-
Call Now
1800-102-2727
Dielectric property and polarization, Effect of dielectric on capacitance, Practice problems, FAQs
In a capacitor when the gap between the metal plates is filled with a bad conductor, the charge retention dramatically increases. Conversely, when the medium is filled with more conducting material the charge retention decreases. Charge retention is nothing but capacitance. The retention of charge depends upon the polarization of the material. The atoms in a material have a dipolar property which basically dictates the dielectric nature. Let’s learn more about this in this article!
Table of content
- Dielectric property and polarization
- Effect of dielectric on capacitance
- Capacitor with partially filled dielectric
- Practice problems
- FAQs
Dielectric property and polarization:
A dielectric is made up of polar and nonpolar molecules. The net effect of an external field will compel the molecules to align their dipole moments along its direction.
Let us consider a dielectric slab in an electric field which is acting in the direction as shown. The arrangement of charges within the molecules of the dielectric in the electric field is also shown. The positive charges move in the direction of the field and the negative charges in the opposite direction. The electric dipoles align themselves with the direction of the field. In this state, the entire dielectric and its molecules are said to be polarised.
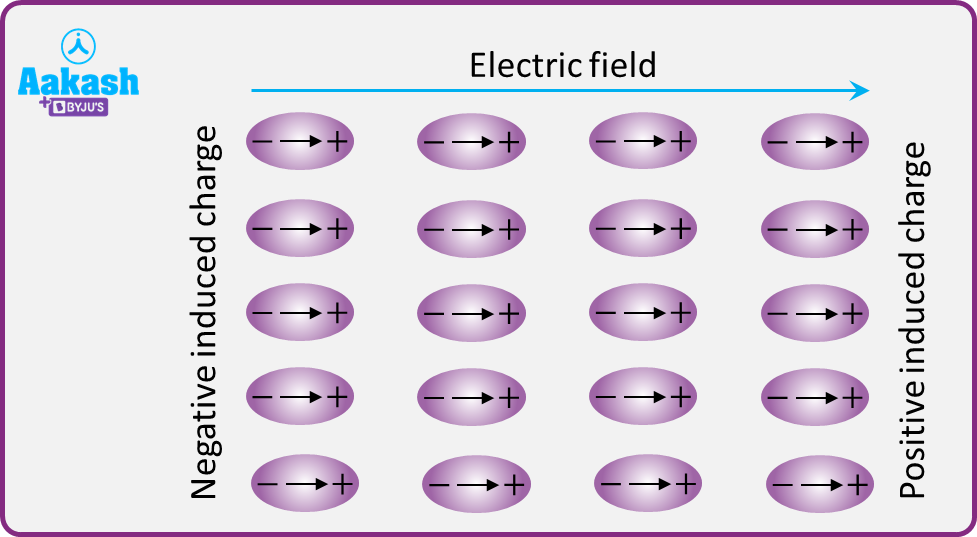
The alignment of the dipole moments of the permanent or the induced dipoles, along the direction of the applied electric field, is called polarisation.
Within the two extremely thin surface layers, there is an excess negative charge in one layer and an excess equal positive charge in the other layer.
The induced charges which are on the surfaces of the dielectric are due to these layers. They are not free but each is bound to a molecule which lies on or near the surface. That is why these charges are called bound charges or fictitious charges. Within the remaining dielectric, the net charge per unit volume remains zero. Thus, although the dielectric is polarized, yet as a whole, it remains electrically neutral.
The positive induced surface charge is equal in magnitude to the negative induced surface charge. Thus, in polarisation, the internal state of the slab is characterised by the relative displacement of the charges within it, not by an excess charge.
Polarisation can be thought of as a phenomenon where an alignment of positive and negative charges takes place within the dielectric which results in no net increase in the charge of the dielectric.
The extent to which a dielectric is polarized is described by a vector quantity P, called polarisation.
Meanwhile, polarisation is defined as the electric dipole moment per unit volume.
It has the same direction as the molecular dipole moment.
The SI unit of P is Cm-2.
The dimensional formula for
Where, I = Current, L = Length, T = Time
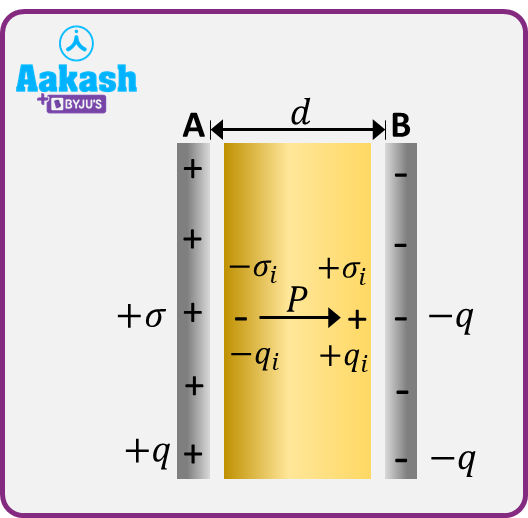
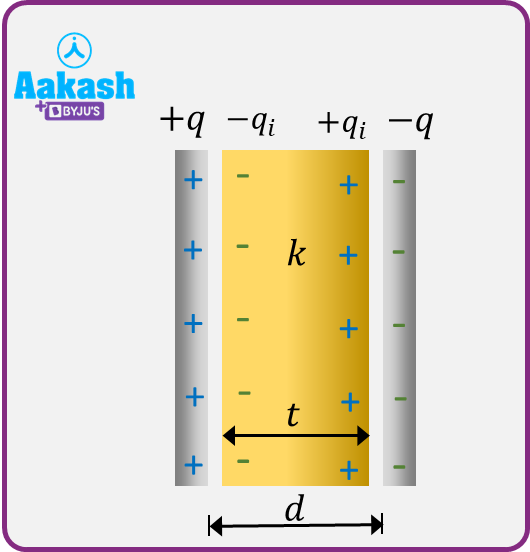
let us consider a dielectric slab of surface area A and thickness d . Let this slab completely fill the space between the plates A and B of a parallel-plate capacitor, having charges +q and –q. Let –qi and +qi be the induced charges on the surfaces of the slab opposite to A and B respectively.
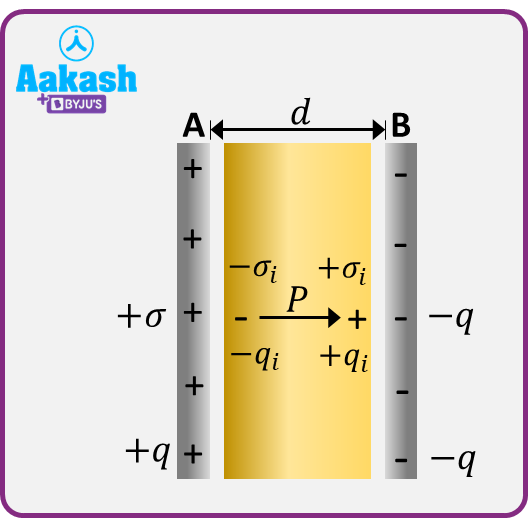
The polarised slab can be considered as a single dipole consisting of the induced charges –qi and +qi at the opposite faces separated by a distance equal to the thickness of the slab.
The dielectric slab fills the entire space between the plates in a capacitor. The small gap left between the surfaces and those of the plates is only for the sake of drawing but actually no gap is there.
Electric dipole moment of the entire slab =qid
Volume of the entire slab =Ad
Thus, polarisation,
i=surface density (charge per unit area) of the induced charge.
When P is normal to the surface, the polarisation is numerically equal to the surface density of the induced charge.
It is found in most of the dielectrics except certain crystals which are called ferroelectrics. The polarisation P is directly proportional to the resultant electric field E exists in the dielectric.
Xe is the susceptibility of a material. It is dimensionless. More the susceptibility, more will be the induced charge i.e., more polarization.
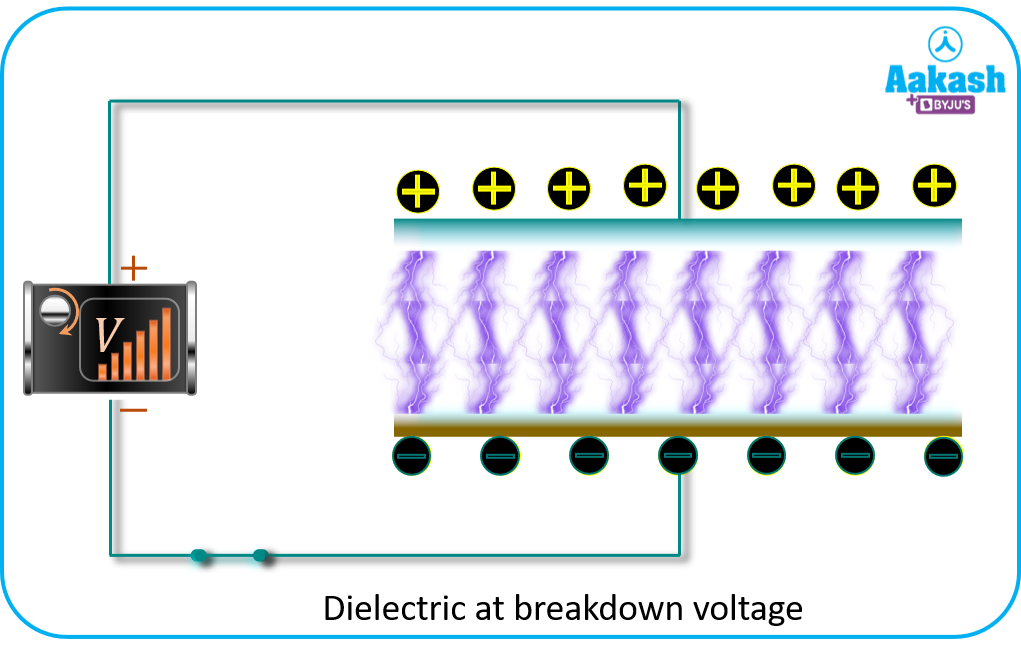
If a strong electric field is applied, dielectric will break and the charges gets separated
and the dielectric acts as a conductor. This is known as Dielectric breakdown.
The maximum potential that the dielectric medium can sustain is known as Dielectric strength of the dielectric medium.
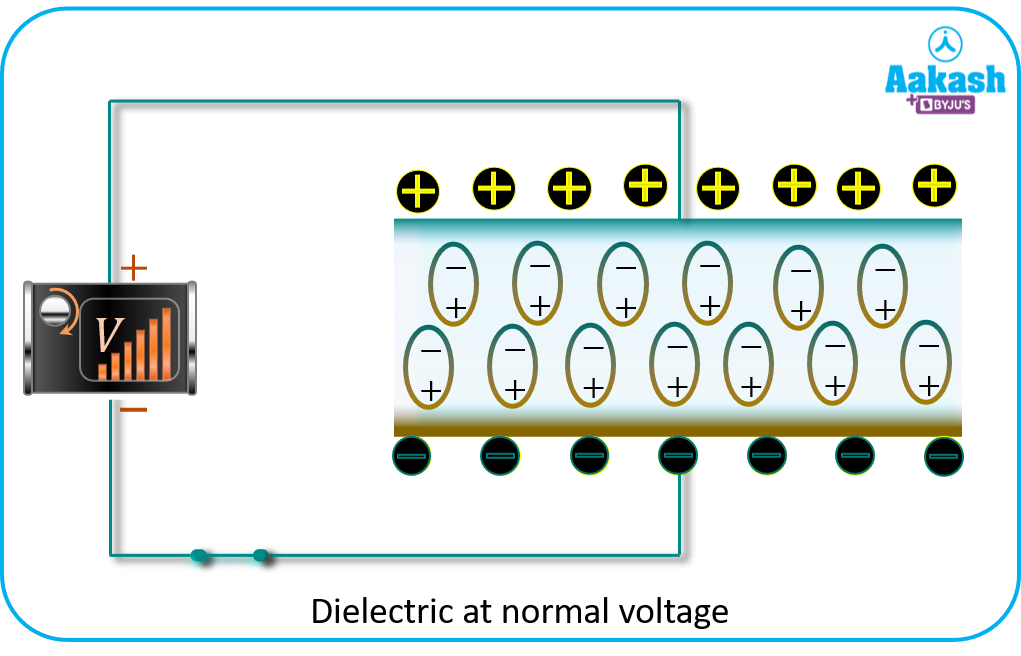
Effect of dielectric on capacitance
Capacitors usually have a non-conducting material or a dielectric between the conducting plates or electrodes. By placing a solid dielectric between the conducting plates of a capacitor, it enables the following three functions
a. a solid dielectric solves the problem of maintaining two large metal sheets at a very small separation without actual contact
b. A dielectric increases the maximum possible potential difference which can be applied between the plates of the capacitor without any electrical breakdown of dielectric placed in between the electrodes.
c. It increases the capacity of charge retention. If a dielectric with relative permittivity K is inserted between the electrodes, the potential difference decreases by a factor K. The electric field decreases because of the induced charge of the opposite sign that appears on the surface of the dielectric. This induced charge produces an electric field inside the dielectric in the opposite direction and as a result the net electric field decreases.
Capacitor with partially filled dielectric
If the space between the two plates of a capacitor be filled partially with a dielectric of dielectric constant k and a charge q is given to the capacitor, an induced charge, qi is developed in dielectric.
Electric field inside the dielectric will be, E=E0k where E0 is the electric field in the region in the absence of the dielectric.
The potential difference between the plates of the capacitor is,
So, the capacitance will be,
Different cases:
a. If there is a conducting slab of
b. If the space is completely filled with a conductor, k→∞, d=t, C→∞
c. If there are n dielectric slabs, C=A0d-t1-1k
d. If the space is completely filled with a dielectric,
Practice Problems
Q. Calculate the polarization produced in 1.5 mm thick polystyrene subjected to 240 V. The relative dielectric of polystyrene is 3.5.
A. We know,
Here,
So, the polarization is,
Q. Electric field inside a capacitor is measured as 50 V/m. The dielectric constant is 4.5. Calculate the polarization.
A. Susceptibility,
So, the polarization is,
Q. Two conducting plates of 0.2 m2 are placed 4 mm apart. A conducting slab of 2 mm thickness is inserted in between. What will be the capacitance of the arrangement?
A. The capacitance for such a system can be calculated as,
Q. Two conducting plates of 0.2 m2 are placed 4 mm apart. A polystyrene slab of 2 mm thickness and dielectric constant 4 is inserted in between. What will be the capacitance of the arrangement?
A.
The capacitance for such a system can be calculated as,
FAQs
Q. What are polar molecules?
A. These types of dielectrics in which the molecules are all asymmetrical in form. H2O, CO2, NO2, and other gases are examples. In the absence of any electric field, the electric dipole moment of these molecules moves in an unpredictable direction. As a result of this, the average dipole moment is 0. In the presence of an external electric field, the molecules assemble in the same direction as the electric field.
Q. What are non-polar molecules?
A. These are symmetrical molecules. So, there is no persistent dipole moment in the molecule. O2 N2, H2 are examples of non-polar molecules.
Q. What is polarizability?
A. When the matter is exposed to an electric field, it has a propensity to develop an electric dipole moment that is proportional to the applied field. Because matter is made up of fundamental particles with an electric charge, such as protons and electrons, it is a property of all matter.
Q. What is dielectric constant?
A. The Dielectric constant is the ratio of the applied electric field strength to the strength of the decreased value of the electric field in a capacitor when a dielectric slab is placed between the parallel plates. The formula is as follows:
here E0 is the applied electric field, E is the net field, & r is the dielectric constant.
Greater the dielectric constant, the greater the amount of charge that can be retained inside. Thus, the capacitance of a capacitor is increased by a factor of the dielectric constant when the gap between the conducting plates is completely filled with a dielectric.
C0 is the capacitance between the plates with no dielectric.


