-
Call Now
1800-102-2727
State of a System - Thermodynamic Terms, State of a System, Difference Between State Functions and Path Functions, Practice Problems and FAQs
Do you travel a lot? Travelling distance has become easy and quicker with modern logistics.
Now travelling between cities can be taken by car, Vande Bharat trains or aircraft. All these logistics take different times between the same starting and destination points- aircrafts being the quickest and cars taking the longest time. What is the reason? You will say immediately that the different logistics take different routes of which the aircraft takes the shortest and most direct path between initial and final points, not talking about their speeds.


The distance they have to cover is the same, but the distances they traveled are different. The distance travelled by them depends on the path followed and is called a path function. The distance to travel will be the same which depends only on the starting and destinations (or initial state and final states) and not on the path and so it is called a state function.
Let us know more about these State and state functions,
TABLE OF CONTENTS
- Thermodynamic Terms
- Types of Systems
- State of the system
- Difference between State Function and Path functions
- List of state functions
- Practice Problems
- Frequently Asked Questions - FAQs
ThermodynamicTerms
You might be knowing the term ‘States within a country’ and physical states of matter. The state is also used frequently in thermodynamics, which involves a study of changes in thermodynamic properties like energy, work, etc of specific substances, upon temperature and work performances. You have to be familiar with and clear about the terms used in thermodynamics before we go any further.
System
A system is a part of the universe in which thermodynamic observations are being made. It could be solid or liquid or gas but separated from the rest of the Universe by clear-cut boundaries.
Surroundings
Anything other than the system under consideration of the Universe is surroundings. But for practical purposes, we take the immediate surrounding of the universe which influence the system.
Boundary
A boundary is a physical barrier that separates the system from its surroundings. A system is segregated from its surroundings by a real or imaginary wall.
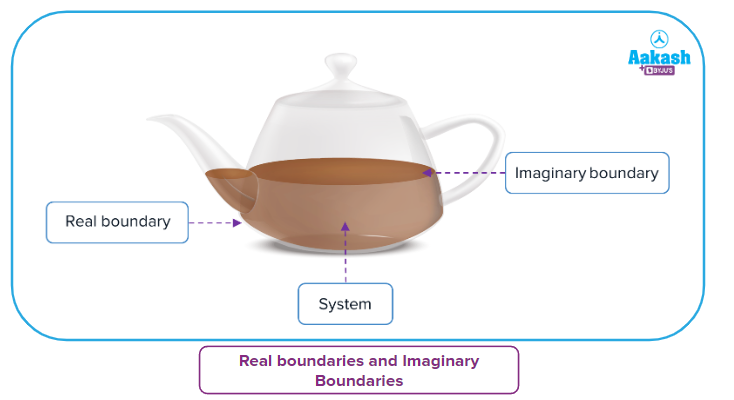
Tea in the vessel is defined as a system and everything else as surroundings. Here, the wall of the container which is separating the system and surroundings which exists, in reality, is known as a real boundary. Sometimes the tea along with the container also can be considered the system, if changes in both of them are taken into consideration.
When a small portion of tea (within the entire volume of the container marked in white colour) is defined as a system, the boundaries of this system are imaginary as it does not exist in reality and it can be changed if some amount of tea is poured out.


Universe
The system and its surroundings together make up the universe.
Types of Systems
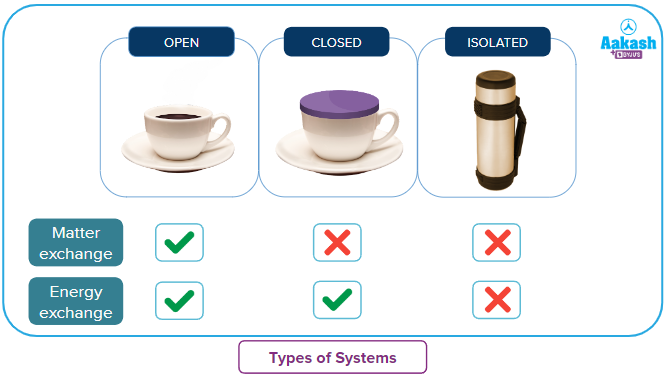
The system under study can be classified into as follows-
- Open System: The system that is open to the surroundings so that it can exchange its contents of mass or matter and energy with the universe or surroundings is referred to as an open system.
An open cup of tea loses both tea vapours and heat to the surroundings so that after some time there is a lesser amount of cold tea left in the cup.
- Closed System: As the name suggests, the system is closed for the exchange of its mass or matter. So, a closed system does not exchange its mass or matter but still, it can exchange energy with the surroundings or universe through the boundary.
A hot tea cup loses only heat with the surrounding, such that the quantity of tea remains the same while it becomes cold after some time.

- Isolated System: In an isolated system, the exchange of both mass and energy with the surroundings does not take place. The boundary is impermeable for both mass and heat.
Thermos flask is an example of an isolated system where the exchange of matter and energy will not take place.

To summarise this:
State of the System
Any matter is characterised in terms of its physical and chemical properties. The physical properties are fixed in terms of variables like pressure, volume, temperature and concentration as moles.
When the macroscopic properties of a system can be characterised by definite values of the variables, the system is said to be in a (definite) state. Whenever there is a change in any one of these macroscopic properties, the system becomes different and is said to be changed into a different state.
As a result, the system's state is determined by its macroscopic properties. The state of the system is the condition in which the system is present. The state is defined by measuring the observable properties of the system like pressure, volume, temperature, composition, etc.
The system before any change is known as the initial state and after the changes, it is in a final state. An initial state may change into a final state following different routes like logistics discussed earlier to reach the same final state.
- State functions: These are the physical quantities whose values depend only on the state of the system and do not depend upon the path by which this state has been attained. A few examples of state functions are pressure, volume, temperature, etc.
- Path functions: These are the physical quantities that change with the path by which the system has achieved a particular state. Heat and work are examples of path functions.

Here we can observe the path-dependent nature of work and the path-independent nature of potential energy. The man is going to the third floor using a lift and the woman by stairs, the work done while reaching the third floor is different for both but since they both arrive at the third floor. Whereas, the potential energy is the same for the man and the woman because both are gaining the same height.
Take another illustration of work as a path function is given in the figure below. Here, the work done by the athletes while running around the track will depend on the path followed by them, i.e., if the athlete chooses to run through another path (i.e., the trajectory of the athlete changes), then the work done by the athlete during running will also change.

Difference Between State Function And Path Function
State functions, as previously described, are attributes whose values are independent of the path taken to reach that function or value. Path functions are the functions that rely on the path followed to get to a given value.
|
State function |
Path function |
|
Independent of the path taken to reach the property or value |
Dependent on the path taken to establish the property or value |
|
Capable of integrating using initial and final values |
Requires multiple integrals and limits of integration in order to integrate |
|
Multiple paths result in the same value |
Different paths may result in different values |
|
Examples: Temperature (T), Pressure (P), Enthalpy (H), Internal energy (U) |
Examples: Heat (q), Work (W) |
List of State Functions
Pressure: Pressure is the average force with which the molecules hit the walls of the container that depends on the volume of the container, temperature and the concentration and not on the path of the molecules they take to hit the walls. So, pressure is a state function.
Temperature: Temperature is the degree of coldness and hotness of a body which does not depend on how the temperature is reached. So, the temperature is a state function.
Volume:
The volume of the container is the volume of the gas molecules that depends on the pressure, and temperature and hence is a state function.
Mass:
The amount or mass of a substance is a fixed quantity independent of pressure, volume and temperature. So, is a state function
Internal energy:
It is the total energy consisting of a system consisting of bond energies and energy due to motion. These are dependent on the state variable such as pressure, volume, and temperature and hence is a state function.
Gibb’s free energy:
Gibb's free energy of the system defines as G = H – TS, changes with H and S which are state functions and hence will be a state function.
Entropy:
Entropy is a measure of the randomness of the system. It is unique to the current state of the system, hence entropy is a state function.
Recommended Video Link: https://youtu.be/Xq0EipqWJJY
Practice Problems
Q1. A system wherein the exchange of energy and matter takes place with the environment is called:
a. Open System
b. Closed System
c. Isolated System
d. None of these
Answer: (A)
Solution: In thermodynamics, a closed system can exchange energy (as heat or work) but no matter, with its surroundings. An isolated system cannot exchange both energy and matter with the surroundings, while an open system can exchange both energy and matter with the surroundings.
Q2. The system where only energy can be exchanged with the surroundings and not the matter is called:
a. Open System
b. Closed System
c. Isolated System
d. None of these
Answer: (B)
Solution: In thermodynamics, a closed system can exchange energy (as heat or work) but no matter, with its surroundings. An isolated system cannot exchange any heat, work, or matter with the surroundings, while an open system can exchange both i.e. energy and matter.
Q3. Which of the following is a state function?
a. q w
b. q +w
c.
d. q -w
Answer: (B)
According to the first law of thermodynamics;
ΔU=q+w
As we know that ΔU (change in internal energy) is a state function, thus we can say that q+w is a state function.
Q4. Which is not a state function?
a. Enthalpy
b. Entropy
c. Work
d. Internal Energy
Answer: (C)
Solution: Heat and work are not state functions, they are path functions. Work cannot be a state function because it is proportional to the distance travelled by an object, which depends on the path used to get from one state to the next.
Frequently Asked Questions – FAQs
Q1. Is altitude a state function?
Answer: It doesn’t matter how you got to the top of the mountain, the change in altitude will be the same. This is an example of a state function, which is a property whose value is independent of the path used to get there.
Q2. Why entropy is a state function?
Answer: Entropy is a state function since it depends only on the initial and the final states of the system. It does not depend on the path, whichever path it takes to reach a final state from the initial state, the entropy value remains the same for both states and the change in entropy will be constant between the two states.
Q3. Why energy is a state function but heat and works not, even though both are one of the forms of energy?
Answer: Actually energy has two forms:
1. Energy in storage: Energy which can be stored. Examples: chemical energy, internal energy, kinetic energy, potential energy, pressure energy etc. All energies in storage are state functions.
2. Energy in transition: Energy which can not be stored but is in the transition state. Examples: heat, work, electrical energy etc. All energies in transition are not state functions, they are path functions.
Hence energy is a path function but heat and work are not.
Q4. Which of the following is a state function and an extensive property at the same time?
a. Internal energy
b. Pressure
c. Molar heat capacity
d. Temperature
Answer: (A)
Solution: Internal energy only depends upon the initial and final state of the system, not on the path followed. As a result, the internal energy function is a state function.
The amount of matter has an impact on internal energy. The internal energy increases as the amount of the substance increases, causing more collisions. So, internal energy is an extensive property.


