-
Call Now
1800-102-2727
Paramagnetic materials, properties, behaviour in an external field, Curie’s law, practice problem, FAQs
Have you ever tried to stick a magnet on the aluminium utensils or foil? If you had you’d know that they are not as strongly attracted to the magnets as iron does. But what made them to behave differently after all they both are metals and share many common properties. To know the reason you must know about the magnetic properties it carries.
Paramagnetic materials
The substances which, when placed in an external magnetic field, acquire a feeble magnetization in the same sense as the applied field are known as paramagnetic materials.
Properties of paramagnetic substances
- Magnetic Susceptibility: 𝜒 > 0 (Small and positive)
- Relative permeability:𝜇 : Slightly more than one
In the absence of an external field, these dipoles are randomly distributed in volume giving a net magnetic moment zero.
For eg. O2 (at STP),𝑁𝑎,𝐹𝑒𝑂,𝐴𝑙
Explanation for behaviour of paramagnetic substance
In paramagnetic substances, each atom has a finite dipole moment and thus behaves like a small magnetic dipole whereas the net dipole moment becomes zero due to the thermal agitation and because of that the atomic dipole achieves random orientation. These atomic dipoles get aligned and result in a net dipole moment when the substance is placed in an external magnetic field.
Magnetic moment for each atom: mi≠0. This means each atom should have unpaired electrons.
Magnetic moment of the whole material: MT=Σmi=0
Behaviour in an external magnetic field
- The dipoles are aligned in the direction of the applied external magnetic field.
- This causes weak attraction to the external magnetic field and this phenomenon is called Paramagnetism.
- The alignment is partial.
- The Paramagnetic property of a substance depends on temperature.
- Induced field I is small and along the external field H.
- For paramagnetic substances, the induced internal magnetic field and the external magnetic field are along the same direction. Hence, the net magnetic field inside the material slightly increases. As a narrow spaced magnetic field denotes higher intensity, for the case of paramagnetic substances we can say that the magnetic field lines become denser inside paramagnetic materials when placed in an external magnetic field.
- A paramagnetic rod sets itself parallel to the field because the constituent atoms of the rod behave as magnetic dipoles and try to align themselves along the direction of the external field.
Curie’s law
The law states that for a paramagnetic material, magnetisation is inversely proportional to the absolute temperature.

Where x = susceptibility of the material
T = Absolute temperature
C = Curie constant
Beyond saturation value of magnetization ( ), at which all dipoles are perfectly aligned with the field, Curie’s law is no longer valid.
), at which all dipoles are perfectly aligned with the field, Curie’s law is no longer valid.
Practice problems
Q.The magnetic susceptibility of a paramagnetic material is 0.0075 at -73oC. Find susceptibility at
-173oC.
[Assume the states are far away from saturation]
A.
For paramagnetic material at states far away from saturation,
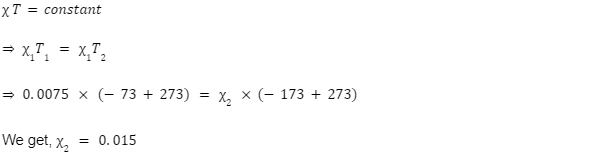
Q. The x versus graph for an alloy of paramagnetic nature is shown in the figure. Find the Curie constant for this alloy.
[Assume the states are far away from saturation]
A. We know that, for paramagnetic material, at states far away from saturation,
Comparing the above expression with the equation of a straight line, passing through origin, i.e. y, we can say that, the slope of x versus curve is equal to the Curie constant.
Therefore, slope,
Q.For a magnetising field of intensity , aluminium at acquires intensity of magnetisation of . What will be the intensity of magnetisation if the temperature of the metal is raised to 320 K ?
A.
Given:
At, 
At;
;
Using the relation, , we get
Now, according to the Curie's law,
Now, the intensity of magnetisation,
Since, h is independent of the temperature, i.e.,
∴
Q.A sample of paramagnetic salt contains atomic dipoles each of dipole moment . The sample is placed under a homogeneous magnetic field of , and cooled to a temperature of . The degree of magnetic saturation achieved is equal to 15%. In the magnetic field of What will be the total dipole moment of the sample at a temperature of ?
(Assume Curie's law is valid)
A.
Given:
Number of atomic dipoles, n =
Dipole moment of each atomic dipole,
When the magnetic field,
Temperature,
Total dipole moment of the atomic dipole,
Magnetic saturation is achieved at 15%
Hence, an effective dipole moment,
When the magnetic field,
Temperature,
Its total dipole moment
Using Curie's law, for a paramagnetic material,

∴
FAQs
Q. If the magnetic dipole moment and susceptibility of an atom of diamagnetic material and paramagnetic materials are denoted by , and , respectively, then
- (d)
Diamagnetic materials have no net magnetic moment without the presence of an external applied magnetic field  . Diamagnetic materials have a small negative magnetic susceptibility xd
. Diamagnetic materials have a small negative magnetic susceptibility xd
Thus,
Materials having unpaired electrons, whose spins do not cancel each other, are called paramagnetic.
The susceptibility of paramagnetic material is small and positive.
Thus,
Q.A spherical cavity with a paramagnetic substance is present inside a diamagnetic sphere at its centre and is placed in a uniform magnetic field B. What can we deduce about the field inside the paramagnetic substance?
- more than |B| and parallel to B.
- less than |B| and antiparallel to B.
- less than |B| and parallel to B.
- Zero
A. (d)
Magnetic field lines do not pass through the diamagnetic sphere, as shown
Therefore, the magnetic field will be absent inside the cavity of the sphere. Therefore, the paramagnetic substance will not experience any force.
Q.The liquid in the watch glass in the following figure is -
- Paramagnetic
- Diamagnetic
- Non-magnetic
- Cannot predict
A. (a)
The liquid shown in the figure is paramagnetic in nature.
Because when a paramagnetic liquid is placed in a non-uniform magnetic field, they have a tendency to move from the weaker part of the magnetic field to the stronger part of the field.
The magnetic field is stronger near the poles of the two magnets, and therefore most of the liquid accumulates in that region
Q.A block of certain material is placed in a uniform magnetic field. The image below shows distribution of magnetic field lines around the block. If the temperature of the block is increased, what will happen to the number of magnetic field lines passing through the block?
A.
In an external magnetic field, some of the magnetic field lines are passing through the block, which means that the block is made of paramagnetic material. As the temperature increases, the susceptibility of paramagnetic material decreases.
Hence, the magnetization of the block will decrease with increase in temperature. Therefore, the number of magnetic field lines passing through the block will continue decreasing, as the temperature increases.


