-
Call Now
1800-102-2727
ZnCl2- Introduction, Preparation, Properties, Uses, Practice Problems, FAQs
There was a girl whose name is Meenal, studying in class 12. Her mother, around 45 years old, was suffering from a dental problem. Her mother asks Meenal to take her to the dentist. Meenal took an appointment with one of the dentists at a hospital nearby her house.
When they visited the dental clinic, the doctor checked her mother’s teeth and then said that there is a cavity present inside, and that is creating an issue due to this her mother is suffering from pain. The doctor advises filling the Cavities, which are small holes created by portions of a tooth that are rotting, and are frequently treated with dental fillings. Dentists will fill these holes during a filling procedure using a material like composite or amalgam.
One of the famous mixtures where filling material is zinc chloride. This is mixed with zinc oxide to make a syrupy-like liquid used in the dental filling.
Zinc is an important element which has many important properties and compounds of zinc are beneficial for humans. Let us study one of the critical compounds of zinc i.e, zinc chloride in a bit detail.
TABLE OF CONTENT
- ZnCl2-Introduction
- ZnCl2-Preparation
- ZnCl2-Properties and reactions
- ZnCl2-uses
- Practice Problems
- Frequently Asked Questions-FAQs
ZnCl2-Introduction:
Inorganic chemical compounds having the formula ZnCl2 and its hydrates are known as zinc chloride. Nine crystalline forms of zinc chlorides are known, and they are all colourless or white. They are also very soluble in water. This salt is hygroscopic and even deliquescent. In metallurgical fluxes, chemical synthesis, and textile processing, zinc chloride is widely used.
ZnCl2-Preparation:
- It is created by combining, zinc compounds, like zinc oxide, zinc carbonate, or zinc hydroxide with dil. hydrochloric acid. The solution produces hydrated zinc chloride crystals, ZnCl2.2H2O, after concentration and cooling.
- Hydrated zinc chloride may undergo hydrolysis to form the basic chloride (zinc hydroxy chloride), which on further heating yields zinc oxide. Hence the anhydrous zinc chloride cannot be obtained by heating a hydrous zinc chloride.
- Anhydrous zinc chloride is made by heating zinc in dry chlorine or dry HCl gas environment.
- Zinc powder can be distilled with mercuric chloride to from anhydrous zinc chloride.
ZnCl2-Properties and Reactions:
- Anhydrous zinc chloride is a deliquescent white solid that is water-soluble.
- It melts at 660 ℃ and boils at 730℃
- Heat transforms hydrated zinc chloride into zinc hydroxy chloride or zinc oxychloride.
- A whitish substance forms when H2S is circulated through the solution. A zinc sulphide precipitate is generated.
- When NaOH is added, a white zinc hydroxide precipitate forms, which dissolves in excess sodium hydroxide to create sodium zincate.
- When you add NH4OH solution, a white zinc hydroxide precipitate forms, which dissolves in excess ammonia to form a complex salt.
- A white precipitate of basic zinc carbonate is generated when zinc chloride solution is treated with sodium carbonate solution.
When a sodium bicarbonate solution is employed, however, a white precipitate of typical zinc carbonate is created.
- Ammonia gas is absorbed by anhydrous zinc chloride, which forms an addition compound called adduct.
- It dissolves cellulose with its syrupy solution.
- When combined with zinc oxide, ZnO, the syrupy solution forms a hard substance called oxychloride,ZnCl2.3ZnO.
ZnCl2-Uses
- The Lucas test uses a solution of anhydrous zinc chloride in strong hydrochloric acid to distinguish and classify primary, secondary, and tertiary alcohols. The Lucas reagent is the name given to this mixture. Tertiary alcohol interacts with the Lucas reagent upon addition, producing an alkyl chloride as a precipitate right away. If the precipitate appears a few minutes later, the alcohol is secondary. If no precipitate is acquired in cold, alcohol is considered a primary one.
- A typical laboratory reagent that is helpful as a Lewis acid in organic chemistry is zinc chloride.
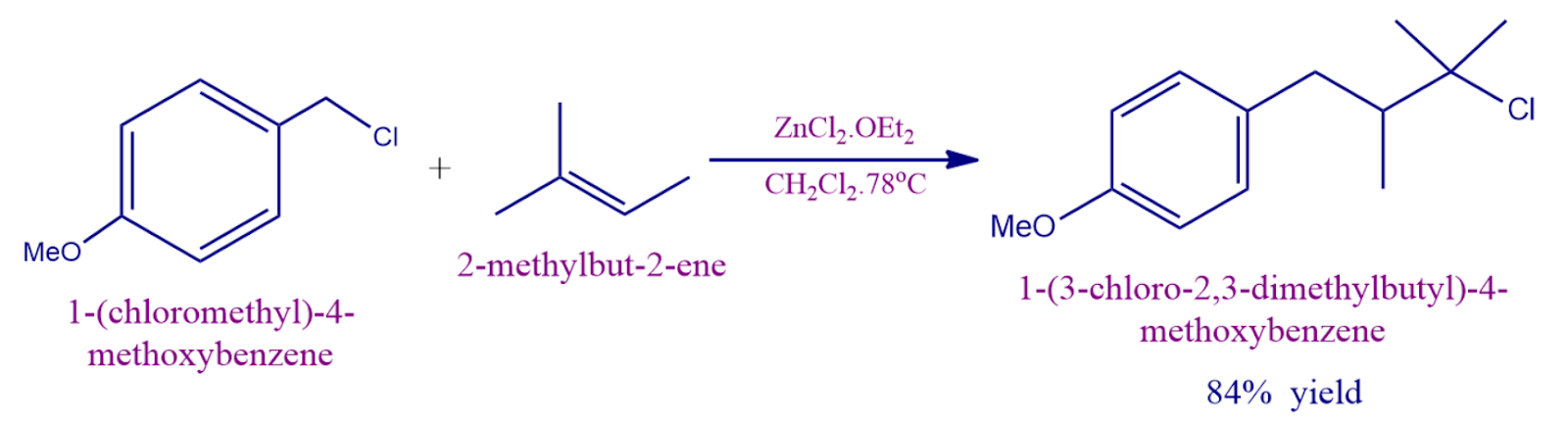
- Additionally, zinc chloride activates benzylic and allylic halides so that weak nucleophiles like alkenes can replace them.
- Starch, silk, and cellulose may all dissolve in concentrated zinc chloride aqueous solutions (greater than 64% weight/weight of zinc chloride in water).
- ZnCl2 is employed as a fireproofing agent and in fabric "refreshers," such as Febreze, which is pertinent to its affinity for these materials. Paper is soaked in concentrated zinc chloride to create vulcanised fibre.
- Timber is impregnated with a strong solution of zinc chloride to protect it from being destroyed by microorganisms.
- Dental fillings are made up of a syrupy zinc chloride solution and zinc oxide.
- It is used to clean the surface of metals before soldering since it is acidic in an aqueous solution.
- It's used in producing parchment paper, vulcanised fibre, and cold water glues and adhesives. It's also utilised in dry cell manufacturing.
Practice Problems:
Q1. A white precipitate of ____________ is generated when zinc chloride solution is treated with sodium carbonate solution.
A. Basic zinc carbonate
B. zinc oxide
C. sodium zincate
D. None of the above
Answer: A
Solution: A white precipitate of basic zinc carbonate is generated when zinc chloride solution is treated with sodium carbonate solution.
Q2._____________ is the oxidation state of Zinc in zinc chloride.
A. 0
B. +1
C. +2
D. +3
Answer: C
Solution: Let the oxidation state of zinc in ZnCl2 be x.
For calculating this we know that the oxidation state of the chloride we consider is -1.
Now, calculating this we get,
Hence, +2 is the oxidation state of zinc in ZnCl2 .
Q3. When you add NH4OH solution, a ____________ coloured precipitate forms.
A. White
B. Black
C. Red-brown
D. Yellow
Answer: A
Solution: When you add NH4OH solution, a white-coloured precipitate of zinc hydroxide is formed. Here is the mentioned reaction for this.
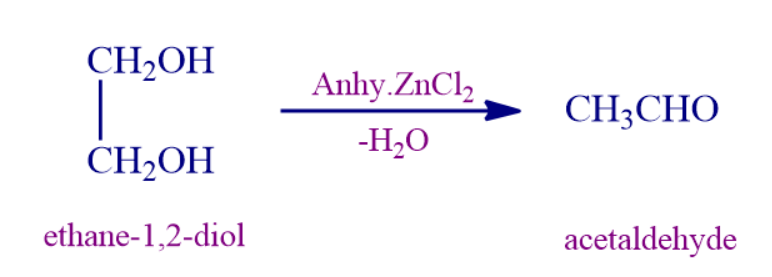
Q4. With anhydrous zinc chloride, ethylene glycol gives ____________ as the major product.
A. Acetylene
B. Acetaldehyde
C. Formaldehyde
D. Acetone
Answer: B
Solution: With anhydrous zinc chloride, ethylene glycol gives Acetaldehyde as the major product.
Frequently Asked Questions - FAQs:
Q1. Is it safe to consume zinc chloride?
Solution: Zinc chloride has strong irritant and corrosive properties when inhaled. Ingestion of even 10 mL of a 35% solution of zinc chloride can cause oropharyngeal and stomach burns, epigastric discomfort, pharyngeal oedema, haematemesis, and melaena due to the severe corrosiveness of the substance.
Q2. Zinc chloride is covalent or ionic. Explain.
Solution: Ionic compounds include zinc chloride. This chemical has zinc as the cation and chlorine as the anion. However, it also possesses some covalent characteristics.
Q3. Is zinc chloride a neutral salt, acidic or an alkali base?
Solution: Acidic salts are produced by the salts of strong acid and weak base. Because ZnCl2 is is created by the reaction of a strong acid,HCl and a comparatively weak base, Zn(OH)2, zinc chloride is a weakly acidic salt. Thus,ZnCl2 has a mildly acidic character.
Q4. What other applications does zinc chloride have?
Solution: Zinc chloride is a crystalline (sand-like), white, odourless granule or powder. It is employed in the production of agricultural chemicals, pharmaceuticals, textiles, screening smoke for crowd control, iron galvanising, soldering flux, crowd control smoke, and wood preservative.


