-
Call Now
1800-102-2727
Gas laws in KTG - Boyle’s Law, Charles's law, Gay-Lussac's law, Avogadro's law, practice problems, FAQs
You may have played cricket. When a bowler throws a ball at an angle it collides with the ground and applies a force on the ground. As per Newton's third law of motion the ground also applies a reaction force on the ball. Due to this reaction force the ball moves further.
Similarly a gas molecule can also be considered a tiny ball which collides with the wall of the container as shown in the figure below and applies a force on the wall of the container. This behaviour of the gas gives the birth to a theory known as the kinetic theory of gases. This theory consists of some laws which describe the behaviour of the gases. These laws are Boyle’s Law, Charles's law, Gay-Lussac's law and Avogadro's law. Let's learn about these laws in detail!

Table of content
- Gas laws
- Boyle’s Law
- Charles's law
- Gay-Lussac's law
- Avogadro's law
- Practice problems
- FAQs
Gas laws
The gas laws are a set of rules that establish relationships between the various parameters of the gas like: pressure, volume, temperature and number of molecules. These laws govern the behaviour of gases. The four basic laws of gases are;
- Boyle’s Law
- Charles's law
- Gay-Lussac's law
- Avogadro's law
We will understand each law one by one.
Boyle’s Law
In 1662, Robert Boyle gave this law. According to which, for a given mass of an ideal gas at a constant temperature, the volume V of the gas is inversely proportional to its pressure P.
Mathematically
Or,
Or, PV=k
Where k is the proportionality constant.
If V1 represents the volume of a quantity of gas at pressure P1, and V2 represents the volume of the same quantity of gas at pressure P2, the above equation can be written,
P1V1=P2V2
Or,
The graph between pressure P and volume V can be represented as,
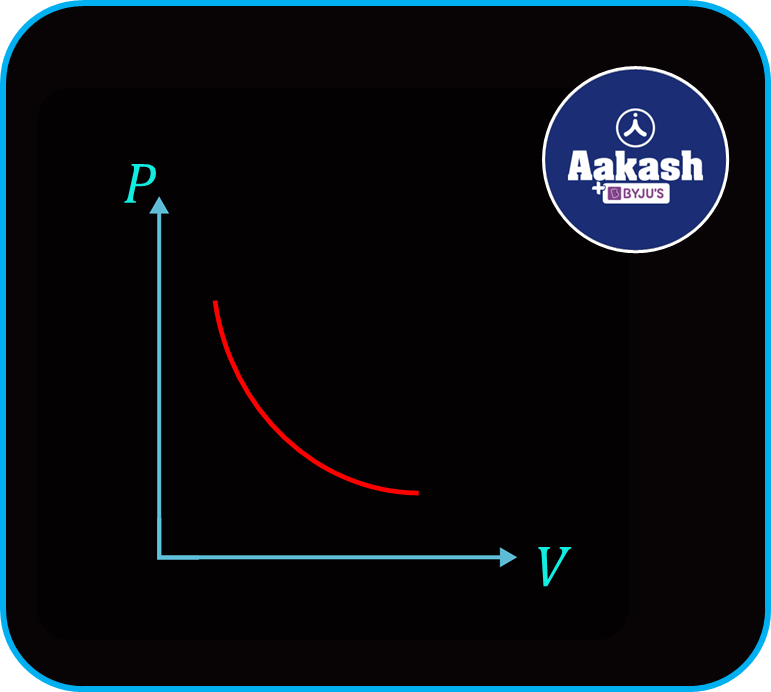
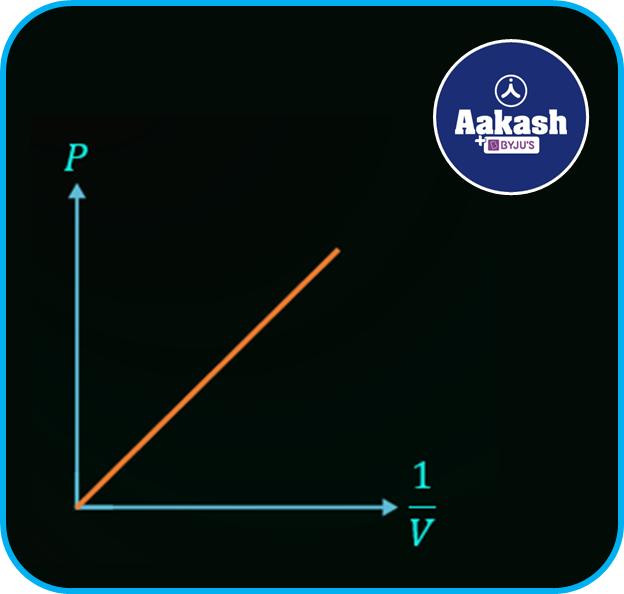
Also the graph between pressure P and reciprocal of volume 1V can be represented as,
Charles's law
This law was given by a French scientist- and inventor Jacques Charles around 1787.
According to Charles's law, for a constant pressure, the volume of the given mass of a gas is directly proportional to its absolute temperature.
Mathematically, VT
Where k is constant.
Let us assume that when a gas with initial volume V1 at temperature T1 is heated to a temperature T2, the volume becomes V2 at a constant pressure. Then
The graph between the volume V and absolute temperature T is shown in the figure below,
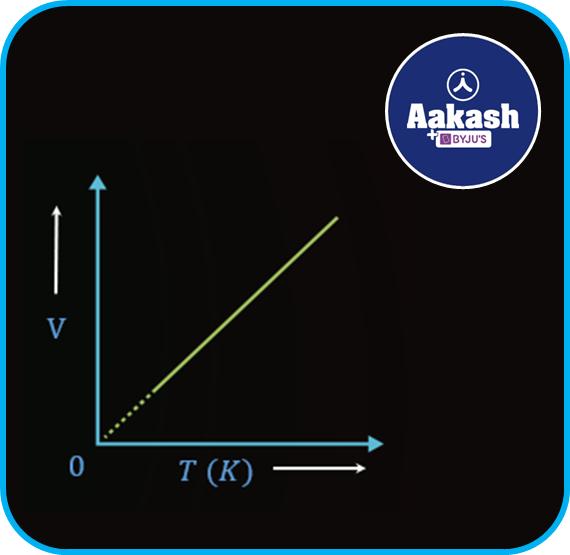
Gay-Lussac's law
It is given by the a French chemist Joseph Louis Gay-Lussac in 1808. According to Gay-Lussac's law, for a constant volume, the pressure of the given mass of a gas is directly proportional to its absolute temperature.
Mathematically, PT
Or P=kT
Or PT=k
Where k is a constant.
Consider a gas having a temperature T1 exert a pressure P1. If the temperature of the gas is increased to T2, then pressure of the gas become P2. Then
The graph between the Pressure P and absolute temperature T is shown in the figure below,
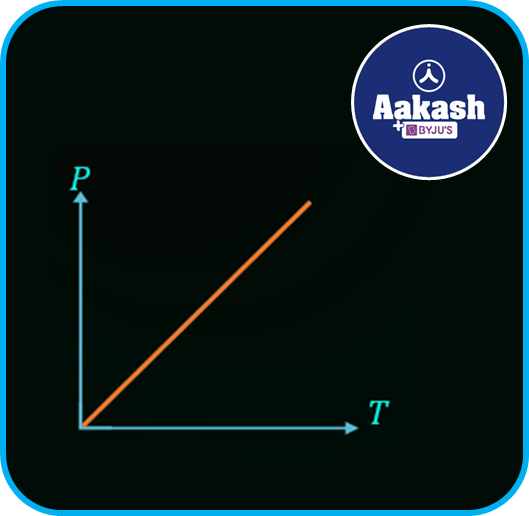
Avogadro's law
Amedeo Avogadro, an Italian scientist, proposed this law in 1811. According to the avogadro’s law, for the same condition of temperature and pressure, equal volumes of different gases contain equal numbers of moles.
Mathematically, Vn
Or V=kn
Or
Where n is the number of the moles and k is a proportionality constant.
If a balloon contain n1 mole of a gas and having volume V1 and another balloon contain n2 mole of another gas and having volume V2 . Then
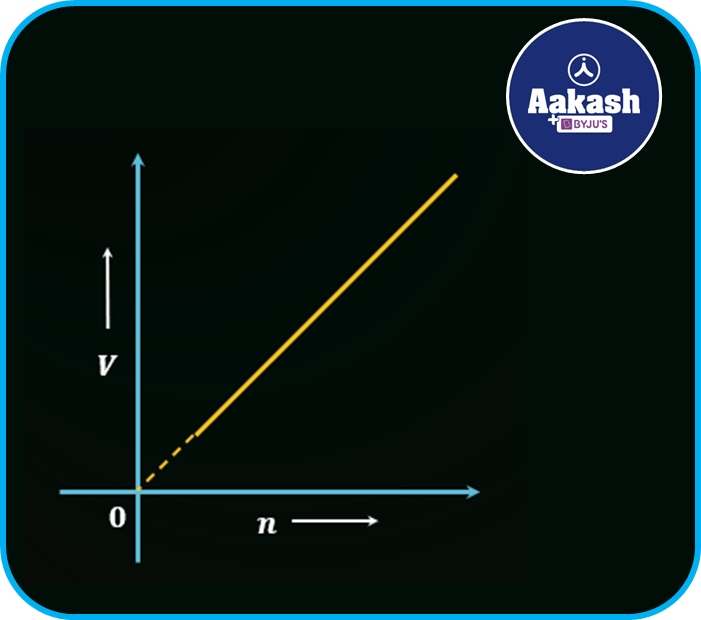
The graph between the volume V and number of mole n is shown in the figure below,
Practice problems
Q. At 5 atm, a 20 mL sample of gas is present. If the quantity of the gas and temperature remain constant and pressure rises to 7 atm, what will be the volume of the gas?
A. Given P1=5 atm and P2=7 atm
V1=20 mL
Using the Boyle’s Law, we have
V2=14.28 mL
The new volume of the gas is 14.28 mL.
Q. A sample of carbon dioxide with a volume of 25 mL and a temperature of 27oC is in a pump. Find the new volume of carbon dioxide in the pump if the temperature is raised to 77oC while the pressure stays constant.
A. Given
V1=25 mL
Using Charles's law, we have
V2=29.16 mL
Hence the volume of the gas is 29.16 mL
Q. A bottle filled with the gas is heated to 327oC, at which point it bursts, from room temperature of 27oC and a pressure of 2 atm. Determine the heated soda bottle's pressure.
A. Given T1=27+273 K=300 K and T2=327+273=500 K
P1=2 atm
Applying the Gay-Lussac’s Law,
P2=3.33 atm
Hence the pressure at the time of bursting is 3.33 atm.
Q. Consider 50 moles of hydrogen gas with a volume of 6.95 m3 . Calculate the volume of 80 moles of nitrogen gas under the same pressure and temperature.
A. Given n1=50 mole and n2=80 mole
V1=6.95 m3
According to the Avogadro’s Law
The volume of 80 mole nitrogen is 11.12 m3
FAQs
Q. What is ideal gas?
A. An ideal gas is one that obeys Boyle's law, Charle's law, Gay - Lussac’s and Avogadro's law under all temperature and pressure conditions. Such gas does not exist in reality; it is simply a hypothetical concept.
Q. What is the kinetic theory of gases?
A. According to this theory, gases are made up of many molecules, which are moving at a certain speed and in an arbitrary direction. Their position or speed can be used to determine the macroscopic properties of gases like temperature, pressure and volume.
Q. A syringe and a bicycle pump works on which law?
A. Syringes and a bicycle pump are based on Boyle's law. As the volume of the cylinder is reduced , the pressure inside the cylinder increases.
Q. What is the daily life example of Gay - Lussac’s Law?
A. The pressure cooker is a good example of Gay-Lussac’s Law. As the temperature rises, the pressure of the fluid inside the cooker increases, when a pressure cooker is placed on a heat source.


