-
Call Now
1800-102-2727
Zeroth Law of Thermodynamics - Definition, Cases, Equations, Practice problems, FAQs
Anish and Nikita always score first and second in the class, while Shriya scores third in the class. Shriya being Nikita’s good friend, agrees with her in most cases. While solving a physics problem, Anish and Nikita get the correct answer as option (b). So it would be correct to say that they two are in good agreement with each other.On the other hand, Shriya, who solved the same problem also gets the answer as option (b). So would it be appropriate to say that Anish and Shriya are in agreement with each other? The answer is yes – since Nikita and Shriya share a good relation, she has no choice but to agree with Anish. Let’s call this harmony “equilibrium”. In physics, thermal equilibrium refers to substances being maintained at the same temperature. The Zeroth Law of thermodynamics aims to explain this thermal equilibrium in detail. If a coffee mug at 600C and a water mug at 600C are in thermal equilibrium with each other, then they both are in thermal equilibrium with a bowl containing chicken soup at 600C. In this article, we will explore the zeroth law of thermodynamics in detail.
Table of contents
- Heat and temperature
- Thermometers
- Thermal equilibrium and steady state
- Definition of zeroth law of thermodynamics
- Practice problems
- FAQs
Heat and temperature
Heat is the cause while temperature is the effect. Temperature helps to quantify heat. Higher the temperature, hotter the body. Lower the temperature, colder the body. Temperature is measured in different scales–Kelvin scale, Celsius and Fahrenheit Scale. Celsius ( 0C),Fahrenheit, ( 0F)and Kelvin ( K ) are the different units to measure temperature.
Thermometers
Thermometers are used to measure and record temperature. They work on the principle of thermometric property– a physical quantity which can change with temperature.
For instance, pressure, volume, length and resistance exhibit thermometric properties– they vary with temperature. Let X denote the physical quantity which has a relation with temperature. Let X0, X100 and Xt be its values at 00 C, 1000 C and t0 C respectively. Then the temperature t can be expressed as,
Temperature, t=
Types of thermometers
1)Mercury thermometer
Some amount of mercury is filled in these thermometers. When a patient running a fever is checked with a thermometer, the length of the mercury column fluctuates–and helps record the temperature. Mercury is used in thermometers because it has a high coefficient of expansion–meaning that it is sensitive to changes in temperature –hence it would give more accurate temperature readings when used to record a patient's temperature. Let L0 indicate the length of the mercury at 00 C and L100 be its length at 1000 C. Let Ltindicate the length at t0 C. Then,
2)Constant volume gas thermometer
This type of thermometer makes use of constant volume of the gas being filled inside the thermometer. But the pressure of the gas column varies since the temperature varies. If p0, p100 and pt indicate the pressures of the gas at 00C, 1000 Cand the unknown temperature t0C.Then,
t=
In the case of constant pressure gas thermometers, the volume would vary. Therefore,
t=
3)Platinum resistance thermometer
Resistors are electrical components that resist the flow of current through them. Platinum resistance thermometers make use of resistors. Let R0, Rt and R100 indicate the resistance value( in ohms) at 00C, t0C and 1000C. Then,
t=
The temperature coefficient of resistance refers to the increase in resistance for every 10C rise in temperature of the resistor.
4)Pyrometers
Pyrometers record the temperatures of the body by detecting the intensity of signals coming from it. Stefan’s law states that the amount of thermal energy emitted per unit area per unit time is directly proportional to the fourth power of its absolute temperature. Temperatures in the range of 8000 C to 60000 C can be recorded using pyrometers.
Thermal equilibrium and steady state
Thermal equilibrium refers to two bodies being maintained at the same temperature. Consider a rod whose length is dx, and the temperature difference between the ends is dT. The rate of heat flow through can be written as,
Consider two rods having thermal conductivities K1 and K2 . The length of each rod is L and area of cross section area A. They are joined end to end as shown in the figure. The left end of the rod is maintained at temperature T1. The right end of the second rod is maintained at temperature T2(T1>T2). After reaching steady state, all points of the system are at the same the common temperature T.
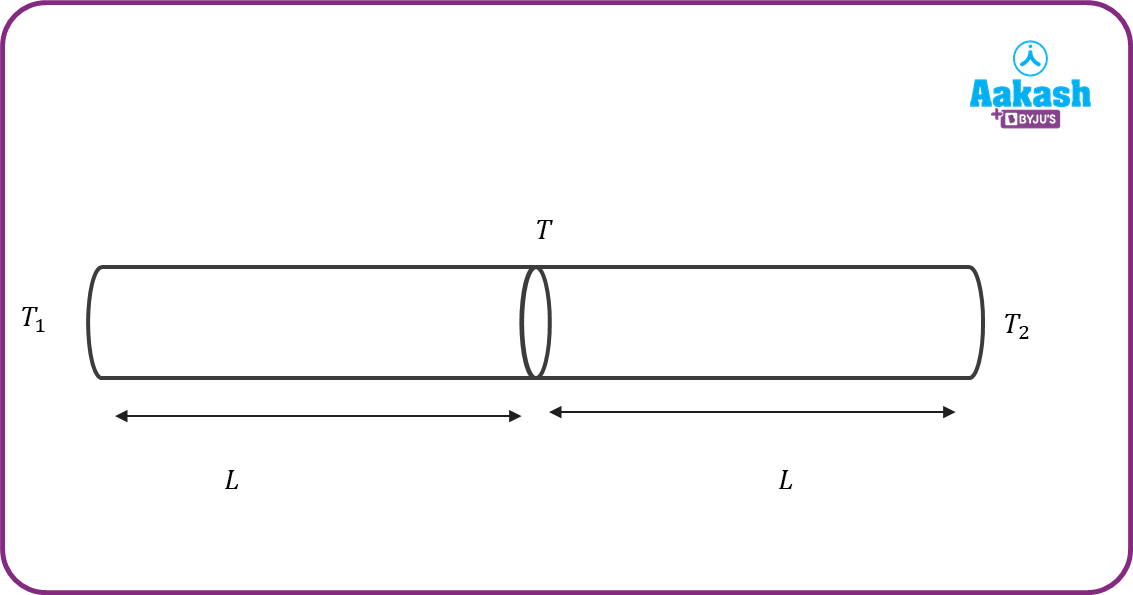
Heat flux through rod 1= heat flux through rod 2
From the above equation, the common temperature Tcan be calculated.
The unit of thermal conductivity is and its dimensional formula is .
Definition of zeroth law of thermodynamics
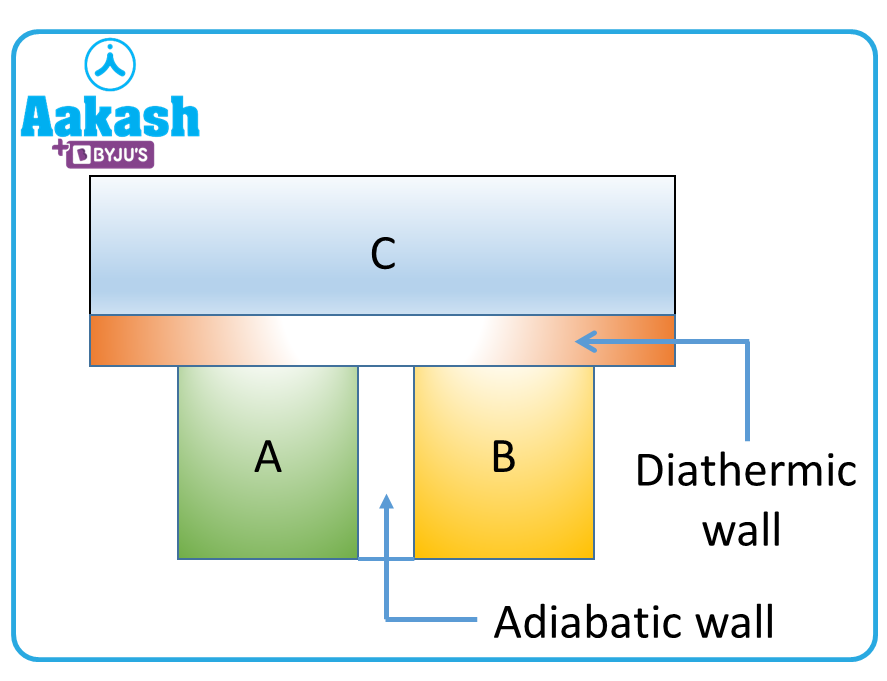
Let us consider two systems A and B which are separately in thermal equilibrium with C.
Then their temperatures can be related as,
and
This implies that systems A and B are in thermal equilibrium with each other.
Systems A and B are in contact with the third system C through a diathermal wall–which can facilitate heat transfer but not mass transfer between the systems. A and B are separated by an adiabatic wall. The variables associated with A and B would vary until they are in equilibrium with C.
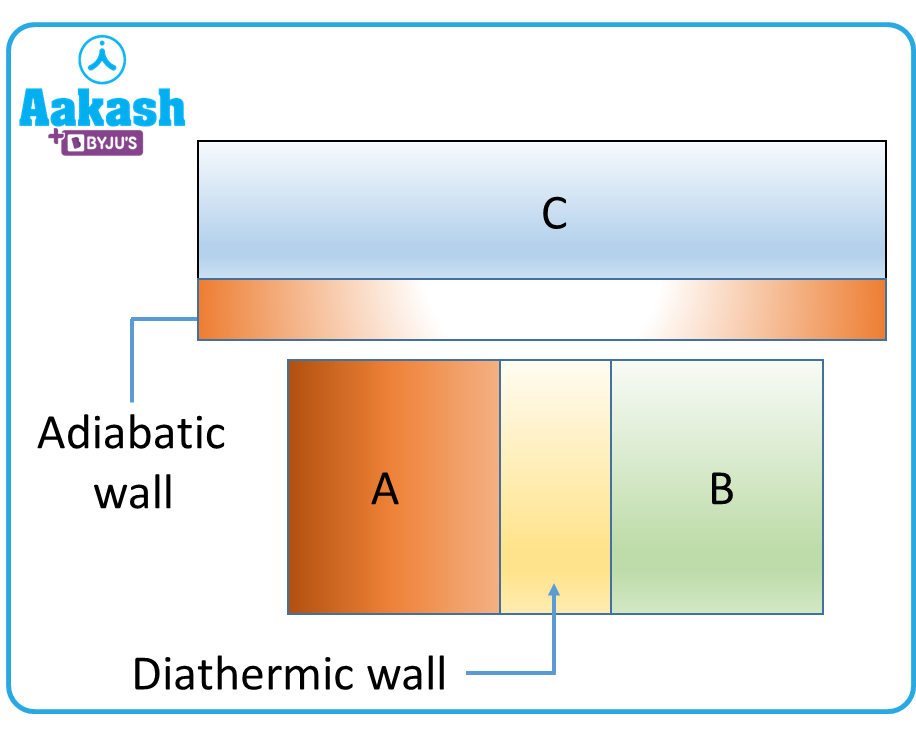
Now if the adiabatic wall between A and B is replaced by a diathermal wall and an adiabatic wall separates A and C. Now A and B are found to be in thermal equilibrium with each other.
Practice problems
Q1.A metal rod having length 2 m has cross sectional areas 2 A and A as shown in the figure. The ends of the rods are maintained at temperatures 1000 C and 700 C. The temperature at the junction C would be
(a)800 C (b)850 C (c)900 C (d)950 C

Answer. c
Here, ,
Let K1=K2=K indicate the thermal conductivities of the rod.
Let T indicate the temperature of the middle point. Applying the concept of steady state,
Q2.A silver rod with a circular cross section has a radius of 1 cm. One end of the rod is kept at 1000 C and the other end is kept at 00 C. Calculate the thermal resistance and the thermal current through the rod, if the length of the rod is 2 m. Given, the thermal conductivity of silver is .
Answer.
Given,
Length of the rod, l= 2 m.
Radius of the rod, r= 1 cm=10-2 m.
Thermal conductivity of the rod,
Area of the rod,
Thermal resistance,
Temperature difference,
Thermal current, , i=6.3 W
Q3.The temperature of a body P is found to be 100C and it is in thermal equilibrium with another body Q. The temperature of a body R is also 100C. P and Q and are separately in equilibrium with R. Calculate the temperature of the body Q.
Answer.
Given,
Temperature of the body , Temperature of the body
Temperature of the body
According to the Zeroth law of thermodynamics, ;
Then
Hence, temperature of the body Q=100 C
Q4.Two wires, A and B have identical lengths. The radius of the wire A is twice that of B, ie . The temperature difference between the two ends of the rod are the same, and the wires conduct heat at the same rate. The relation between their thermal conductivities are
(a) (b)(c) (d)
Answer. d
Given, radius of wire B ( twice that of wire B).
The rate of heat transfer,
Equating the rate of heat transfer through the wires at steady state,
Ratio of the thermal conductivities,
FAQs
Q1.What is the best example of the zeroth law?
Answer. A best example of the zeroth law of thermodynamics would be a thermometer and a thermostat. The thermostat of an AC would maintain the room temperature at 700 Fahrenheit(say). If the thermometer registers the temperature of a person also as 700 Fahrenheit, the thermometer and the AC are said to be in thermal equilibrium,meaning that the person and the AC are in thermal equilibrium– thereby satisfying the zeroth law of thermodynamics.
Q2.How does zeroth law explain the concept of temperature?
Answer. Zeroth law talks about temperature to quantify the degree of hotness or coldness of a body. Additionally, it also talks about the concept of thermal equilibrium. For instance, a heating pad at 800C and a boiling chicken soup at 800C are said to be in thermal equilibrium–since they have the same temperature.
Q3.Give a few applications of the zeroth law of thermodynamics.
Answer.The zeroth law finds applications in: constant gas volume thermometer, constant gas pressure thermometer, electrical resistance thermometer and thermocouple. Since these instruments revolve around the concept of temperature, the zeroth law helps to explain their working principle.
Q4.What is the necessary condition for thermal equilibrium?
Answer.When two bodies are in thermal contact with each other, heat transfer between them is possible. The rate of heat transferred per unit time is proportional to the coefficient of thermal conductivity, the area of cross section of the conductor and inversely proportional to its length. If heat transfer between the systems is restricted, then they cannot reach thermal equilibrium.


