-
Call Now
1800-102-2727
Variation of Molar Conductivity with Dilution in Strong and Weak Electrolytes: Molar Conductance Behaviour of Strong and Weak Electrolytes, Kohlrauch’s law, Practice Problems and FAQs:
If you have to choose between a copper wire and aluminum wire for your household wiring, which one will you choose?

Obviously, you will opt for copper wire because it has higher conductivity and lower resistivity value than aluminum wire.
Well, we learnt that not only metals but some aqueous solutions do conduct electricity. These are called electrolytes. There are two types of electrolytes: strong electrolytes and weak electrolytes. Both have a different conductivity values.
Let’s understand how the molar conductivity values of weak and strong electrolytes are getting affected due to dilution.
Table of content
- Conductance (G)
- Conductivity/ Specific conductance
- Molar Conductivity
- Equivalent Conductivity
- Strong Electrolytes
- Weak Electrolytes
- Molar Conductance of Strong Electrolytes
- Molar Conductance of Weak Electrolytes
- Practice Problems
- Frequently Asked Questions
Conductance (G)
It represents the ease by which current can flow through the conductor.
It is a measure of degree through which conductor can conduct electricity.
Greater the value of conductance, greater the conduction.
It is generally inverse of resistance.
Mathematically we can write,
Unit of conductance is simen (S)
Conductivity/ Specific Conductance
The conductance of a solution with a length of 1 cm and a cross-sectional area of 1 sq cm is used to determine a solution's conductivity. The opposite of resistivity() is conductivity, or specific conductance. It is represented by the symbol "K".
Mathematically
= conductance of 1 (unit)3 of conductors or 1 (unit)3of solution.
l= distance between electrodes
A= area of cross section of electrodes
S.I unit of K
Common unit :
Molar Conductivity
It is the conductivity of 1 mole of solution of electrolyte dissolved in V volume of solution.
As we know
Molarity =
Since n=1
Hence,
If the unit of is in Sm-1
If the unit of is in S cm-1
S.I unit of molar conductivity
Common unit of molar conductivity
Limiting molar conductivity
- Limiting molar conductivity is the constant conductivity of one mole at infinitely dilute solution. In other words, the molar conductivity is known as limiting molar conductivity when the concentration of the electrolyte approaches zero.
- Limiting molar conductivity is designated as
Equivalent Conductivity
Conductance of 1g eq. of conductors/electrolyte dissolved in V volume of solution,
1g eq. = amount of substance deposited/liberated by 1 mole of electrons/ 1F
As we know,
Normality
Hence,
Strong Electrolytes
- Strong electrolytes are substances that totally dissociate into ions. Sodium chloride (NaCl), potassium chloride (KCl), lead bromide (PbBr2), sodium hydroxide (Na(OH)2), potassium hydroxide (KOH), hydrochloric acid (HCl), nitric acid (HNO3), sulfuric acid (H2SO4), and others are some examples.
- Only ions and no electrolyte molecules will be present in the solution. Strong electrolytes are excellent electrical conductors, but only in aqueous solutions or molten form.
- To gauge the electrolyte's relative strength, use a galvanic cell. The strength of the electrolyte has a direct relationship to the voltage generated.
- Salts, strong acids, and bases, on the other hand, are strong electrolytes. The strong electrolyte ionisation can be witten as follows:
Weak Electrolytes
- Weak electrolytes are poorly ionised in aqueous solution. The solution will contain more of unionised molecules and less of ions.of the electrolyte. Ionisation of weak electrolytes are around 1% to 10%,while that of strong electrolytes are almost 100%.
- Examples of weak electrolytes are acetic acid, carbonic acid and ammonia.. They have low electrical conductivity. The ionisation of the week electrolytes leads to an equilibrium between the ionised and ionised molecules.represented by a double sided arrow. The ionisation of week electrolyte is represented as:
Conductance of Strong Electrolytes
According to the following equation, the molar conductivity of strong electrolytes varies with concentration:
where A is a constant depending upon the type of the electrolyte, the nature of the solvent and the temperature, and is the molar conductivity at infinite dilution, called limiting molar conductivity. This equation is applicable at lower solute concentrations and known as Debye Huckel-Onsager equation. Thus, if is plotted against sqrt(c) a linear graph is obtained for low concentrations (with slope = - A) but it is not linear for higher concentrations.
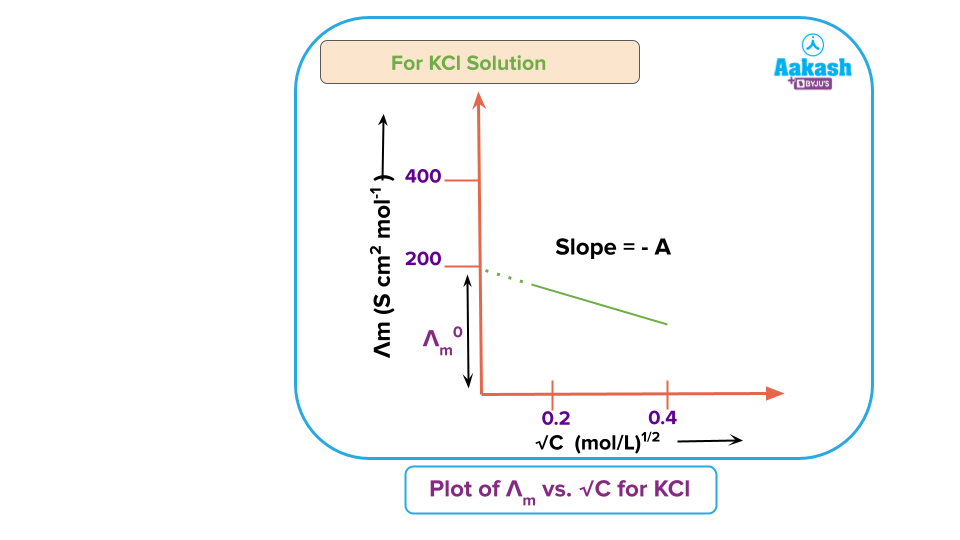
Further, the curve obtained for a strong electrolyte shows that there is only a small increase in conductance with dilution. This is due to the fact that a strong electrolyte completely dissociates in solution, maintaining a constant ion concentration. The greater inter-ionic attractions at higher concentrations cause the ions to move more slowly, which causes the conductance to decrease as concentrations increases. With decrease in concentration, with dilution, the ions are far apart and, therefore, the interionic attractions decrease due to which the conductance increases with dilution and approaches a maximum limiting value at infinite dilution, designated as
Conductance behaviour of Weak Electrolytes
As weak electrolyte, dissociates to a much lesser extent as compared to a strong electrolyte, for the same concentration, the conductance of a weak electrolyte will be substantially lower than that of a strong electrolyte.
Additionally, the curve created for a weak electrolyte demonstrates that conductance increases significantly with dilution, particularly when it approaches infinite dilution. This is because at low concentrations, weak electrolyte is more ionized. On dilution,increase in the number of ions increases the conductance of the solution. But it never reaches a limiting value.
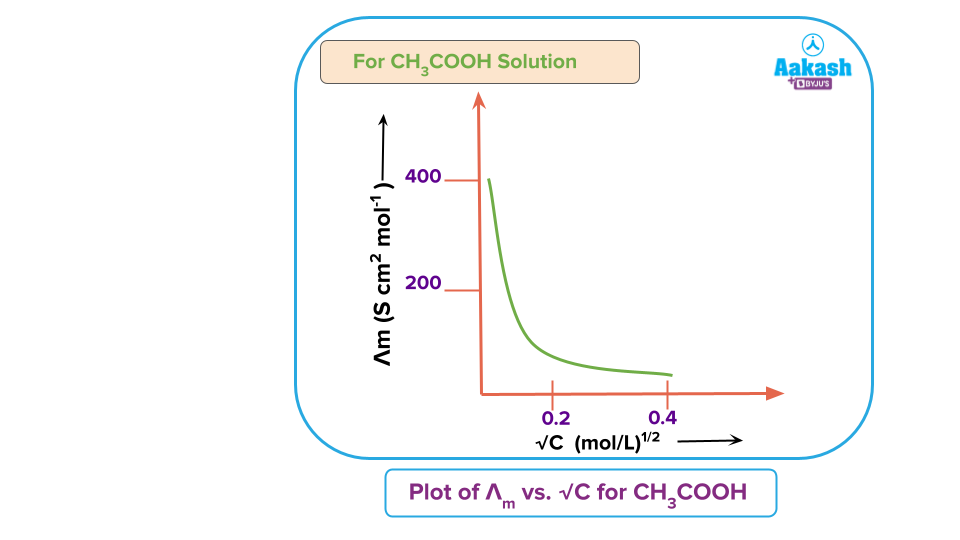
Further, from the plots shown above it is observed that the plot for a strong electrolyte becomes linear near high dilutions and thus can be extrapolated to zero concentration to get the value of the molar conductivity at infinite dilution, i.e., However, for a weak electrolyte increases steeply on dilution, especially near low concentrations. Hence, an extrapolation to zero concentration is not possible, as is clear from the plot for acetic acid shown above Experimentally also, it is not possible to determine the value for a weak electrolyte because though the dissociation is complete, the concentration of ions per unit volume is so low that the conductivity cannot be measured accurately. The problem was finally solved by Kohlrausch who put forward a law known as Kohlrausch law.
Kohlrausch Law
Definition:
“The limiting molar conductivity of an electrolyte (i.e. molar conductivity at infinite dilution) is the sum of the limiting ionic conductivities of all cations and all anions present in one formula unit of the electrolyte.”
Mathematically,
for
where is the limiting molar conductivity of the electrolyte.
are the limiting molar conductivities of the cation (Ay+) and the anion (Bx-) respectively.
For example,
for
Practice Problems
Q.1 What is the relation between the concentration of solution and the solution’s specific conductivity?
Answer: The specific conductance is the conductivity of a solution present within a length of 1 cm and a cross-sectional area of 1 sq cm. Higher the concentration, higher the number of ions present and higher the specific conductivity. Hence the specific conductivity is directly proportional to the concentrzation of the solution..
Q.2 In aqueous solution cupric ions are more stable than cuprous ions. Give reasons.
Answer: In aqueous solutions, ions are generally hydrated. Higher the charges and smaller the ions higher the hydration energy released amd more stable the ions. Cupric ion is smaller and higher charge than cuprous ion. Unstable curious ion disproprtionates to cupric ions and neutral copper.
Q.3 Debye Huckel- onsager equation is
B.
C.
D.
Answer: (A)
Solution: The molar conductivity of strong electrolytes is found to vary with concentration according to the equation:
where A is a constant depending upon the type of the electrolyte, the nature of the solvent and the temperature, and m° is the molar conductivity at infinite dilution, called limiting molar conductivity. This equation is applicable at lower solute concentrations and known as Debye Huckel-Onsager equation.
Q.4 Unit of molar conductivity is
- S cm mol-1
- S cm2 mol2
- S cm2 mol-2
- S cm2 mol-1
Answer: (D)
Solution:
If the unit of K is in Sm-1
If the unit of K is in S cm-1
S.I unit of molar conductivity
Common unit of molar conductivity
Frequently asked questions
Q.1 Is molar conductivity an intensive property?
Answer: Because molar conductivity is independent of electrolyte concentration and mass, it is an intensive property.
Q.2 What impact does temperature have on the molar conductivity?
Answer: With the increase in the temperature, the molar conductivity of an electrolyte increases with an increase in the interaction of the ions.
Q.3 Does volume affect conductivity?
Answer: In short, molar conductivity does not depend on the volume of the solution.
Q.4. Does concentration affect the molar conductivity?
Answer: Yes, concentration of the solution is an important factor factor in determining the value of molar conductivity. When solution is diluted, it’s molar conductivity increases. The total number of ions increases when a solution is diluted because the degree of dissociation increases. Molar conductance subsequently increases with dilution.


