-
Call Now
1800-102-2727
Structure of Acid Derivatives: Introduction of Acid Derivatives, Structure and Nomenclature of Acid Derivatives, Reactivity of Acid Derivatives, Practice Problems & Frequently Asked Questions(FAQs)
Who is your favourite actor? You may have many reasons for your liking. Everyone will have their own favourites.

But, there are only a few actors who are celebrated as good actors. Can you say what are the qualities of a good actor? It is simple. Any actor who identifies himself /herself in the different role they play, making us forget about the actor but to identify them with the character they play is a versatile actor.
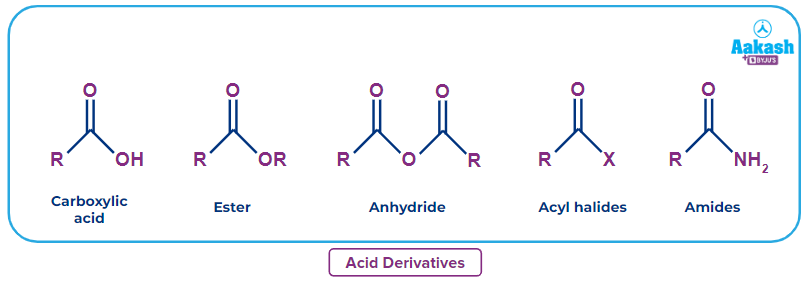
Likewise there are some organic compounds or groups that can change a little bit and exhibit totally different properties. One such group is carboxylic acid. Carboxylic acid can form with a small change to an anhydride, ester, carbonyl chloride, amide, etc. these are called acid derivatives as they can be prepared from the carboxylic acid functional group.
The family of compounds we will be studying is known as carboxylic acid derivatives.
Table of Contents:
- Introduction of Acid Derivatives
- Structure and Nomenclature of Acid Derivatives
- Reactivity of Acid Derivatives
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of acid derivatives:
Alkyl and/or aryl groups are linked to hydroxyl, alkoxy, amino, and halo substituents, respectively, to form the significant families of organic compounds known as alcohols, phenols, ethers, amines, and halides.
Carboxylic acids are a type of chemical molecules with the functional group -COOH and are used in a wide variety of goods, including hormones and medications that are synthesised. Carboxylic acids have an acyl group (R-C=O) with a hydroxyl group (-OH) attached to the carboxyl carbon. Functional derivatives are created by replacing the hydroxyl group with substituents such as -NH2, -X, -C ≡ N,–OOCR .
By switching out the -OH group in the carboxylic acid's structure, several carboxylic acid derivatives are created. The typical formula for carboxylic acids is R-COOH, where R is an alkyl group. The following acid derivatives are examples of typical -OH group replacements:
- When the -OH group is substituted with a halide group (-X), acyl halides are produced.
- Amides are created by substituting an amino group (-NH2) for the -OH group.
- When the -OH group is replaced by a -OR group, esters are produced.
- Since amide can be degraded, nitriles (R-C ≡ N) are also an acid derivative.
- When a –OOCR group is added in place of the -OH group, acid anhydrides and other compounds are created.
- Thioesters are designated as per IUPAC guidelines, with the carboxylic acid ending in -oic acid replaced by -thioate and the sulphide alkyl chain acting as a substituent.
Structure and Nomenclature of Acid and Acid Derivatives:
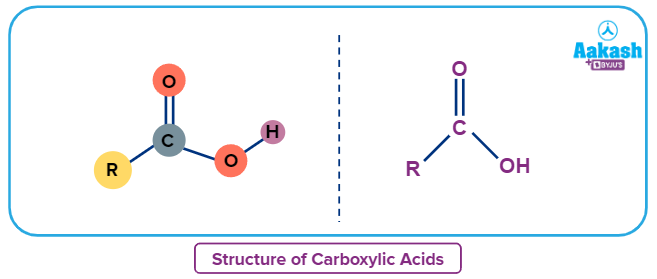
The general formula for carboxylic acids is R-COOH, where R stands for the rest of the molecule to which the carboxyl group is connected and -COOH represents the carboxyl group. A single sigma bond connects the carbon atom in this carboxyl group to the hydroxyl group, and a double bond connects it to the oxygen atom. CnH2nO2 is the general formula.
Below are a few examples of acid derivatives along with their structures.
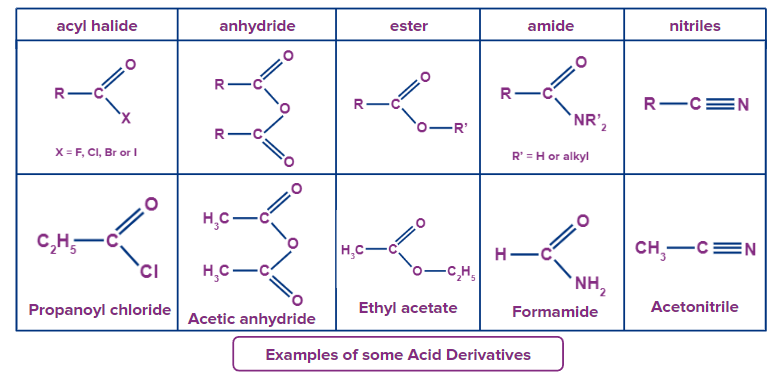
Acid Halide:
The name of the relevant carboxylic acid comes first in the nomenclature of acid halides. The -ic acid ending is dropped in favour of the ending -yl, which is then followed by the name of the halogen and a -ide. Both common and IUPAC terminology are valid in this regard. The first position number is assigned to the carbonyl carbon. Since it is believed that the functional group will be at the end of the parent chain, the location number need not be included in the name.
Example: 1. CH3COCl - Ethanoyl chloride
2. CH3CH2COCl - Propanoyl chloride (or propionyl chloride).
Acid anhydride:
A functional group is known as an acid anhydride when two carboxylic acids combine and lose water (anhydride implies without water). Symmetrical acid anhydrides are termed similarly to carboxylic acids, with the distinction that the suffix -anhydride is used in place of the ending -acid. This is true for both the IUPAC and the Common nomenclature.
Use the alkanoic general technique to name both unsymmetrical acid anhydrides, and then arrange the names alphabetically. As a result, give each component a name before organising it alphabetically, followed by spaces, and finally the word anhydride.
Example: CH3(CH2)2CO2O - Butanoic anhydride is a symmetrical anhydride
(CH3COOCOCH2CH3) - Ethanoic propanoic anhydride (or acetic propionic anhydride) is an unsymmetrical anhydride.
Esters:
The alkyl chain from the alcohol is referred to as a substituent when naming esters. This alkyl chain doesn't have a designated number. This is followed by the name of the parent chain, which consists of the carboxylic acid portion of the ester with the ending -e removed and replaced with the ending -oate.
Example: CH3(CH2)2CO2C2H5 - Ethyl butanoate (or ethyl butyrate).
Lactones are cyclic esters. The position of the alkyl oxygen in relation to the carboxyl carbonyl group is indicated by a Greek letter.
Amides:
Primary Amides:
In order to name primary amides, the name of the acid is modified by deleting the -oic acid or -ic acid ends and adding -amide. The position number for the carbonyl carbon is 1. Assuming that the functional group will be at the end of the parent chain, the location number is not required to be included in the name.
Example: CH3CONH2 - Ethanamide (or acetamide)
HCONH2 - Methanamide (or formamide)
C6H5CONH2 - Benzamide
Secondary and Tertiary amides:
When designating secondary amides, a capital letter N is used to indicate that the alkyl group is located on the nitrogen atom. The term "substituent" refers to alkyl groups that are joined to the nitrogen. They are identified as being connected to the nitrogen by the letter N. The names of tertiary amides are similar.
Examples: CH3(CH2)2CONHC2H5 - N-ethylbutanamide, (CH3)2NCOH - N,N-dimethylmethanamide (or N,N-dimethylformamide).
Lactams are the name for the cyclic amides. The position of the nitrogen on the alkyl chain in relation to the carboxyl carbonyl group is indicated by a Greek letter.
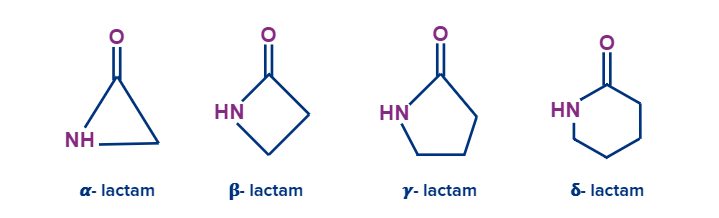
Nitriles:
The suffix nitrile is attached to the name of the appropriate alkane to create the names of simple acyclic nitriles (same number of carbon atoms). The first carbon in the chain to be numbered is the nitrile carbon. In place of the oic acid or ic acid ending of an analogous carboxylic acid, nitrile is frequently used. A nitrile substituent, such as one found on a ring, is known as carbonitrile.
Example: CH3CN- Ethanenitrile
CH3CH2CN- Propanenitrile
CH3CH2CH2CN - Butanenitrile
Thioesters:
According to their names, thioesters are substitutes for the alcohol's alkyl chain. This alkyl chain has not been given a number. After that, an alkane with the ending -thiooate is used to identify the parent chain from the carboxylic acid portion of the thioester.
Example: CH3 COS CH3 - Methyl Ethanethiooate
CH3 CH2 COS CH3 CH2 - Ethyl Propanethiooate.
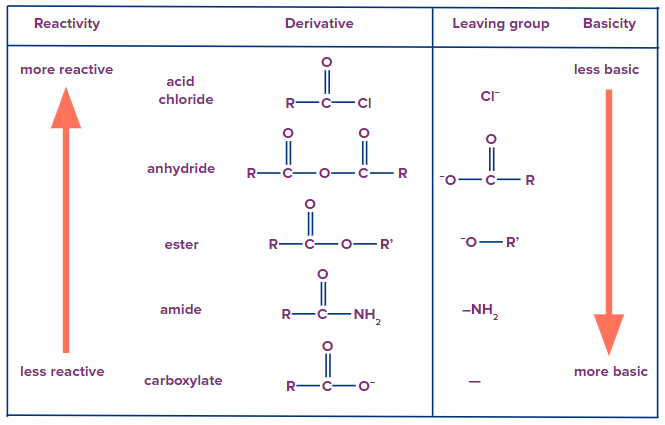
Reactivity of acid derivatives:
The anticipation of the compound's reactivity requires two steps in the synthesis of carboxylic acid derivatives, each of which is essential. Electronic and steric factors are important to take into account when contrasting the reactivities of carboxylic acids and their derivatives.
Bulky groups have a considerable impact on the pace of the first reaction during the synthesis of an acid derivative. It is more difficult for the nucleophile to attack the carbonyl carbon. More bulky the alkyl group linked less will be derivative reactivity. Evaluating reactivity requires taking steric parameters into account.
The polarisation of acyl molecules is important for predicting reactivities. Acyl compounds with greater degrees of polarisation react more quickly.
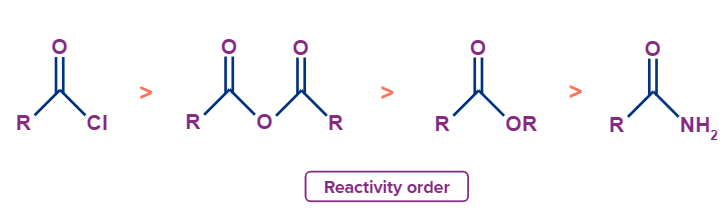
Bimolecular nucleophilic acyl substitution occurs on acid derivatives. A weaker base will function better as the leaving group and an acid derivative will be more reactive. Following is a list of the acid derivatives reactivity order to nucleophilic acyl substitution.
Practice Problems:
Q1. What is the IUPAC name of (CH3)2CHCH2CN?
(A) Ethanenitrile
(B) Butanenitrile
(C) Propanenitrile
(D) 3-methyl Butanenitrile
Answer: (D)
Solution: To produce the names of simple acyclic nitriles, the suffix nitrile is added to the name of the corresponding alkane (same number of carbon atoms). The nitrile carbon is the first carbon in the chain to be counted. Nitrile is frequently used in place of the oic acid or ic acid ending of an equivalent carboxylic acid. Carbonitrile is the name for a nitrile substituent, which is found on the ring and IUPAC name of the given compound is 3-methylbutanenitrile (or isovaleronitrile).
Q2. Derivatives of Carboxylic acids are
(A) Polar
(B) Non polar
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: Due to the polar nature of the carbonyl group and the presence of a heteroatom in the group , carboxylic acids and their derivatives are polar compounds.
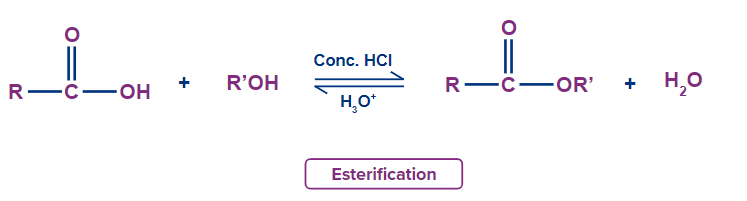
Q3. The process by which alcohol and carboxylic acid combine to form ester in the presence of acid is referred to as
(A) Esterification
(B) Alcoholysis
(C) Alkylation
(D) Acylation
Answer: (A)
Solution: In the presence of H+ ions, the esterification reaction between an alcohol and a carboxylic acid results in esters. Below is a general reaction for esterification.
Q4. Among the following, which has the highest boiling point?
(A) RCONH2
(B) RCOCl
(C) (RCO)2O
(D) RCOOH
Answer: (A)
Solution: Acid chlorides have the lowest boiling points due to the impossibility of hydrogen bonding.
Amides have high boiling points because they can form hydrogen bonds with up to two H atoms per amide group. The decreasing boiling point order is
RCONH2 >(RCO)2O >RCOOH > RCOCl
Frequently Asked Questions(FAQs):
Q1. Why does acyl chloride react more quickly?
Answer: Acyl chlorides are the derivatives of carboxylic acids that are most reactive. The electronegative chlorine atom in the C-Cl bond attracts electrons in its direction, making the carbonyl carbon more electrophilic. This makes a nucleophilic approach stronger. Furthermore, the chloride ion (Cl-) is an excellent leaving group, speeding up that process. Acyl chlorides are a crucial synthesis intermediate that are easily converted into esters and amides due to their reactivity.
Q2. Why do alcohols of equivalent molar mass have lower boiling temperatures than carboxylic acids?
Answer: Because two intermolecular hydrogen bonds can form between carboxylic acids, forming dimers. carboxylic acids have higher boiling temperatures than alcohols with comparable molecular weights.
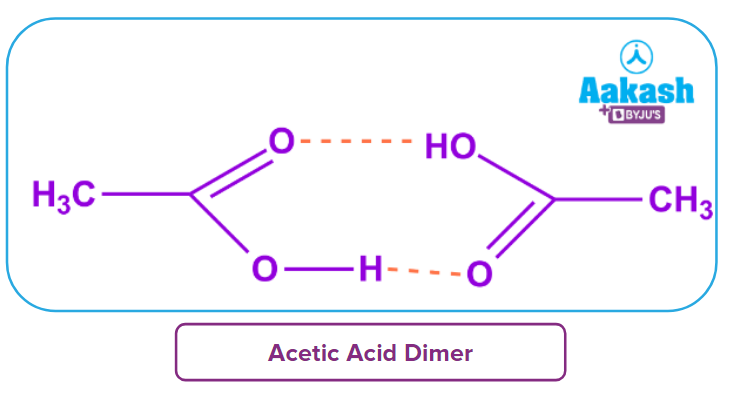
Additionally, it implies that low molecular weight carboxylic acids are water soluble. Higher molecular weight carboxylic acids are insoluble in water because when the carboxylic acid's molecular weight rises, the hydrophobic nature of the alkyl component eventually balances the polar nature of the functional group.
Q3. Why is Chloride ion a superior leaving group than hydroxide ion?
Answer: For a variety of reasons, Chloride ion is a superior leaving group to hydroxide ion. When compared to chloride ion, hydroxide ion is a superior Lewis base and has a higher nucleophilicity.
Compared to hydroxide ion,chloride ion is less electronegative. Since chloride ion is larger than hydroxide ion, its charge density is smaller than that of the hydroxide ion, making it more stable. As a result, it is easier for chloride to exist as a so-called free ion than for hydroxide, making it a superior leaving group.
Q4. Explain the concept of bonding in acid derivatives?
Answer: The inclusion of a group with an electronegative heteroatom, typically oxygen, nitrogen, or sulphur, bound directly to the carbonyl carbon distinguishes carboxylic acid derivatives from aldehydes and ketones. A derivative of a carboxylic acid can be thought of as having two components. The acyl group, which consists of the connected alkyl (R) group plus the carbonyl group is one component. The heteroatom-linked group is located on the other component. A strong bond and a weaker bonds make up the bonds in the carbonyl (CO) group. The carbonyl group is polarised so that the oxygen is slightly negative and the carbon is slightly positive because oxygen is more electronegative than carbon. Thus, carbon can behave as an electrophilic centre and oxygen as a nucleophilic centre.


