-
Call Now
1800-102-2727
Heat - specific heat, modes of heat transfer, Practice problems, FAQs
You might have observed in summers that when an ice cube is placed in an open, it starts melting. But from where and how did it get the energy to start the melting process? The energy involved in this process is heat. Heat is involved in many processes in our everyday life and thus it becomes important to know about this form of energy flow. Let’s read more about heat in this article.
Table of contents
- Heat
- Specific heat
- Modes of Heat Transfer
- Heat conversion to work
- Practice problems
- FAQs
Heat
Heat is a form of energy in transit that flows due to the virtue of temperature difference. Heat is a mode of transferring energy to a system just as work.

Heat is a low grade energy as compared to work which is high grade energy. This means that complete conversion of work to heat is possible whereas the reverse i.e. complete conversion of heat into work is not possible.
Heat transfer takes place from a hot body to a relatively colder body. The hotness or coldness of a body is defined by its temperature.
Also it is not a state variable but rather a path variable i.e. the heat transferred to a system does not depend on the states of the process but on the path followed.
When energy transfer takes place with a system undergoing a certain process, the internal energy of the system changes. The relation between the three can be found by using the energy conservation law i.e.
Where
heat supplied to the system from the surrounding
work done by the system on the surroundings
change in internal energy of the system
Equation (i) is also known as the First law of Thermodynamics.
The internal energy is a state property and thus we can say that the difference between heat supplied and work done is also a state property or in other words path independent.
Specific heat
Heat transfer generally produces a major effect of change in temperature. For ex: heating a body raises its temperature such as in the cooking process whereas cooling a body lowers the temperature of the body.
Heat transfer depends upon the temperature change of the body, quantity of the substance and properties of the body. The heat transfer is given by,
Where m is mass of the body
s is specific heat of the body
is change in temperature of the body
Specific heat is a property of a substance which is the amount of heat required to raise the temperature of the unit mass of a substance by unity.
Units of specific heat:
Dimensions of specific heat:
Modes of Heat Transfer
There are three major modes of heat transfer:
- Conduction:
- Heat conduction is the flow of internal energy from a region of higher temperature to that of a lower temperature by the interaction of adjacent particles (atoms, molecules, ions, electrons, etc.) in the intervening space.
- Heat transfer happens only due to the collisions between the neighbouring molecules.
- The rate of heat transfer through a medium via conduction is given by Fourier’s Law of Heat Conduction,
Where k = thermal conductivity of the material
A = cross-sectional area
temperature gradient across the medium.
- Convection:
- Heat transfer by the bulk movement of molecules within fluids such as liquids and gases.
- Convection heat transfer is further of two types i.e. natural and forced convection depending upon the involvement of external factors.
- The rate of heat transfer by convection is given by,
h = Convection coefficient
A = Surface area of the body
T = Temperature of the body
T0 = Temperature of the surrounding
- Radiation:
- It is a type of heat transfer that does not require any intervening medium.
- Radiation heat transfer uses electromagnetic waves to transfer heat from one body to another.
- There are many laws that govern radiation heat transfer but the most important is Stefan Boltzmann law.
- According to Stefan Boltzmann law, radiant energy emitted by a body is given by
Where ε= emissivity of the material.
A= surface area of the body.
𝜎= Stefan’s constant
T= absolute temperature of the body
Heat conversion to work
For the conversion of heat into work, a device is used which makes a system undergo a cyclic process. This device is known as a heat engine. The basic feature of a heat engine is shown in the image below.
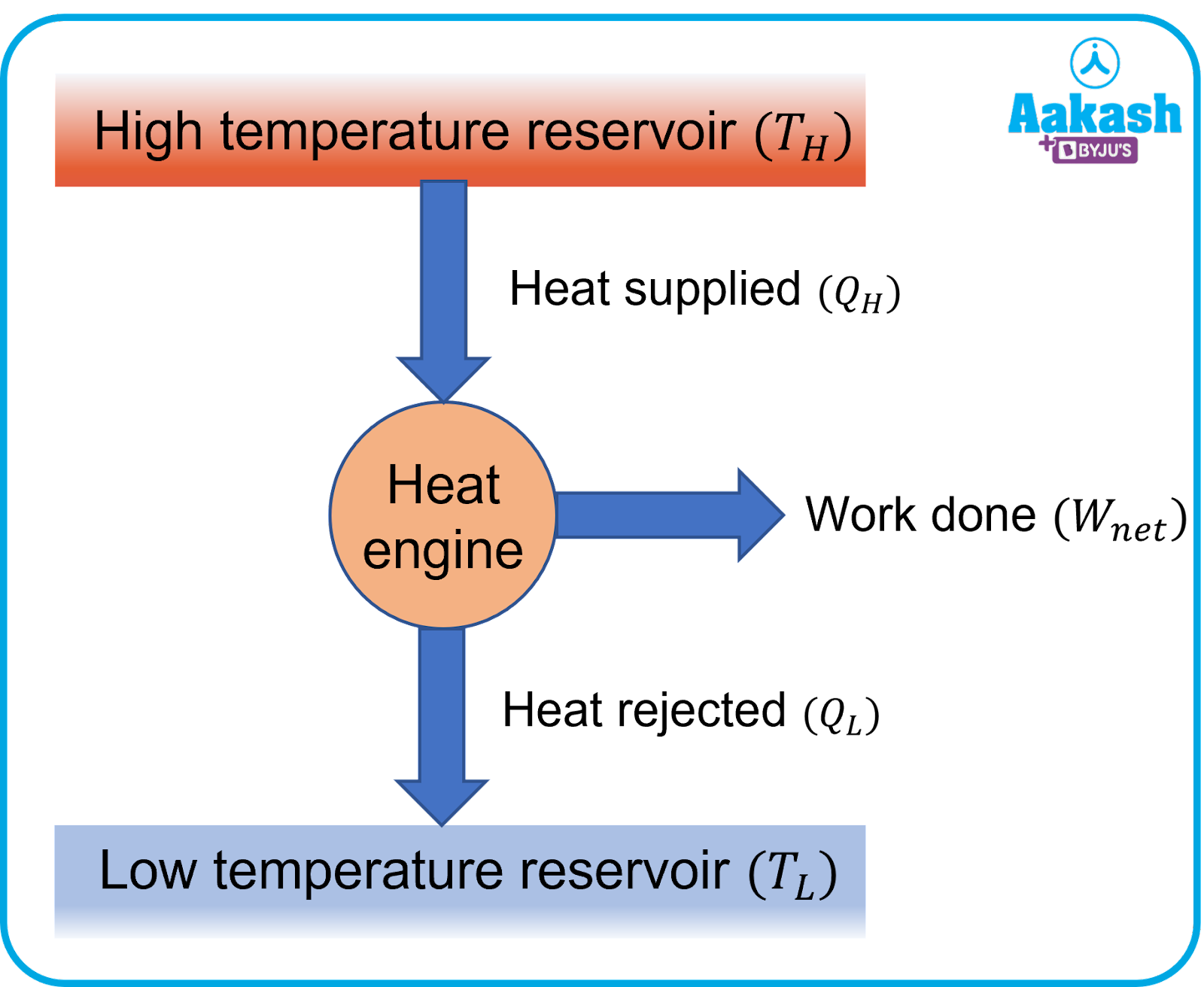
The mechanism involved in conversion of heat to work varies as per different heat engines. For ex: an external source is used to heat the system (steam) as done in steam engines or it may occur due to exothermic reaction such as in internal combustion engines.
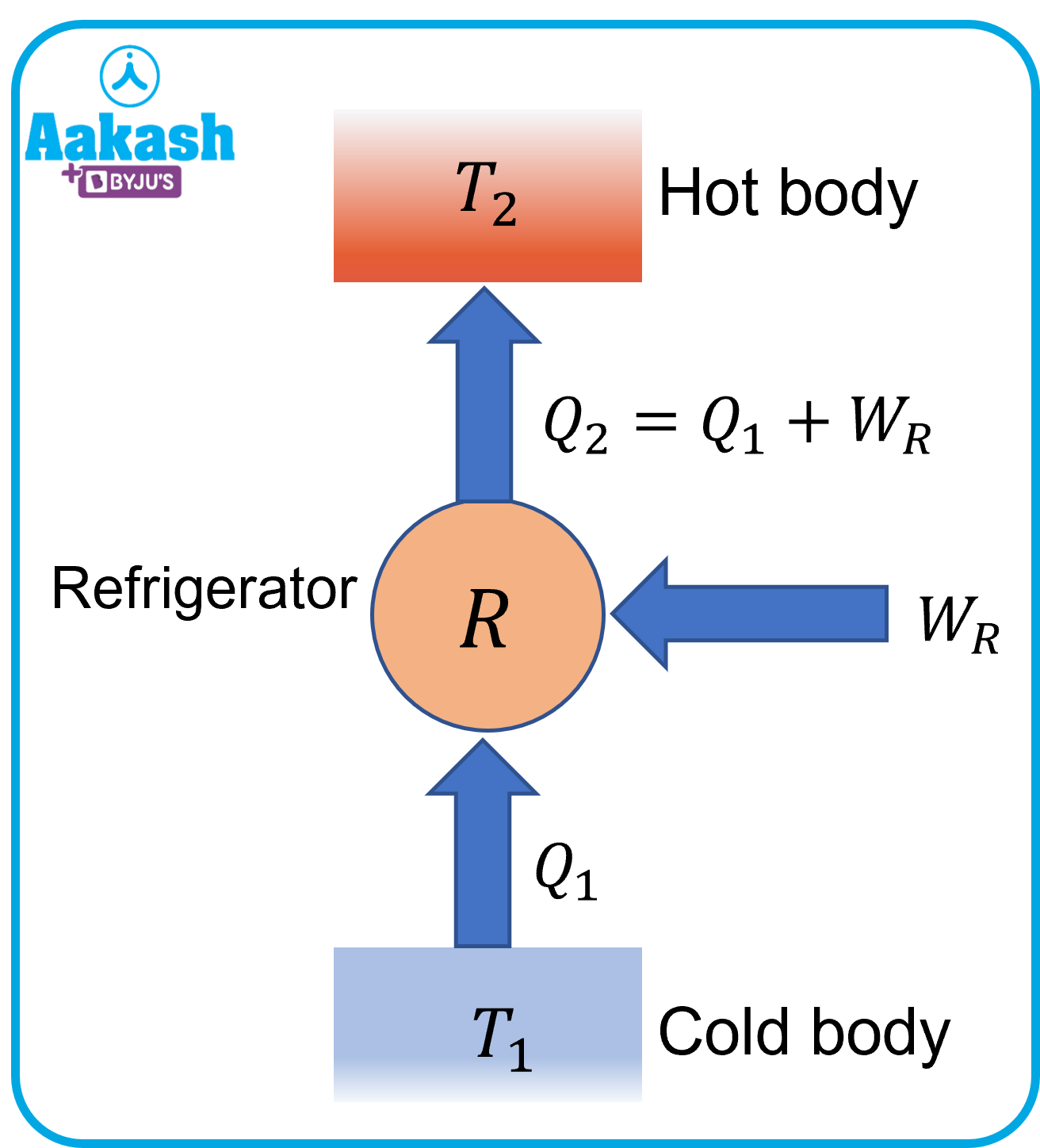
If we use the heat engine in reverse i.e. if we supply work to the engine, we can make the heat flow from low temperature to high temperature. The working of a refrigerator is one of the most common applications that is based on this process.
Practice problems
Q1. A real heat engine has an efficiency of 40%. The work output of the engine is 100 kJ per cycle. Calculate the amount of energy extracted per cycle from the high temperature reservoir.
Answer: Given,
Efficiency of heat engine, = 40 %
Work output, W=100 kJ
Using the formula,
Efficiency
Heat extracted,
Q2. The area enclosed by a PV graph for one thermodynamic cycle of a heat engine is 610 J. The thermal energy absorbed by the gas is 1300 J in one cycle. Determine the efficiency of the cycle.
Answer. Given that, the area enclosed by a PV diagram for one thermodynamic cycle of a heat engine is 610 J.
It means, work done, W=610 J
Thermal energy absorbed, Q=1300 J
Efficiency of heat engine in one cycle
Efficiency
Q3. Considering an ideal refrigerator, heat from its inside at a temperature of 277 K is transferred to a room at a temperature of 300 K. Find the amount of heat extracted for each Joule of electrical energy consumed by the refrigerator.
Answer.
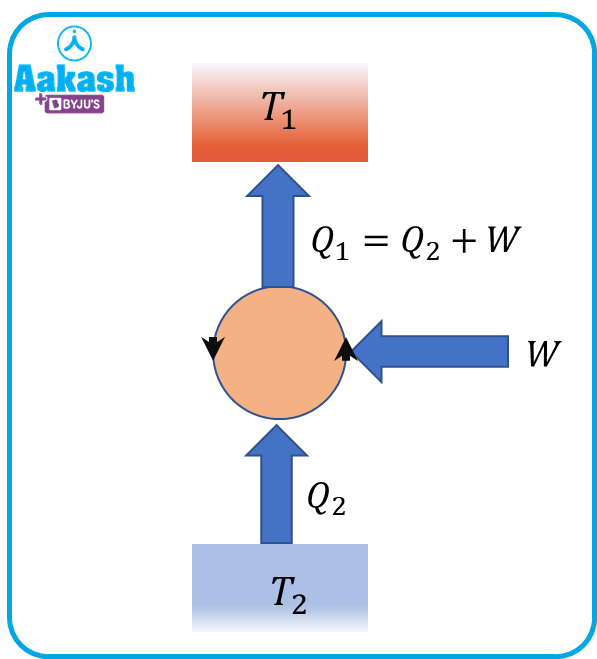
Coefficient of performance,
Given,
W = Electrical work consumed by refrigerator
W=1 J
From Eq.(i) we get,
Hence, the required amount of heat extracted by the refrigerator will be 12 J.
Q4. A real heat engine has an efficiency of 50%. The work output of the engine is 120 kJ per cycle. Calculate the amount of energy extracted per cycle from the high temperature reservoir.
Answer. Given,
Efficiency of heat engine, = 50 %
Work output, W=120 kJ
Using the formula,
Efficiency
Heat extracted,
FAQs
Q1. Can a process occur without any heat transfer across the system?
Answer: Yes. Adiabatic process occurs without any heat transfer across the system.
Q2. Is it possible that the temperature of a system decreases with the addition of heat?
Answer: Yes, it is possible that the temperature of the system may decrease upon heat addition. This occurs in polytropic processes which have negative specific heat. Basically the system does more work than the amount of heat added and due to this the internal energy of the system decreases, thus resulting in decrease in temperature of the system.
Q3. What is the unit of heat?
Answer: Being a form of energy, heat has the same units as that of energy i.e. Joules(J).
Q4. Which mode of heat transfer is fastest ?
Answer: Radiation is the fastest mode of heat transfer. In vacuum the heat transfer takes place at the speed of light .


