-
Call Now
1800-102-2727
Third Law of Thermodynamics – Statement, Alternate Statements, Residual Entropy and Mathematical Explanation
The Antarctic is breathtakingly beautiful. Even the thickest winter coats can't stop the biting cold from penetrating through all that beauty.
Even in these extreme conditions, a well-known resident thrives. Any suggestions as to which animal I am referring to?
The magnificent Emperor penguins, indeed.
These kinds of birds have developed a tactic called huddling to combat the extreme cold of the Antarctic, where temperatures drop to and the wind chill makes it even lower!
These penguins form tightly packed huddles and reduce their movements to the bare minimum. And all of them face in the same direction to ensure the tightest packing.
And as the temperature plummets, they become almost still like statues. The cold winds do not then manage to penetrate to reach all of them.
This is how they fight and survive the extreme cold through the bitterest of the Antarctic winters.
There is something strikingly parallel in the world of thermodynamics.
We know that when we increase the temperature, the average kinetic energy of the atoms or molecules increases, and when we lower the temperature, the average kinetic energy decreases.
But what if we keep on decreasing the temperature to a certain point where the atoms or molecules completely stop?
That temperature in thermodynamics is known as ‘Absolute Zero’, and it’s the lowest possible temperature. It is equal to .
And just like our penguins, at a temperature of 0 K, most atoms and molecules get arranged in a definite, symmetrical pattern, just like in a perfect crystal and do not move around.
We already know that when the matter is in a state of maximum order, it will have the least randomness or entropy.
So, we can say that the entropy of a perfect crystalline substance approaches zero when the temperature of the crystal approaches 0 K.
From this observation, the third law was developed by chemist Walther Nernst during the years 1906–12, and is therefore often referred to as the Nernst's theorem or Nernst's postulate. Let’s discuss it in detail !!
TABLE OF CONTENTS
- Entropy
- Third Law of Thermodynamics
- Residual Entropy
- Alternate Statements of the Third Law of Thermodynamics
- Why is it Impossible to Achieve a Temperature of Zero Kelvin?
- Absolute Entropy of a Pure Substance
- Standard Entropy Changes in Chemical Reactions
- Practice Problems
- Frequently Asked Questions – FAQ
Entropy
The disorder or the degree of randomness of a system can be expressed by a thermodynamic term called Entropy. In other words, entropy can be defined as the measure of randomness or disorder in a system,
Entropy is denoted by the symbol, S. Entropy is a state function and an extensive property. The change in entropy can be denoted as ΔS. The change in enthalpy can be calculated using the formula,
The entropy change of a system can be defined as the integral of all the terms involved in heat exchange (q) divided by the absolute temperature (T) during the infinitesimally small change of the process carried out reversibly at a constant temperature.
ΔS=
ΔS is positive (entropy increases) if heat is absorbed, and negative (entropy decreases) when heat is evolved. ΔS depends on the initial and final states of the system, and is independent of the way in which the change is brought about.
As entropy is expressed as heat divided by temperature, the unit of entropy is calories per degree. In SI units, entropy is expressed as Joules per Kelvin (J K-1). As entropy is an extensive property i.e. it depends on the mass of the substance, its unit can be expressed as J K-1 mol-1.
Third Law of Thermodynamics
The third law of thermodynamics deals with the entropies of perfectly crystalline solids at absolute zero of temperature.
According to the third law of thermodynamics,
“As the temperature gets closer to absolute zero, the entropy of any pure crystalline substance decreases until it is zero.”
This is known as the third law of thermodynamics, i.e., in a pure crystalline compound at absolute zero temperature, there is a state of perfect order.
Residual Entropy
Since entropy is related to the disorder, according to the third law of thermodynamics, at absolute zero, there is a perfect order. As a result, the system's entropy should be zero, but this is only true for pure crystalline solids due to theoretical justifications and real-world evidence that solutions and supercooled liquids do not have zero entropy at absolute zero (0 K).
The entropy possessed by certain substances at 0 K is known as residual entropy. It happens because of the random orientation of any of the molecules in a packed structure. The difference in entropy between a substance's non-equilibrium state and crystal state close to absolute zero is known as residual entropy.
From a thermodynamic point of view, we have chosen the value zero as per our convenience. However, the molecular interpretation of entropy justifies the value of S=0 at 0 K. Therefore, it allows us to define the absolute entropies of substances and to calculate the standard entropy change of a reaction.
Alternate Statements of the Third Law of Thermodynamics
The Nernst statement of the third law of thermodynamics implies that
“It is not possible for a process to bring the entropy of a given system to zero in a set number of operations.”
The American physical chemists Merle Randall and Gilbert Lewis stated this law differently.
“When the entropy of each and every element (in their perfectly crystalline states) is taken as 0 at absolute zero temperature, the entropy of every substance must have a positive, finite value. However, the entropy at absolute zero can be equal to zero, as is the case when a perfect crystal is considered.”
The Nernst-Simon statement of the third law of thermodynamics can be written as
“The related entropy change approaches zero as the associated temperature approaches zero for a condensed system going through a reversible isothermal process.”
Another implication of the third law of thermodynamics is
“The exchange of energy between two thermodynamic systems (whose composite constitutes an isolated system) is bounded.”
Why is it Impossible to Achieve a Temperature of Zero Kelvin?
For an isentropic process (a thermodynamic process in which the entropy remains constant) that reduces the temperature of some substance by modifying some parameter X to bring about a change from 'X2' to 'X1', an infinite number of steps must be performed in order to cool the substance to 0 K.
This is because the third law of thermodynamics states that the entropy change at absolute zero temperatures is zero. The entropy v/s temperature graph for any isentropic process attempting to cool a substance to absolute zero is illustrated below.
From the graph, it can be observed that – the lower the temperature associated with the substance, the greater the number of steps required to cool the substance further. As the temperature approaches zero kelvin, the number of steps required to cool the substance further approaches infinity.
Absolute Entropy of a Pure Substance
For each pure substance, the absolute value of entropy can be computed at any given temperature, in contrast to the absolute value of enthalpy.
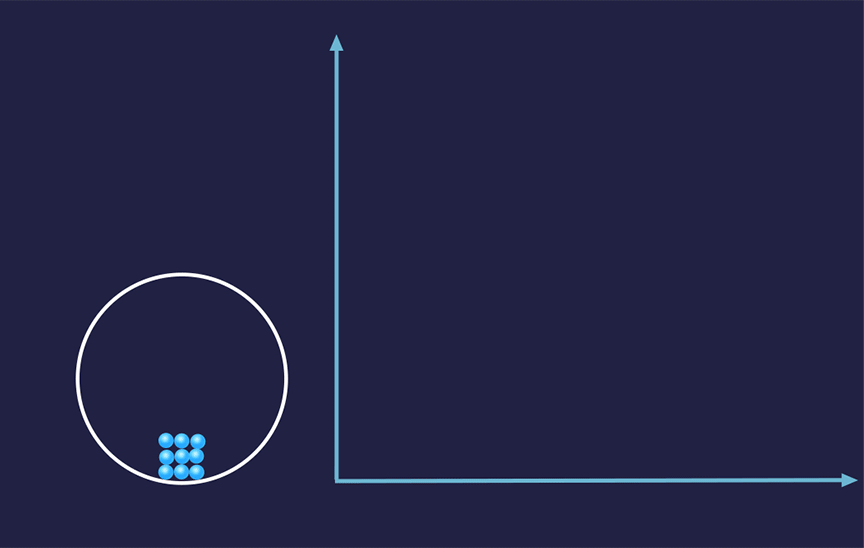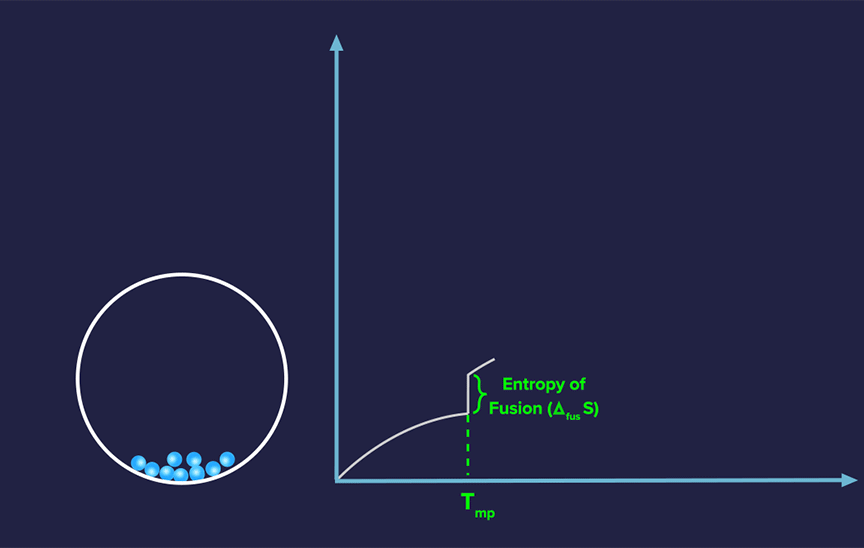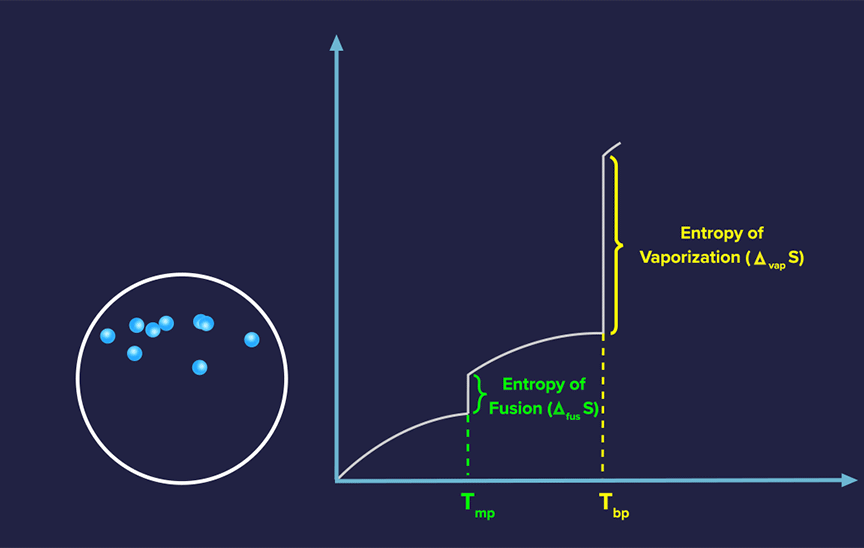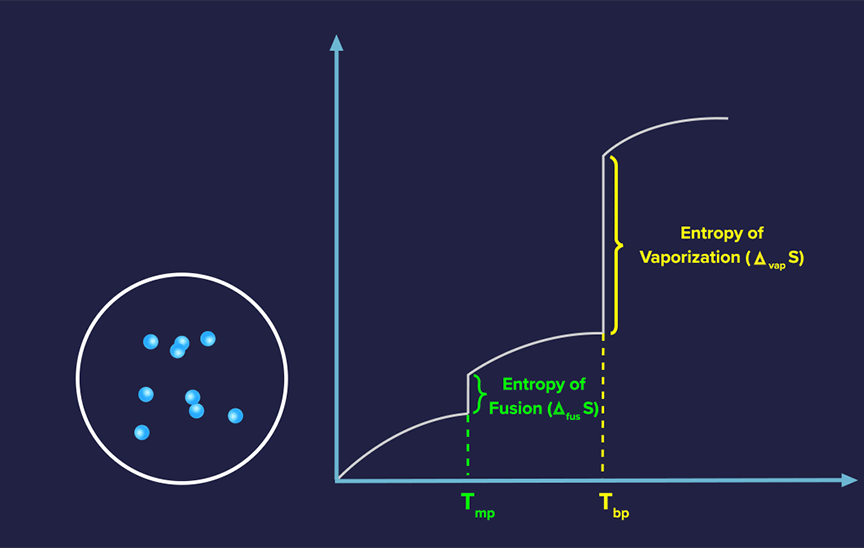
Since randomness increases more at lower temperatures, the slope often decreases as temperature increases for solids.
Standard Entropy Changes in Chemical Reactions
Standard Molar Entropy
Applications of the Third Law of Thermodynamics
- It is used to calculate the absolute entropies of solids, liquids and gases at different temperatures.
- It is used to calculate the entropy changes in a chemical or a physical process.
Limitations of the Third Law of Thermodynamics
- Glassy solids have an entropy greater than zero even at 0 K.
- Zero entropy at 0 K is not observed for solids having a mixture of isotopes. For example, the entropy of solid chlorine at 0 K is not zero.
- The crystals of Co, H2O, NO, NO2, etc do not have perfect order at even 0 K. Therefore, their entropy is not zero at 0 K.
Practice Problems
Q1. When 1 mol of H2O(l) is formed under standard conditions, calculate the entropy change (JK-1) in the surroundings (at 300 K). (Assume that =
Answer: B
Solution: Since at a phase transition, the process is reversible,
So, option B is the correct answer.
Q2. Because the direct translation from A to B is difficult, the following path is followed:
ΔS (A⟶C) = 50 JK-1
ΔS (C⟶D) = 30 JK-1
ΔS (B⟶D) = 20 JK-1
Find the entropy change for the process A⟶B.
Solution: Since entropy is a state function, ΔS (A⟶B)=ΔS (A⟶C)+ΔS (C⟶D)+ΔS (D⟶B)
Since ΔS (D⟶B)=-ΔS (B⟶D), ΔS (A⟶B)=ΔS (A⟶C)+ΔS (C⟶D)-ΔS (B⟶D)
Hence, the entropy change for the process A⟶B is 60 JK-1.
Q3. Calculate the entropy change (JK-1) that occurs when 3 mol of an ideal gas expands from a volume of 100 dm3 to a volume of 1000 dm3 at 300 K in an isothermal reversible expansion.
Answer: C
Solution: The entropy change of a system is given as
As (Isothermal expansion)
So, option C is the correct answer.
Q4. At 250 K, one mole of an ideal gas in thermal contact with the environment expands isothermally from 1 L to 3 L under the constant pressure of 5 atm. Find the change in the entropy of the surroundings ( in JK-1 during this phase. (1 L atm=101.3 J)
- 5.763 JK-1
- 0.405 JK-1
- −1.013 JK-1
- −5.763 JK-1
Answer: B
Solution: The change in entropy of the surrounding is given by,
For an isothermal process
The work done in an isothermal process =
So, option B is the correct answer.
Frequently Asked Questions – FAQ
Q1. Is it possible to have a negative value of entropy?
Answer: The measure of disorder in a system is entropy. Entropy is always rising as everything in the universe strives toward a more chaotic state. The entropy of the cosmos for spontaneous events is always rising, according to the second law of thermodynamics. As a result, the total entropy can never be negative.
Q2. Is it possible to eliminate entropy?
Answer: Entropy is created everywhere and constantly, at every scale, and it can not be removed by any means at any scale. The Second Law of Thermodynamics states that "entropy of an isolated, closed system (or cosmos) is always rising." This is a required but not sufficient condition.
Q3. Is entropy increased by freezing?
Answer: Entropy favours melting because water has higher entropy than ice. Freezing is an exothermic process, which means that energy is lost from the water and dissipated in the environment. As a result, as the environment becomes hotter, it gains more energy, increasing the entropy of the environment.
Q4. What is the significance of the third Law of Thermodynamics?
Answer: The third law of thermodynamics has two important consequences. It defines the sign of the entropy of any substance at temperatures above absolute zero as positive, and it provides a fixed reference point that allows us to measure the absolute entropy of any substance at any temperature.


