-
Call Now
1800-102-2727
Wave Nature of Light: Davisson & Germer Experiment, de Broglie Relation, Wave Nature of Light, Practice Problems & FAQs
Which is your favorite superhero movie?
Even if you are not a big fan of movies, you must have heard about Spiderman. A common boy accidentally gets bitten by a spider and mutates into a man having the power of climbing walls and spreading the web like a spider. To protect his identity from others he doesn’t reveal his face and maintains a dual personality. A common boy from the next door and a mighty hero who is always up against evil.

Similarly light possesses dual nature. For ages light was considered a particle. Until Louis de Broglie noticed something different about light. After brief experimentation and examination, he proposed a hypothesis that light has some wave nature.
In this concept page, we will explore the wave nature of light!

Table of Contents
- Davisson Germer Experiment
- Conclusion
- De Broglie Relation
- Characteristics of Light
- Practice Problems
- Frequently Asked Questions-FAQs:
Davisson Germer Experiment:
In an attempt to demonstrate the wave nature of electrons, Clinton J. Davisson and Lester H. Germer looked into the diffraction of an electron beam being scattered by nickel crystals. The experiment also supported the early de-Broglie hypothesis, which proposed that matter contains a wave component in addition to its particle nature.
Experimental apparatus of Davisson Germer experiment:
The illustration used by Clinton J. Davisson and Lester H. Germer to show how electrons behave like waves is shown below. A low tension voltage source heats a tungsten filament in an electron cannon that is part of the arrangement. By creating the necessary potential difference with the aid of a high tension voltage source, the accelerator in the cannon is responsible for speeding up thermally excited electrons from the filament. As a result, the accelerator allows for the modification of electron velocity. Nickel crystal that is in its path is impinged by a fine stream of electrons. To avoid electrons striking atoms in the course of an electron beam, the device is enclosed in a vacuum chamber. The illustration shows the placement of a mobile collector with a spinning galvanometer in the direction of the diffracted electron beams. The collector, which functions like a Faraday cup electron detector since the nickel target is movable, counts the electrons that diffract after striking it from different angles.
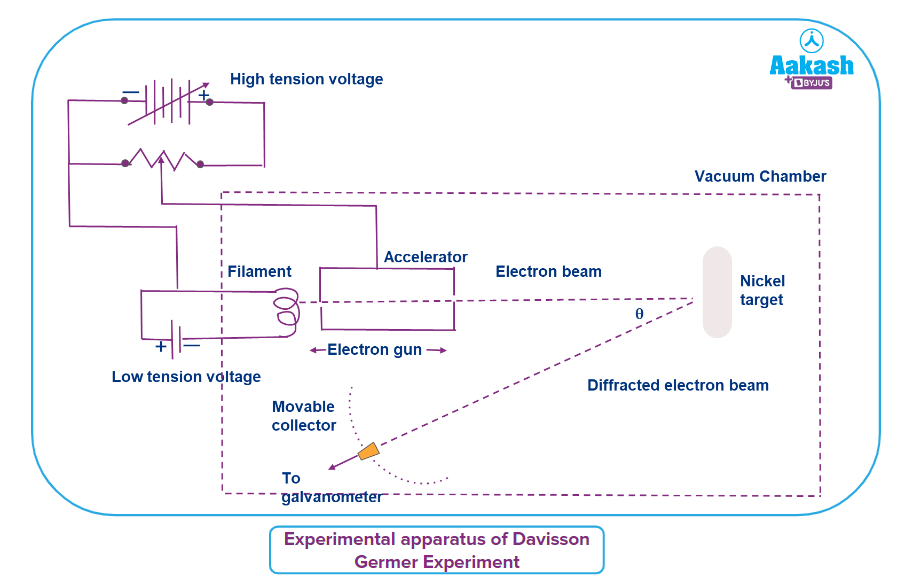
Working of Davisson Germer experiment:
When the filament is heated, electrons are released through thermionic emission. An accelerator is then used to accelerate the released electrons into an electron beam. The standard impact of the electron beam on a nickel target follows. The atoms of the nickel target scatter the incoming electrons in various ways. The intensity of the dispersed electron beam in a certain direction can be calculated by rotating the mobile collector. The galvanometer, which is connected to the mobile collector and measures the photocurrent to ascertain the strength of the electron beam, measures the photocurrent.
Conclusion:
In their experiment, Davisson and Germer discovered some intriguing findings. A distinct peak intensity was seen as the electron beam's accelerating voltage was increased at a particular value. This voltage was determined to be exactly 54 volts. Momentum exchanges in electron collisions cannot account for the peak. The constructive interference of electrons distributed from various crystallographic levels is the only explanation that makes sense. Without a doubt a wave property.
This voltage corresponds to a de Broglie wavelength of 0.167 nm. De Broglie's theoretical discovery of this wavelength for electron matter waves was exact.
This is how the Davisson and Germer experiment clarified the electron's wave-like characteristics and discovered its corresponding wavelength using the de Broglie relation.
De Broglie Relation:
Using Planck's equation and Einstein's mass-energy connection, the wavelength calculated by de Broglie is:
According to the Plank’s equation;
E = h𝜈....(i)
𝜈 is the frequency of the photon
λ is the wavelength of the photon
c is the speed of the light
h is the Planck’s constant
According to the Einstein’s equation;
m is the mass of the particle
c is the speed of the light
Equating equation (i) and (ii), we get;
Similarly for particles (matter),
where p is the momentum of the particle.
Characteristics of Light:
According to the wave theory of light:
- Energy is transmitted in the form of electromagnetic radiation or waves.
- The radiation consists of the electric and magnetic fields that oscillate in a perpendicular direction to each other.
- The electromagnetic waves do not require any medium for propagation.
- All the electromagnetic waves travel with the same velocity as that of the light, i.e., 3 x 108 m per sec.
- There are various types of electromagnetic radiation which differ from one another in frequency or wavelength. These form the electromagnetic spectrum.
Recommended Video:
Wave Nature of Electromagnetic Radiation Class 11 Chemistry - Atomic Structure (L3) | NEET 2024
Practice Problems:
Q1. What causes light to move in waves?
- Photons
- Electromagnetic nature of light.
- Electric field
- Magnetic field
Answer: (B)
Solution: Every form of energy is believed to behave like an electromagnetic wave. As light is a form of energy, light also has electromagnetic waves. This causes light to move in waves.
Q2. What will be the mass associated with a ball having De-Broglie wavelength 6.626 10-37m moving with velocity 100 m sec-1.
- 10 g
- 100 g
- 1000 g
- 10000 g
Answer: (D)
Solution: Let, weight of ball = x g
We can see, order of the wavelength of the daily life particle is extremely small, therefore de-Broglie wavelength calculation for the daily life particle has no physical significance
Q3. The approximate ratio of de-Broglie wavelength of neutron and electron moving with the same speed is about:
- 1:4
- 1:1839
- 1:1
- 1:2
Answer: (B)
Solution:
Mass of electron = mass of 1 proton
mass of electron = mass of 1 neutron
(but for numerical simplicity we consider, mass of a proton = mass of a neutron)
According to the question, both electron and neutron have the same velocity.
( for electron)
( for neutron)
Q4. Which of the following has the lowest de-Broglie wavelength (all have the equal velocity)
- Hydrogen gas molecule
- Electron
- Proton
- Nitrogen gas molecule
Answer: (B)
Solution:
We know,
Species have equal velocity, wavelength is inversely proportional to their mass. So, electrons have the smallest mass in all given options.
Frequently Asked Questions-FAQs:
1. What is the de-Broglie hypothesis's flaw?
Answer: Because the order of the wavelength of a daily life particle is so tiny, de-Broglie wavelength computation for the daily life particle has no physical meaning.
2. What do you understand about the dual nature of electrons?
Answer: Electrons may be thought of as both a particle and a wave at the same time.
3. Which experiment conclusively proved the de-Broglie hypothesis?
Answer: The Davisson and Germer experiment revealed the electrons' wave character, validating de Broglie's previous notion. When electrons are dispersed from crystals with correctly spaced atoms, they display diffraction.
4. De-Broglie was awarded the nobel prize in physics for which achievement?
Answer: De-Broglie awarded nobel prize in physics (1929) for discovery of wave nature of electrons.


