-
Call Now
1800-102-2727
Fehling’s Solution, Practice Problems and FAQs
We always feel tired after some heavy work, sports, or even after walking for a long distance. Do you know the reason for this? The major reason for the tiredness of the body is the lack of energy. So where do we get energy? We get energy through food and the excess energy we need is utilised from the stored energy molecules. This is the same in the case of both plants and animals. Now think about the stored food materials in plants and animals. Plants store food in the form of starch and animals store food in the form of glycogen. What exactly are these starch and glycogen?

Fig: People running in a race
Starch is a carbohydrate and glycogen is a stored form of glucose, which is a carbohydrate and called animal starch. Hence we can say that carbohydrates are the basic components of food and they are the principal source of energy for all the living organisms. Most of the food we eat has carbohydrates.

Fig: Foods with carbohydrates
Carbohydrates can be simple like sugars and can be complex like starches and fibres. This is the one reason we take instant glucose when we feel tired. We will know that our sugar level is low when our body is weak. Right? What happens if the sugar level is more in your body? This may lead to severe health issues like hyperglycemia, diabetics etc.
So we have to find out the presence of sugar in our body. How do we do that? The one process is to test the sugar level in the blood and urine, because blood carries sugar to all parts of the body and the excess sugar is gotten rid of by the kidneys through urine. The sugar in our blood is glucose, which is a monosaccharide. This can be easily detected by using a chemical reagent, called Fehling's reagent. In this article we are going to discuss more about the Fehling’s solution and the test done by using this.
Table of contents
- Fehling’s solution
- Application of Fehling’s test
- Limitations of Fehling’s test
- Practice Problems
- FAQs
Fehling’s solution
The deep blue alkaline solution which helps to identify the presence of aldehydes that contain any aldehyde functional group (-CHO) is called the Fehling’s solution. The German chemist Hermann Von Fehling developed this test in 1849. Hence Fehling's test is one of the tests used to identify the reducing and nonreducing sugar supplementary to Tollen’s reagent test (also distinguishes between aldehydes and ketones). This solution can be also used to differentiate between the ketone functional groups (-C=O) and water soluble carbohydrates.
Types of sugars
According to the ability of sugars to reduce other substances, they can be classified in to two as follows:
- Reducing sugars
- Non reducing sugars
Reducing sugars
The sugars which have an anomeric carbon with OH group attached with it and can reduce other compounds are called reducing sugars. All the monosaccharides such as glucose are reducing sugars and all the disaccharides except sucrose are reducing sugars.
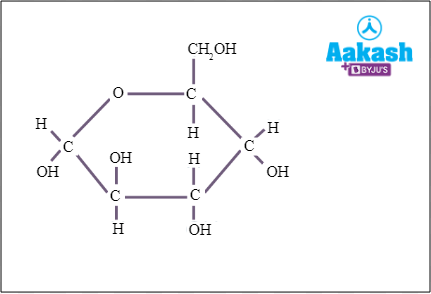
Fig: Glucose
Other examples of reducing sugars are galactose, glyceraldehyde, fructose, ribose, and xylose.
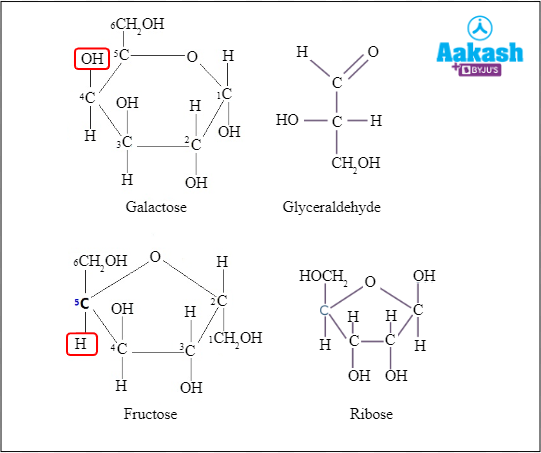
Fig: Reducing sugars
Non reducing sugars
Those sugars without an OH group in the anomeric carbon and not oxidised by a weak oxidising agent are called non reducing sugars. They do not have a free aldehyde or ketone group. Sucrose is a disaccharide, but a non reducing sugar.
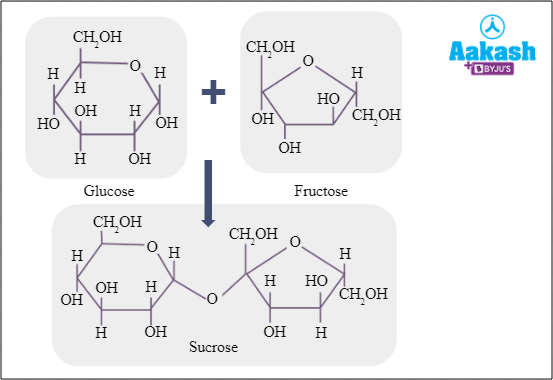
Fig: Sucrose
Other examples of non reducing sugars are stachyose, verbascose, trehalose and raffinose. All polysaccharides are non reducing sugars or carbohydrates.

Fig: Polysaccharides
By using Fehling's solution, the Fehlin’s test is done to find the presence of an aldehyde group or whether it is a reducing or non reducing sugar. In order to carry out this, the substance to be tested has to be heated with Fehling's solution. If an aldehyde group is present, a brick red precipitate is formed in the sample.
Objectives of Fehling’s Test
The objectives of the Fehling’s test can be explained as follows:
- To find out the presence of carbohydrates in a solution.
- To differentiate between reducing and non-reducing sugars.
Preparation of Fehling's solution
In order to get accurate results, Fehling's solution should be freshly prepared. Hence the solution is prepared just before conducting the experiment. Fehling’s solution is made by mixing two previously made solutions in equal volume. The solutions are called Fehling’s A and Fehling’s B solutions. Both the solutions were prepared and stored separately in bottles with rubber stoppers.
Composition of Fehling’s A solution
It is a deep blue aqueous solution of copper sulphate (CuSO4). This solution is prepared by dissolving 70 gms of cupric sulphate pentahydrate in 1L of water. The deep blue colour of the Fehling’s solution is due to the presence of cupric ions (Cu2+) in it.

Fig: Fehling’s A solution
Composition of Fehling’s B solution
It is a colourless solution of potassium sodium tartrate tetrahydrate (KNaC4H4O6·4H2O) and sodium hydroxide (NaOH). Potassium sodium tartrate tetrahydrate is also called Rochelle salt and it is a chelating agent too. A chelating agent is a chemical compound which can bind tightly to metal ions. This solution is prepared by dissolving 350 gms of Rochelle salt in a solution of 100 gms of sodium hydroxide per litre of water.
Equal volumes of Fehling’s A and Fehling’s B solutions are mixed right before the experiment, when there is a requirement.
Principle of Fehling's test
The key principle behind the working of Fehling’s test is the reduction of cupric ions. When there is aldehyde in the sample solution, the copper ions in the Fehling’s solution will oxidise the aldehydes and an acidic compound (aldonic acid) is formed.
RCHO + 2 Cu2+ + 5 OH− → RCOO− + Cu2O + 3 H2O
(Sugar) (Cupric ion) (Copper oxide)
The reduction of cupric ions results in the formation of copper oxide and it appears as a precipitate. The colour of precipitate formed as a result of the reduction of cupric ions of Fehling’s solution to cuprous ions is brick red. This represents that the sample contains reducing sugars.
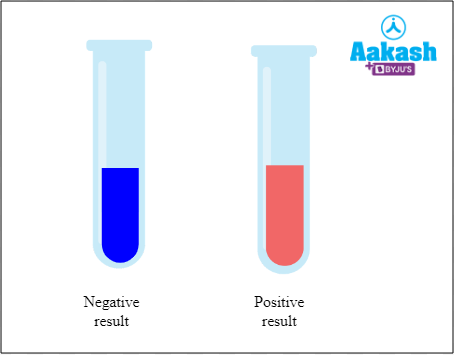
Fig: Result of Fehling’s test
Procedure of Fehling’s test
The heating of solution (aldehyde) with Fehling’s reagent is the basic procedure in the Fehling’s test. But the aromatic aldehydes will not show any reaction to the Fehling’s test and so do the ketones. Hence it is easy to distinguish between the ketones and aldehydes.
The steps followed to conduct the Fehling’s test are the following:
- Take a dry test tube and add 1 to 2 ml of sample into it.
- Take another test tube and add distilled water to it. It is used as control.
- Add 1 ml of Fehling’s solutions A (CuSO4 solution) and B [Rochelle salt (Sodium potassium tartrate) + Sodium hydroxide] one by one to each test tube.
- Heat the test tubes by keeping it inside the boiling water bath.
- Observe the signs of formation of red precipitate in each test tube.
If there is any formation of reddish brown precipitate then the result is positive and there is presence of aldehyde. If there is no indication of such change then the result is negative and the aldehyde is not present in the test tube.
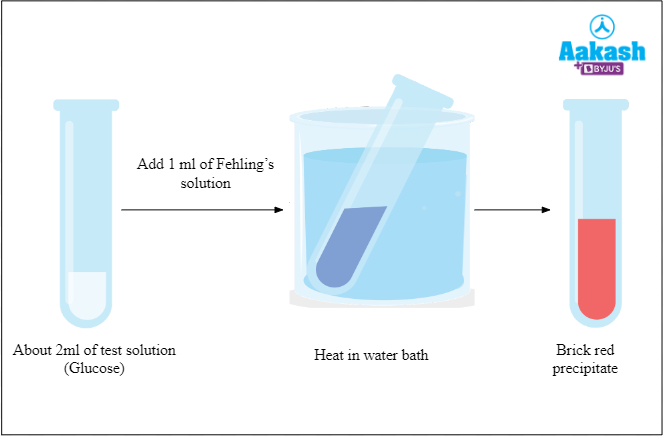
Fig: Fehling’s test
Precautions to be taken
The following precautions should be taken while performing the Fehling’s test:
- Wear protective gloves and goggles must be used while preparing the Fehling’s solution and performing the Fehling’s test. The reason for this is that the Fehling’s reagent is corrosive in nature.

Fig: Safety accessories in lab - gloves and goggles
- Make sure that the sample is not getting overheated or underheated.
- Only 2 to 3 minutes of heating is needed. We may get the wrong colour indications, if the sample is overheated and this will lead to the failure of the test.
Reactions of Fehling’s test
The reaction between copper ions in Fehling's solution and aldehyde of the sample can be represented as follows:
RCHO + 2 Cu2+ + 5 OH− → RCOO− + Cu2O + 3 H2O
In this process, the copper (II) ions of the complex are reduced to cuprous (I) oxide (Cu2O) ions, which is a red coloured precipitate.
When tartrate is added then the reaction will be as follows:
RCHO + 2 Cu(C4H4O6)22− + 5 OH− → RCOO− + Cu2O + 4 C4H4O62− + 3 H2O
The tartrate ions prevent the formation of insoluble Cu(OH)2 from the reaction of copper sulphate and sodium hydroxide present in the solution by forming a bis tartaro cuprate (II) complex. Cupric ions are released slowly for reduction by this complex. This will prevent the formation of black cupric oxide. This black precipitate of cupric oxide can also be formed if the Fehling’s solution is heated in the absence of reducing sugars.
Copper II ions (Cu2+) are reduced to copper I oxide when the redox reaction is completed. Copper I oxide forms the red precipitate and it is insoluble in water. The sodium salt of the acid is left behind in the solution at the end.
Applications of Fehling’s solution
One of the most widely used reagents in the chemistry labs to identify the reducing and non reducing sugars is the Fehling’s solution. Some of the major applications and uses of Fehling's solution are as follows:
- To differentiate between the ketone and aldehyde functional groups, because aldehydes show positive results in the Fehling’s test and ketones do not. An exception to this is the α-hydroxy ketones as they develop colour upon the addition of Fehling’s reagent.
- Since both aldose and ketose monosaccharides can develop red colour in the presence of Fehling’s solution, this can be used for testing the presence of monosaccharides and many other sugars.
- Diagnosis of diabetes can be done by using Fehling’s solution, because the elevated glucose level will be there in the blood and urine.
- Starch can be broken down into maltodextrin and glucose syrup by using Fehling’s solution. Maltodextrin is further used as a food preservative.
Limitations of Fehling’s test
There are some limitations for Fehling’s test and these are as follows:
- Fehling’s can not be used to detect aromatic aldehydes.
- Fehling’s reactions can only take place in an alkaline environment. In an acidic environment the copper (II) ions would be stabilised and not easily oxidised. This will give a negative result for the test.
Practice Problems
1. Which of the statements is wrong about Fehling's solution?
- Fehling’s solution helps to identify the presence of aldehydes that contain any aldehyde functional group (-CHO)
- Fehling's solution is used to identify the reducing and nonreducing sugar
- This solution can not be used to differentiate between the ketone functional groups (-C=O) and water soluble carbohydrates
- Generally if a ketone group is present, a brick red precipitate is formed in the sample
Solution: The deep blue alkaline solution which helps to identify the presence of aldehydes that contain any aldehyde functional group (-CHO) is called the Fehling’s solution. The German chemist Hermann Von Fehling developed this test in 1849. Hence Fehling's test is one of the tests used to identify the reducing and nonreducing sugar supplementary to Tollen’s reagent test (also distinguishes between aldehydes and ketones). This solution can be also used to differentiate between the ketone functional groups (-C=O) and water soluble carbohydrates. By using Fehling's solution, the Fehlin’s test is done to find the presence of an aldehyde group or whether it is a reducing or non reducing sugar. In order to carry out this, the substance to be tested has to be heated with Fehling's solution. If an aldehyde group is present, a brick red precipitate is formed in the sample. Generally for a sugar having a keto group, the Fehling’s test shows a negative result. Hence the correct option is d.
2. A student was conducting Fehling’s test, in order to distinguish between the reducing and non reducing sugar. But he did not get any positive result. Which of the following reasons can be true?
I) Fehling’s solution was not freshly prepared.
II) Equal volumes of Fehling’s A and Fehling’s B solutions were mixed.
III) Fehling’s A solution was prepared by dissolving 70 gms of cupric sulphate pentahydrate in 1L of water.
IV) Fehling’s B solution was made by using 100 gms of sodium hydroxide per litre of water.
- I, II, III and IV
- I and III
- I and IV
- Only I
Solution: By using Fehling's solution, the Fehlin’s test is done to find the presence of an aldehyde group or whether it is a reducing or non reducing sugar. In order to get accurate results the Fehling’s solution should be freshly prepared. Hence the solution is prepared just before conducting the experiment. Fehling’s solution is made by mixing two previously made solutions in equal volume. The solutions are called Fehling’s A and Fehling’s B solutions. Both the solutions were prepared and stored separately in bottles with rubber stoppers. Fehling’s A solution is a deep blue aqueous solution of copper sulphate (CuSO4). This solution is prepared by dissolving 70 gms of cupric sulphate pentahydrate in 1L of water. The deep blue colour of the Fehling’s solution is due to the presence of cupric ions (Cu2+) in it. Fehling’s B solution is a colourless solution of potassium sodium tartrate tetrahydrate (KNaC4H4O6·4H2O) and sodium hydroxide (NaOH). Potassium sodium tartrate tetrahydrate is also called Rochelle salt and it is a chelating agent too. A chelating agent is a chemical compound which can bind tightly to metal ions. This solution is prepared by dissolving 350 gms of Rochelle salt in a solution of 100 gms of sodium hydroxide per litre of water. Equal volumes of Fehling’s A and Fehling’s B solutions are mixed right before the experiment, when there is a requirement. Hence the correct option is c.
3. Find out the correct order of procedure for the Fehling’s test.
i) Add 1 ml of Fehling’s solutions A (CuSO4 solution) and B [Rochelle salt (Sodium potassium tartrate) + Sodium hydroxide] one by one to each test tube.
ii) Take another test tube and add distilled water to it and use it as control.
iii) Observe the signs of formation of red precipitate in each test tube.
iv) Test tubes are kept in a boiling water bath.
v) Take a dry test tube and add 1 to 2 ml of sample into it.
- I - ii - iii - iv - v
- V - ii - i - iv - iii
- I - iii - ii - iv - v
- V - ii - i - iii - iv
Solution: The heating of solution (aldehyde) with Fehling’s reagent is the basic procedure. But the aromatic aldehydes will not show any reaction to the Fehling’s test and so do the ketones. Hence it is easy to distinguish between the ketones and aldehydes. The steps followed to conduct the Fehling’s test are the following:
- Take a dry test tube and add 1 to 2 ml of sample into it.
- Take another test tube and add distilled water to it. It is used as control.
- Add 1 ml of Fehling’s solutions A (CuSO4 solution) and B [Rochelle salt (Sodium potassium tartrate) + Sodium hydroxide] one by one to each test tube.
- Test tubes are kept in a boiling water bath.
- Observe the signs of formation of red precipitate in each test tube.
If there is any formation of reddish brown precipitate then the result is positive and there is presence of aldehyde. If there is no indication of such change then the result is negative and the aldehyde is not present in the test tube.
4. Assertion: Diagnosis of diabetes can be done by using Fehling’s solution.
Reason: There will be elevated glucose levels in the blood and urine of a diabetic patient.
Which of the following statements are correct about the assertion and the reason given above.
- Both the assertion and the reason are true, and the reason is the correct explanation of the assertion
- Both the assertion and the reason are true, but the reason is not the correct explanation of the assertion
- Assertion is true, but the reason is false
- Both the assertion and the reason are false
Solution: The deep blue alkaline solution which helps to identify the presence of aldehydes that contain any aldehyde functional group (-CHO) is called the Fehling’s solution. Hence Fehling's test is one of the tests used to identify the reducing and nonreducing sugar supplementary to Tollen’s reagent test (also distinguishes between aldehydes and ketones). This solution can be also used to differentiate between the ketone functional groups (-C=O) and water soluble carbohydrates. The sugars which have an anomeric carbon with OH group attached with it and can reduce other compounds are called reducing sugars. All the monosaccharides such as glucose are reducing sugars and all the disaccharides except sucrose are reducing sugars. There are many applications for the Fehling’s test and one of the major applications is the detection of diabetics. Diagnosis of diabetes can be done by using Fehling’s solution, because the elevated glucose level will be there in the blood and urine. So while testing the blood and urine of a diabetic patient, if a positive result is shown in Fehling’s test, then he is having high sugar content in his body. So here both the assertion and the reason are true and reason is the correct explanation of the assertion. Hence the correct option is a.
FAQs
1. Can fructose show positive results in Fehling's test?
Answer: Yes, Fructose reduces Fehling’s solution and shows a positive result in the Fehling’s test. Even though Fehling’s test is used for the identification of sugars with aldehyde as a functional group, fructose with ketone functional group shows the red precipitate in Fehling’s test. Fructose gives rise to an equilibrium mixture of glucose, fructose and mannose while warming with dilute alkali. The isomerisation of fructose to glucose and mannose is the reason why the fructose shows positive results. This isomerisation of fructose is called Lobry de Bruyn and Elkenstein rearrangement.
2. Is there any aldehyde which can not show a positive Fehling’s test?
Answer: Those aldehydes which lack alpha hydrogens will not show a positive result for Fehling’s test. They can not form an enolate under usual conditions. An example for this is benzaldehyde or pivalaldehyde (2, 2-dimethylpropanal).
3. What is Benedict's reagent?
Answer: The chemical reagent used to detect the presence of reducing sugars which is a complex mixture of sodium carbonate, sodium citrate and copper (II) sulphate pentahydrate is called a Benedict's reagent. If a solution is showing positive results for Benedict's test, then it will show a colour change from clear blue to brick red with a precipitate. This reagent was named after Stanley Rossiter Benedict who is an American chemist. Benedict's test also detects the presence of aldehydes, alpha-hydroxy-ketones and hemiacetals.
4. What is dextrose equivalent?
Answer: The measure of the amount of reducing sugars present in a sugar product is termed as dextrose equivalent or DE. It is a quantity stating the total value of a reducer of starch or a starch modified product in the percent unit. It can also give an indication of the average degree of polymerisation or DP for starch sugars.


