality control testing. This step has to be done every time a product is made, to maintain the quality, quantity and validity of the product. The methods used for quality control testing vary from product to product.
Major Steps of Quality Control
- Determining the composition
- Checking adulteration
- Checking packaging
Practice Problems of Recombinant DNA Technology
Ques:- Downstream processing does not include which of the given processes?
1. Transformation of host cell
2. Separation of product from culture medium
3. Product purification
4. Sampling from bioreactors
A. I and II
B. II and III
C. I and IV
D. I, III and IV
Solution: Once the concentration of the product expressed by the growing host cells in the bioreactor reaches the optimal quantity, it needs to be extracted out for use. Then the downstream processing is done. In that the separation of the expressed gene product from the culture media and its purification are done. These are the components of downstream processing.
Transformation of the host cell with the desired foreign gene, culturing the cell in bioreactors and sampling the product from bioreactors are steps involved in upstream processing.
Hence the correct option is c.
Ques:- In case of drugs, the formulation with suitable preservatives has to undergo_________ before its availability for commercial use.
A. strict clinical trials
B. accurate separation techniques
C. heat shock treatment
D. prolonged restriction digestion
Solution: After the completion of downstream processing, the product is formulated with preservatives to increase the shelf life. If the desired product is a drug, this formulation with preservatives is then subjected to strict clinical trials in order to assess its efficacy, side effects, dosage etc. This is followed by quality control steps.
Hence the correct option is a.
Ques:- Which of the following methods does not help in removal of cellular debris from the culture media during downstream processing of the recombinant protein?
A. Filtration
B. Centrifugation
C. Electroporation
D. Sedimentation
Solution: The very first step of downstream processing involves separation of the insoluble cellular debris and other particulate matter from the culture medium containing the recombinant protein. This can be achieved using several techniques such as filtration, centrifugation, sedimentation, etc. Electroporation is a technique used for the introduction of foreign DNA into bacterial or plant cells by subjecting them to a very high voltage or electrical pulse.
Hence the correct option is c.
Ques:- Choose the correct option to indicate the techniques used for product purification during downstream processing of the recombinant gene product.
1. Chromatography
2. Heat shock
3. Centrifugation
A. I and II
B. II and III
C. I and III
D. Only I
Solution: The technique that is generally used for purification of the desired gene product is chromatography. Chromatography is a technique based on a principle in which molecules in a mixture can be separated on the basis of their solubility. So the technique used for product purification during downstream processing is chromatography.
Hence the correct option is d.
FAQs of Recombinant DNA Technology
Ques:- What is the significance of the large scale production of recombinant products?
Answer: The product formed in the lab is not enough for the purpose of the whole world. Even if we run multiple labs and try to meet the needs, the process is very tedious. It would require trained manpower and continuous monitoring to maintain proper conditions. Hence the large-scale production of recombinant products is required.
Ques:- What are the two steps used in large scale production of recombinant products?
Answer: The process of large scale production of recombinant products can be divided into 2 major steps. They are Upstream processing and Downstream processing. These steps are not just specific to large-scale productions. The major biosynthetic stage in the production of a recombinant product is the upstream processing. But before reaching the market the product will go through another process for assuring the quality, quantity and validity of the product. It is called downstream processing.
Ques:- What are the steps of downstream processing?
Answer: Downstream processing involves recovery and purification of the formed product before it can be ready for marketing as a finished product. There are mainly four steps in the downstream processing. They are:
- Separation of Cells
- Disintegration of cells
- Extraction and Concentration
- Purification
Ques:-What is meant by quality control in downstream processing?
Answer: Quality control is the step to maintain the identity and purity of the formed product. Each product needs to undergo strict quality control testing. This step has to be done every time a product is made, to maintain the quality, quantity and validity of the product. The methods used for quality control testing vary from product to product. Major steps of quality control are Determining the composition, Checking adulteration and Checking packaging.


