
Stability of Fully Filled and Half Filled Orbitals: Factors Affecting Stability, Examples, Practice Problems & FAQs
If you are going to Kota from Delhi, you have a few sitting options like chair car berth and sleeper berth. Which sitting berth is more comfortable for your journey.
We know a body having less energy is considered to be more stable. You feel relaxed with a sleeper berth in comparison to a sitting chair berth.

Table of contents
- Factors affecting the stability of fully and half-filled orbitals
- Exchange energy
- Symmetricity
- Practice problems
- Frequently asked questions-FAQs
Factors affecting the stability of fully and half-filled orbitals
The factors mainly affecting the stability of half-filled and fully filled orbitals are
- Exchange energy
- Symmetricity
Exchange energy
Electrons having the same spin and energy present in degenerate orbitals can exchange their positions and in this exchange process, the energy is released and the released energy is termed exchange energy.
The higher the number of exchanges in a particular configuration, the stability of the configuration becomes higher.
The exchange energy is the basis for Hund's rule, which allows maximum multiplicity, that is electron pairing is possible only when all the degenerate orbitals contain one electron each.
more the number of exchange ∝ more the stability of configuration
Where n is the total number of electrons having same energy and spin
r = 2 (minimum 2 electrons required for exchange)
Symmetricity
Nature likes symmetry. Symmetry leads to stability. Fulfilled and half-filled subshells are symmetrical. So, they are more stable.
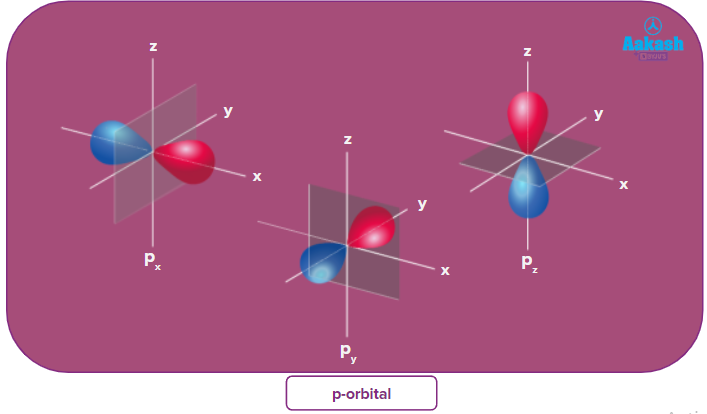
Half filled subshells:
![]()
![]()
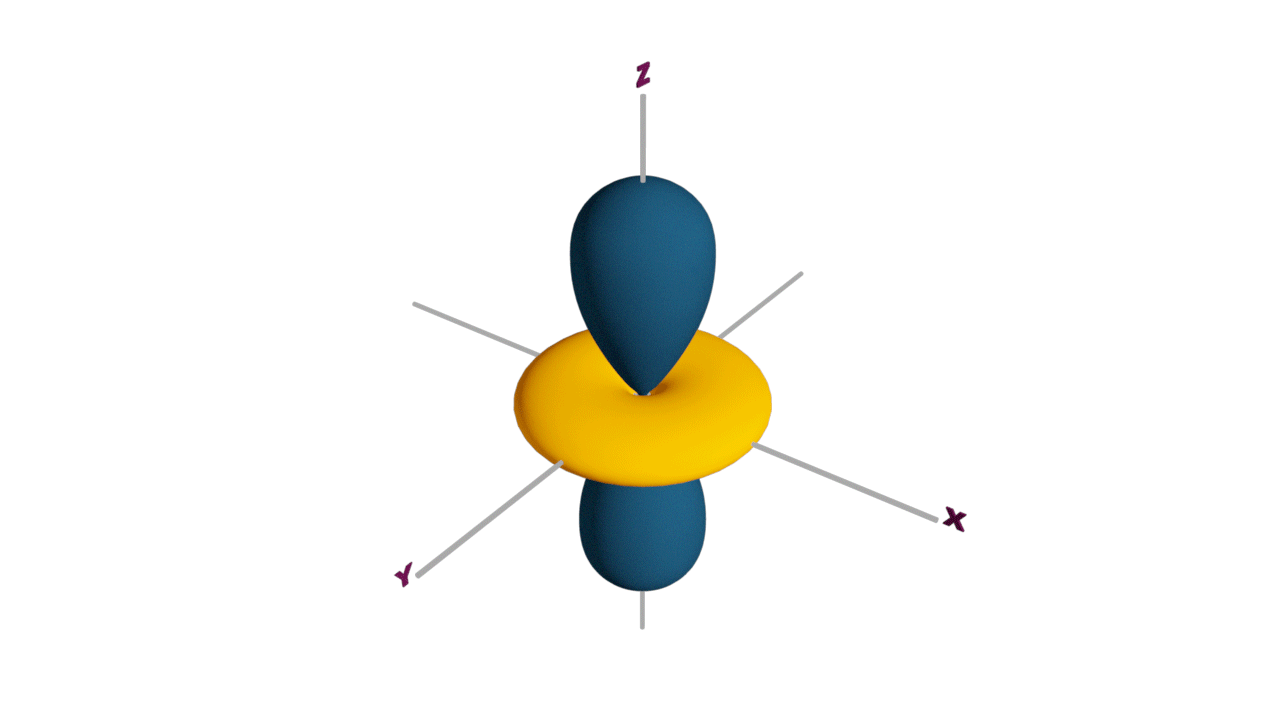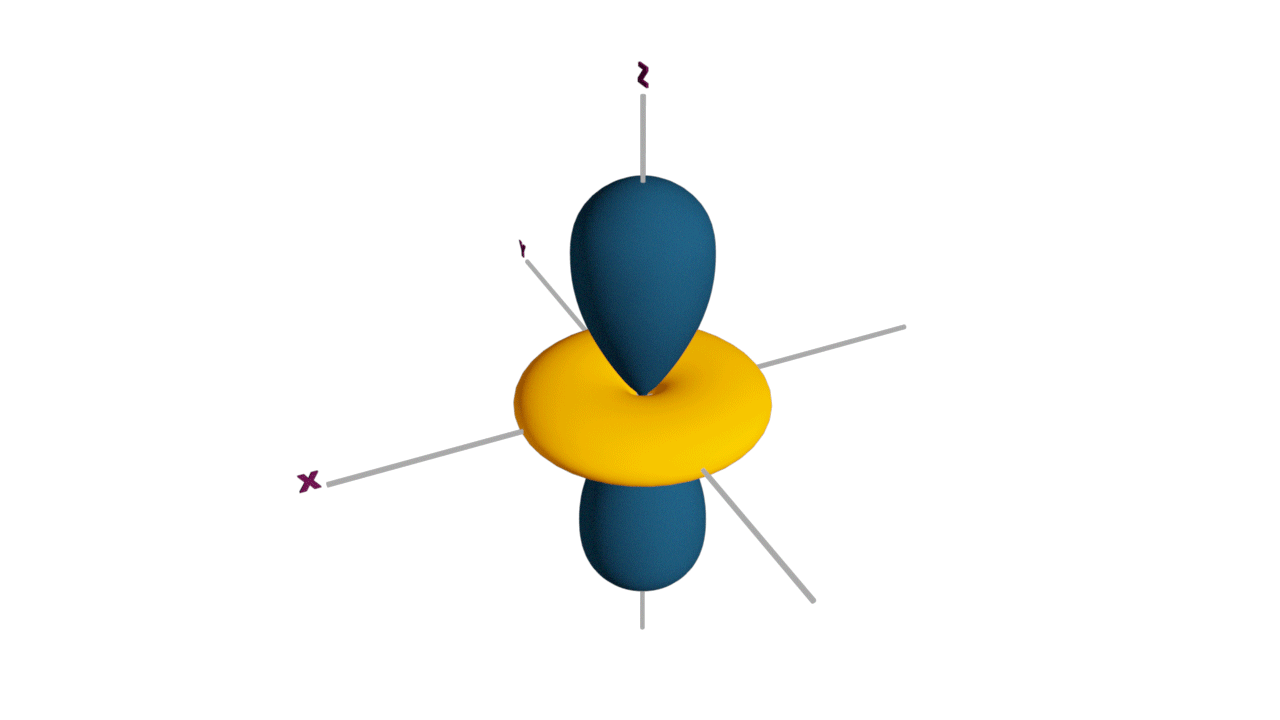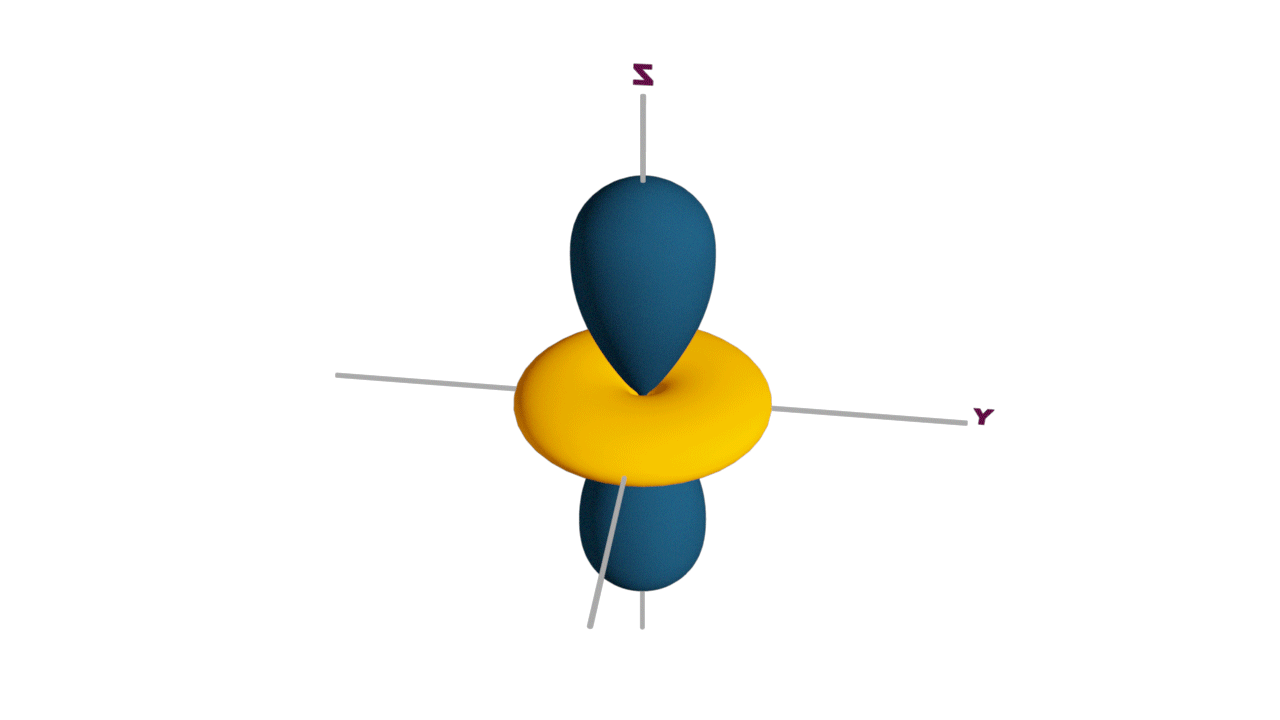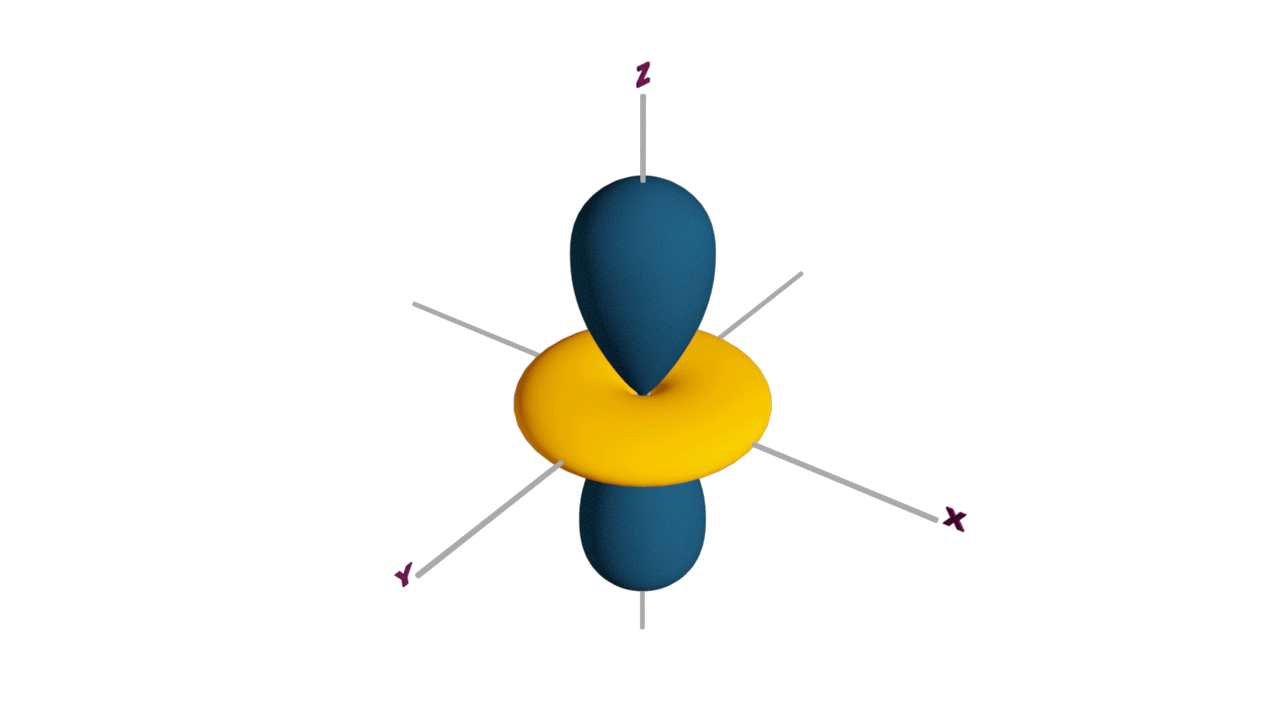
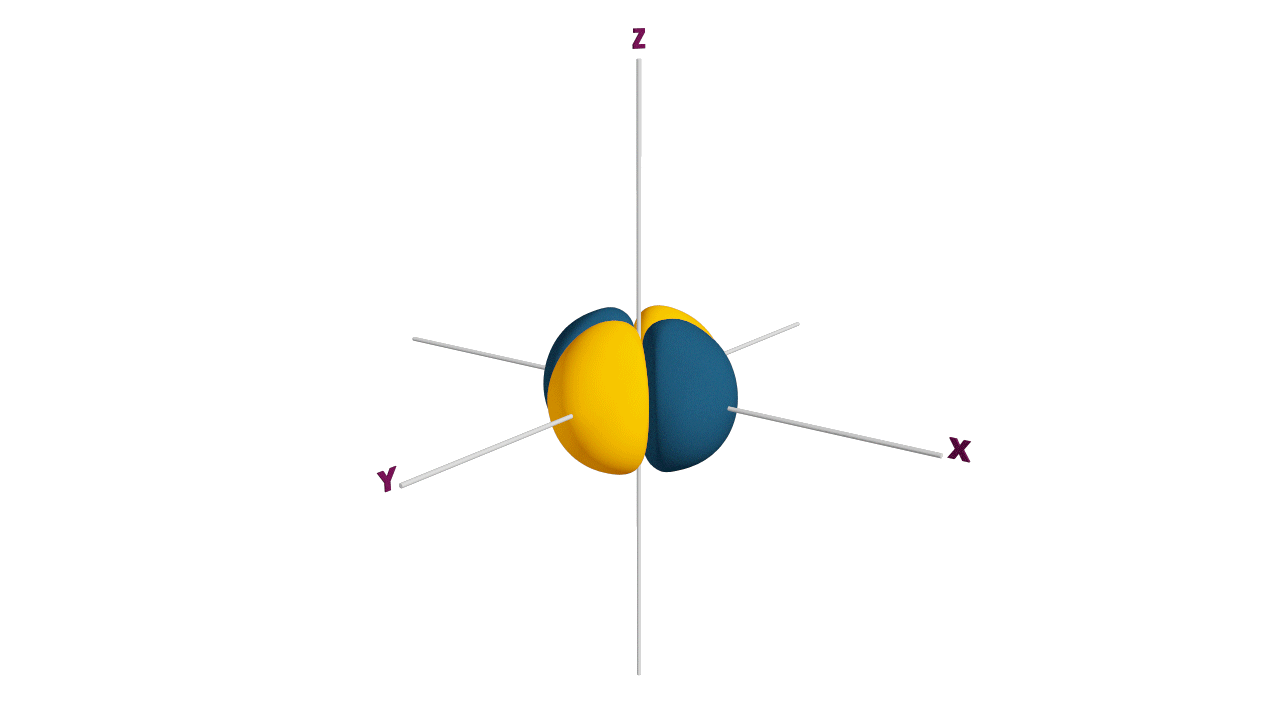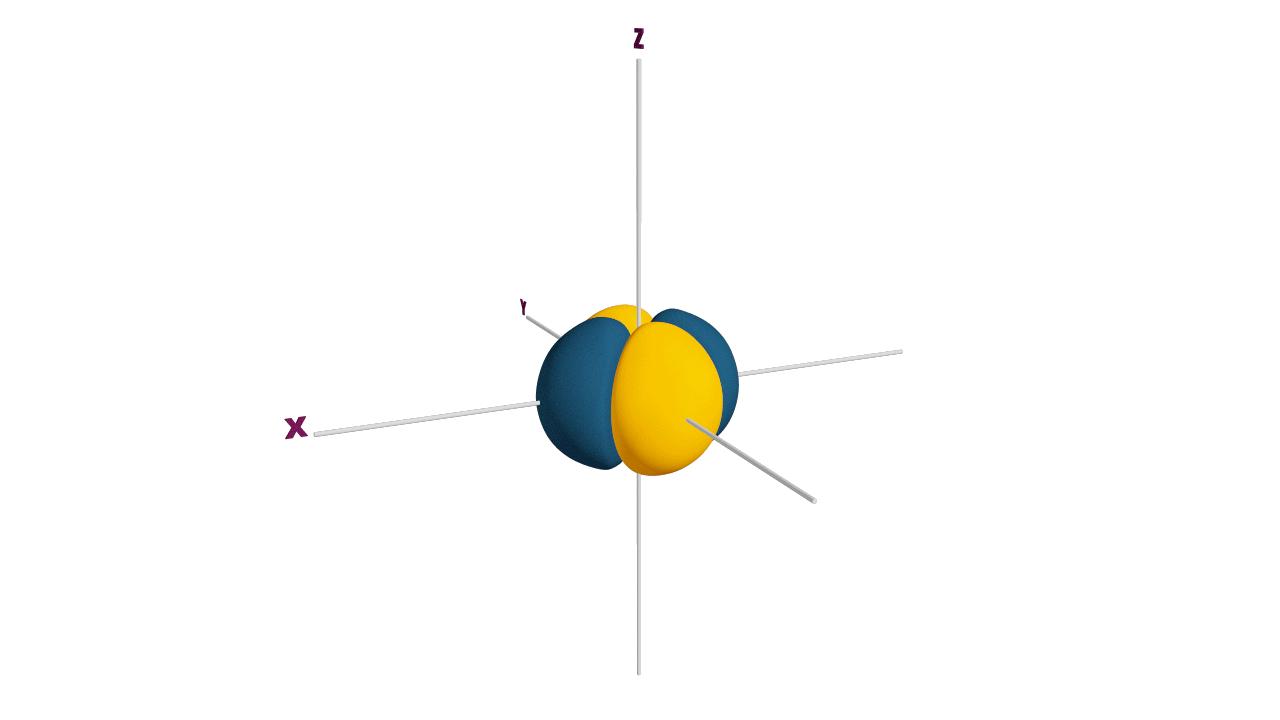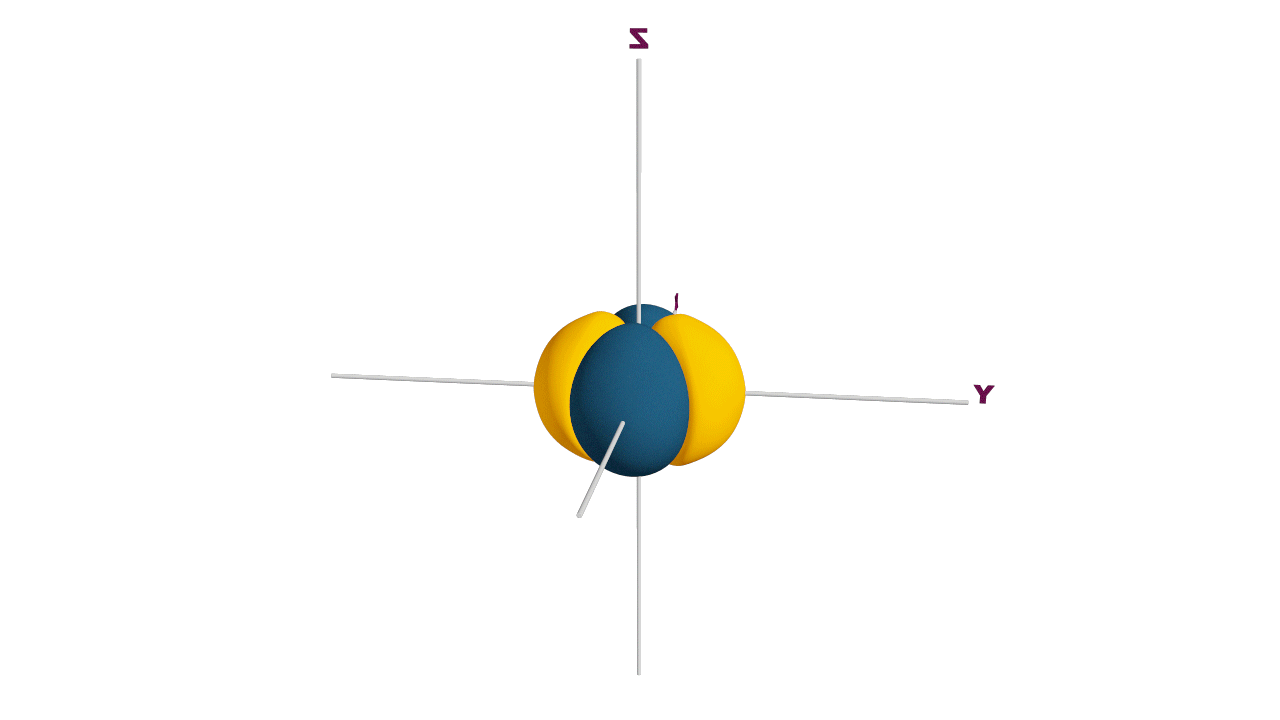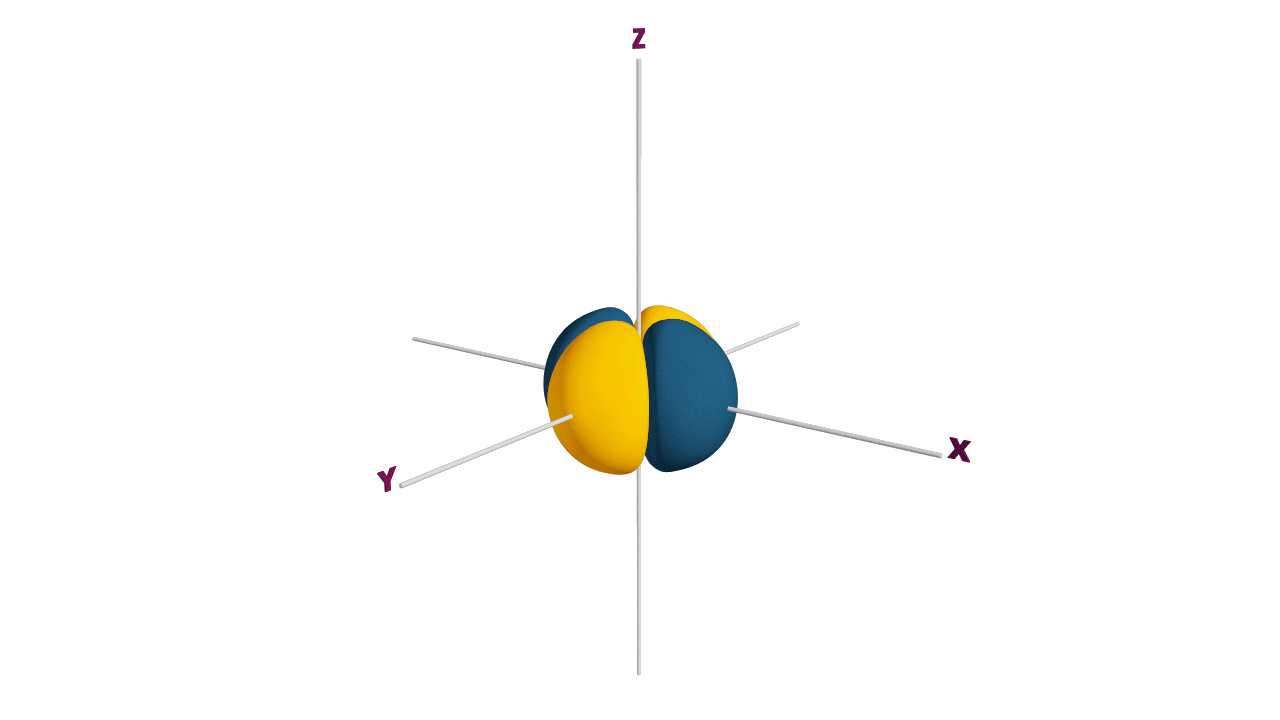
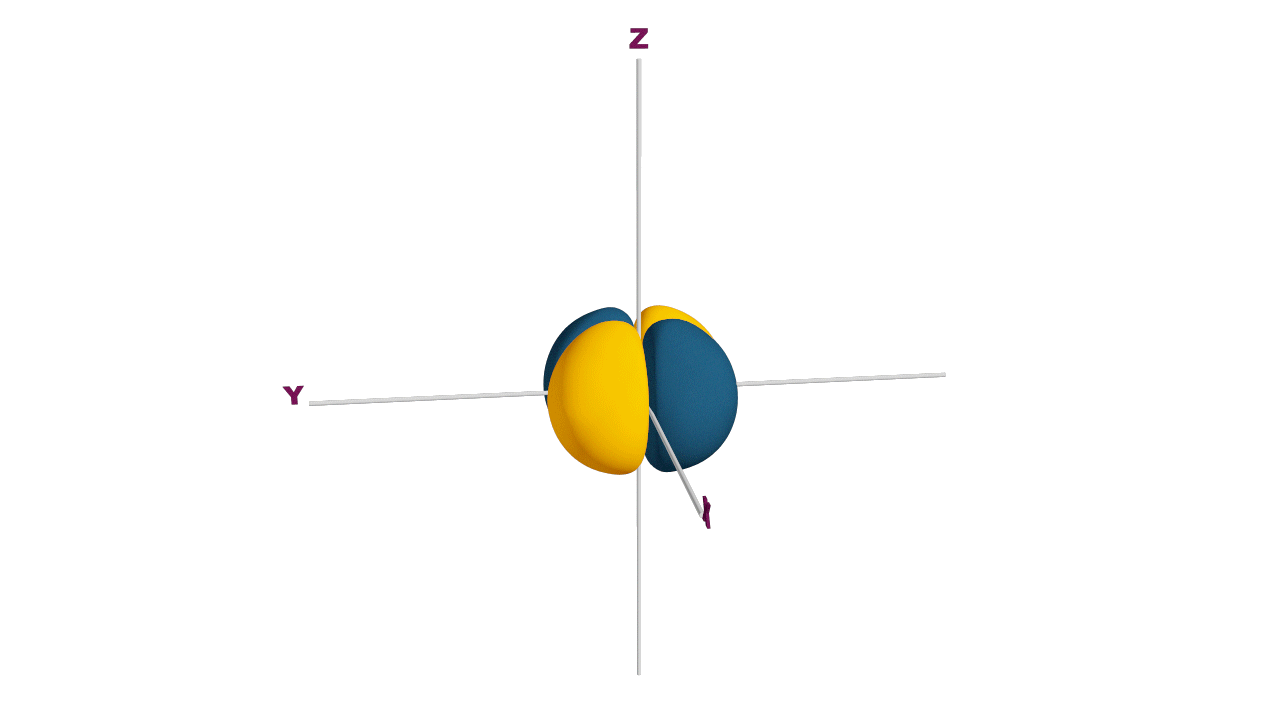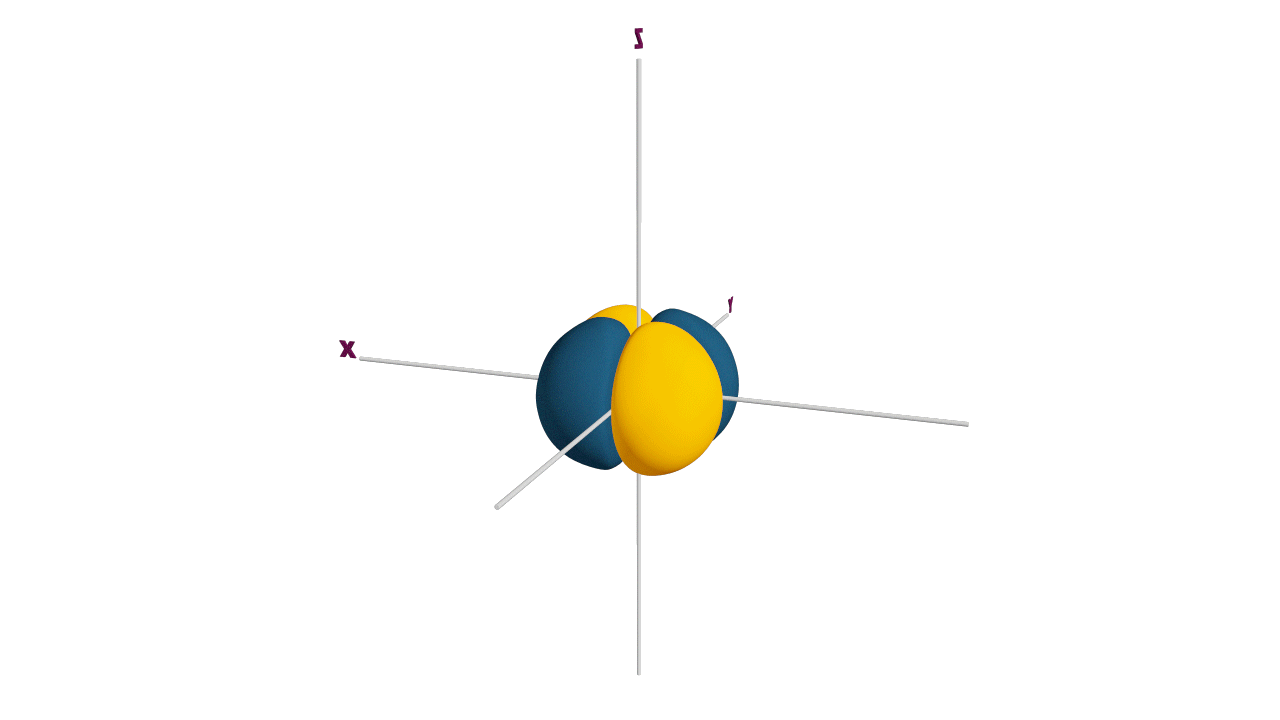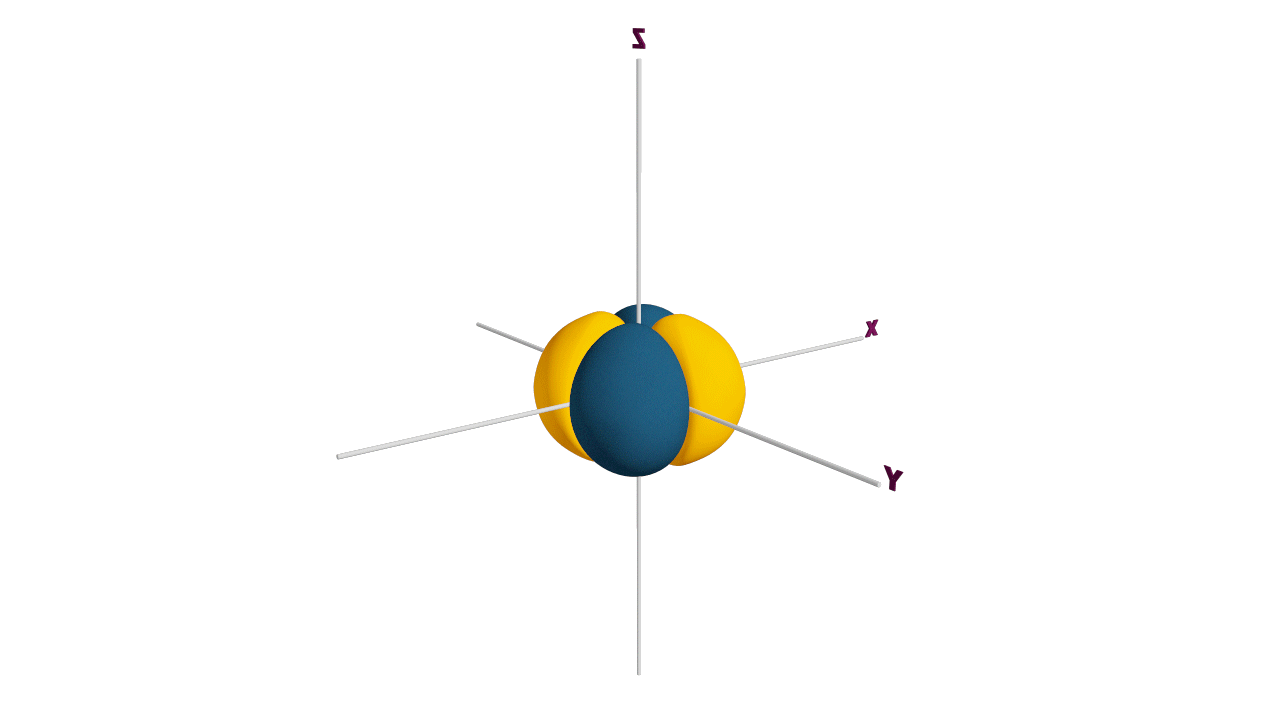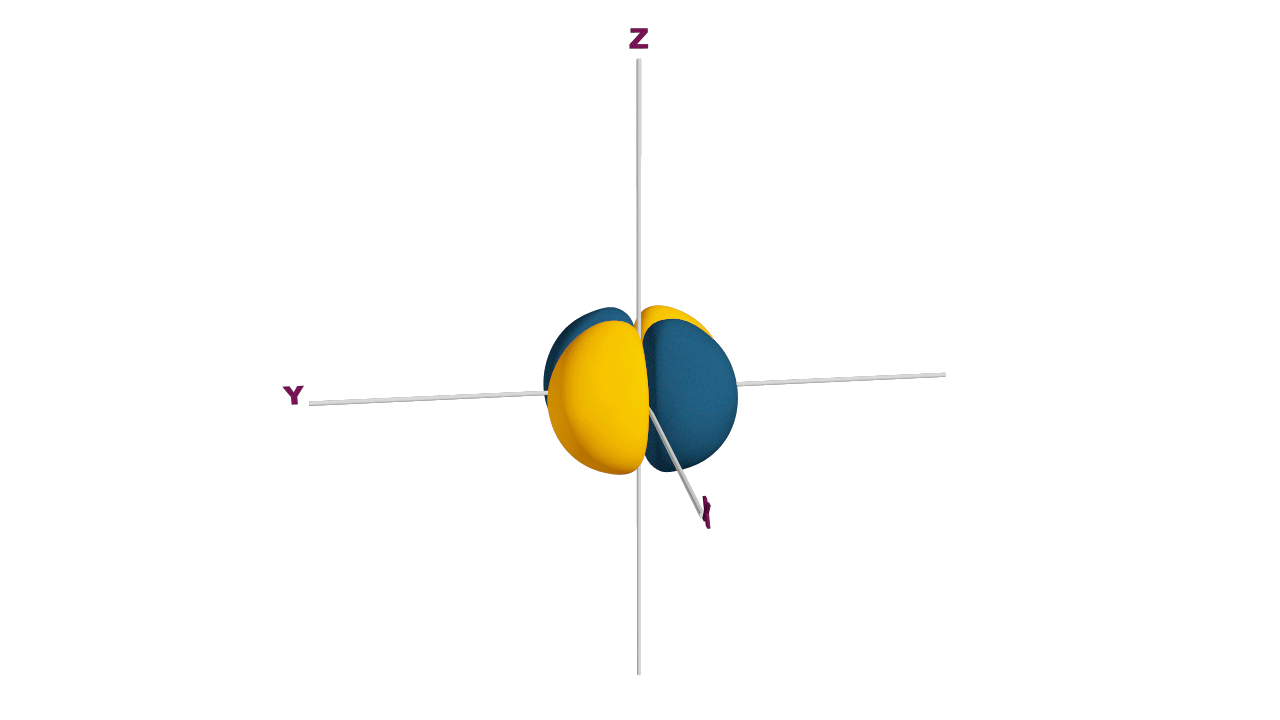
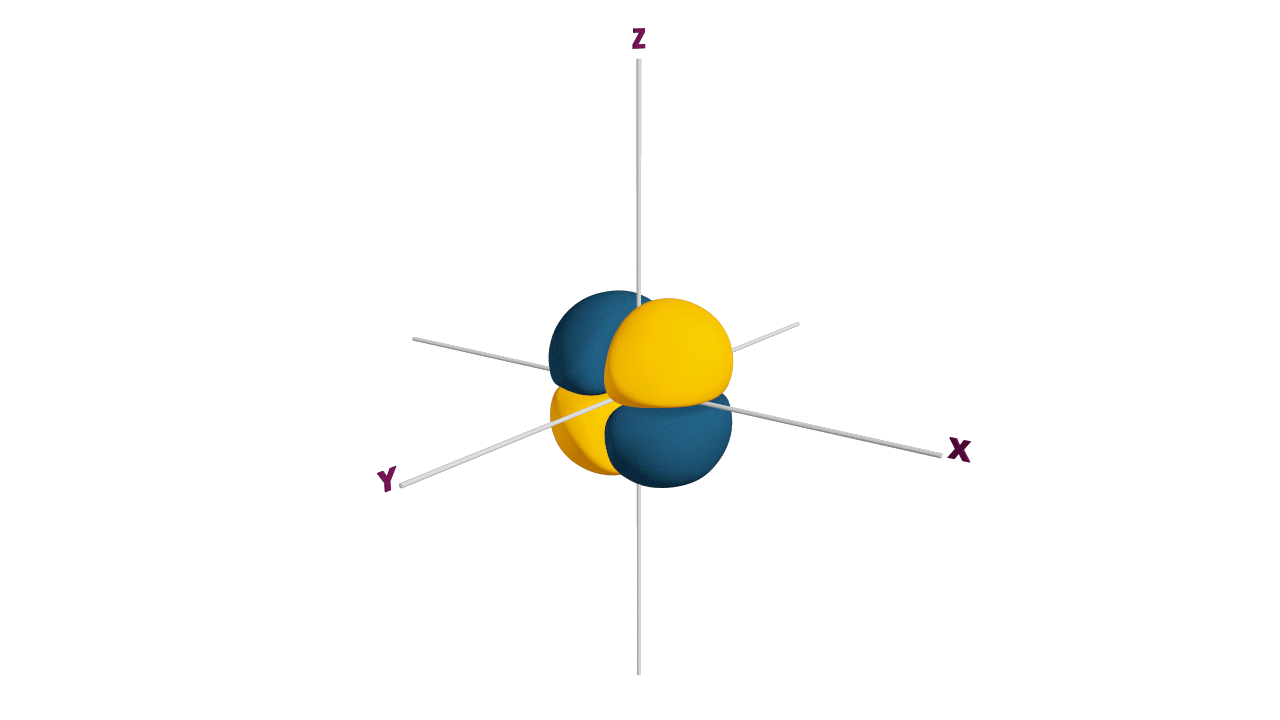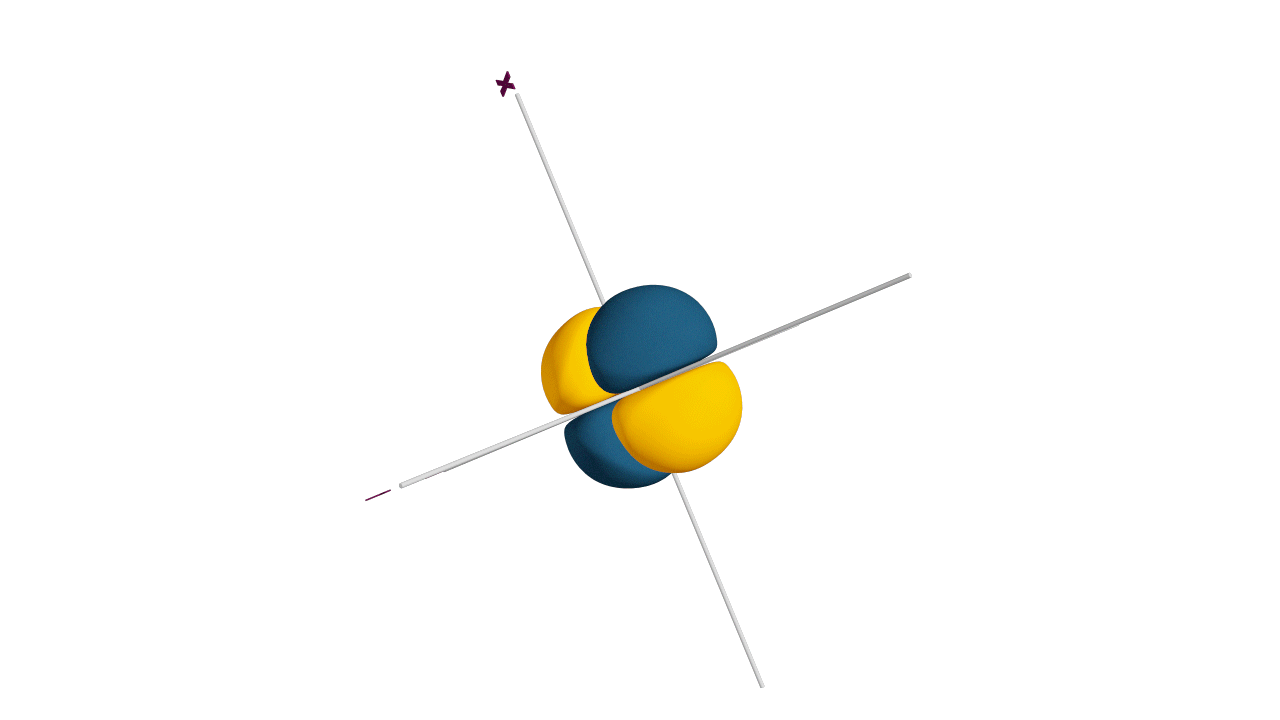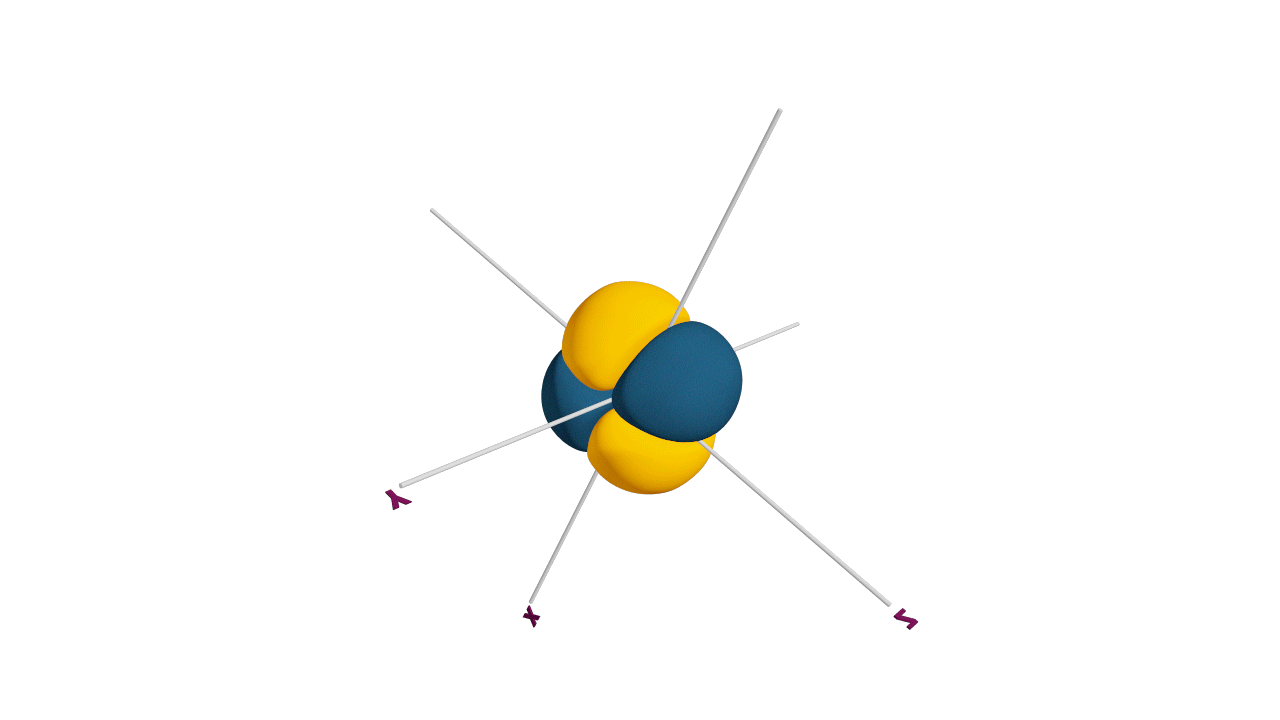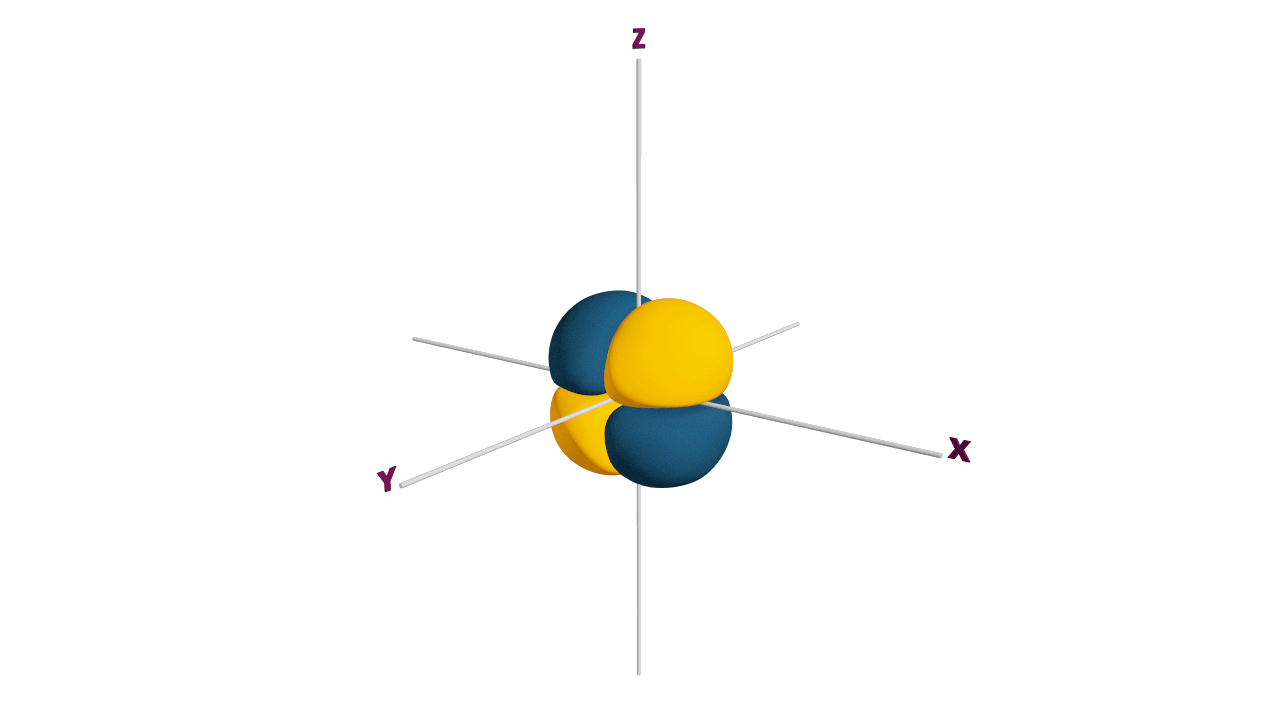
Fully filled subshells:
![]()
![]()
Due to the symmetrical distribution of electrons in degenerated orbitals (having different orientations), having less shielding with respect to others and electrons attracted more towards the nucleus, hence increasing stability.

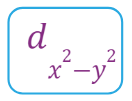

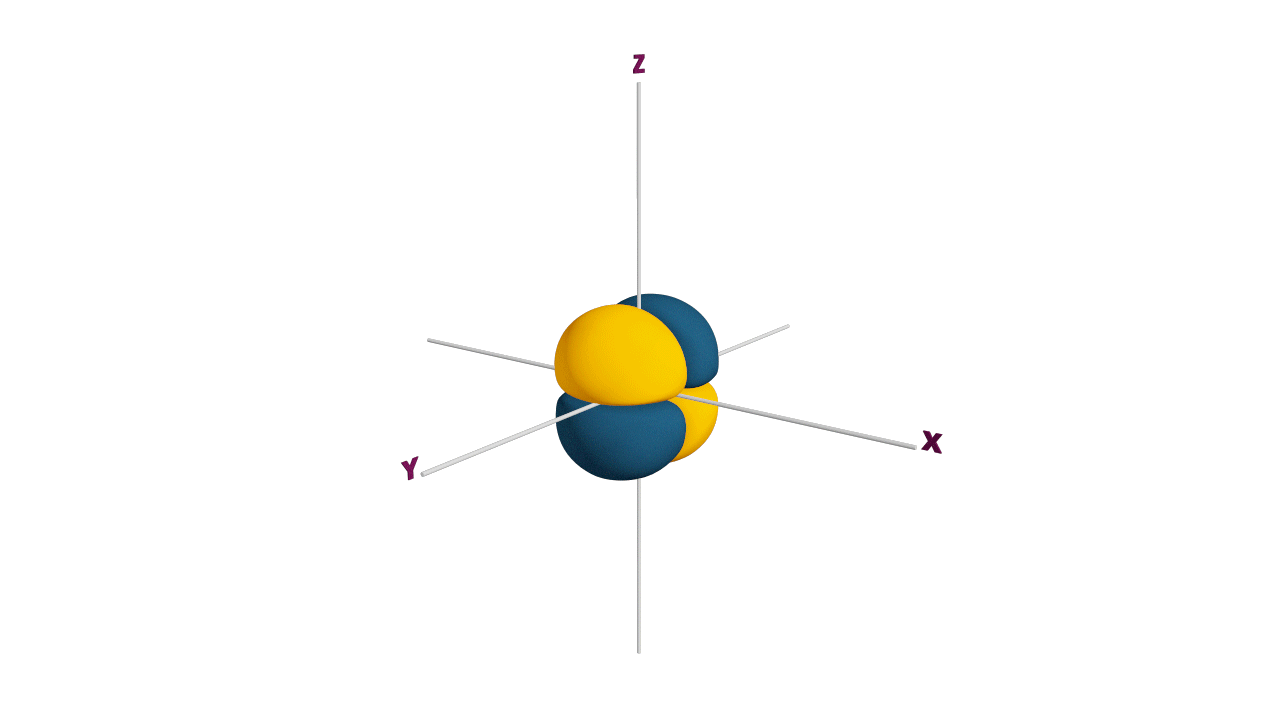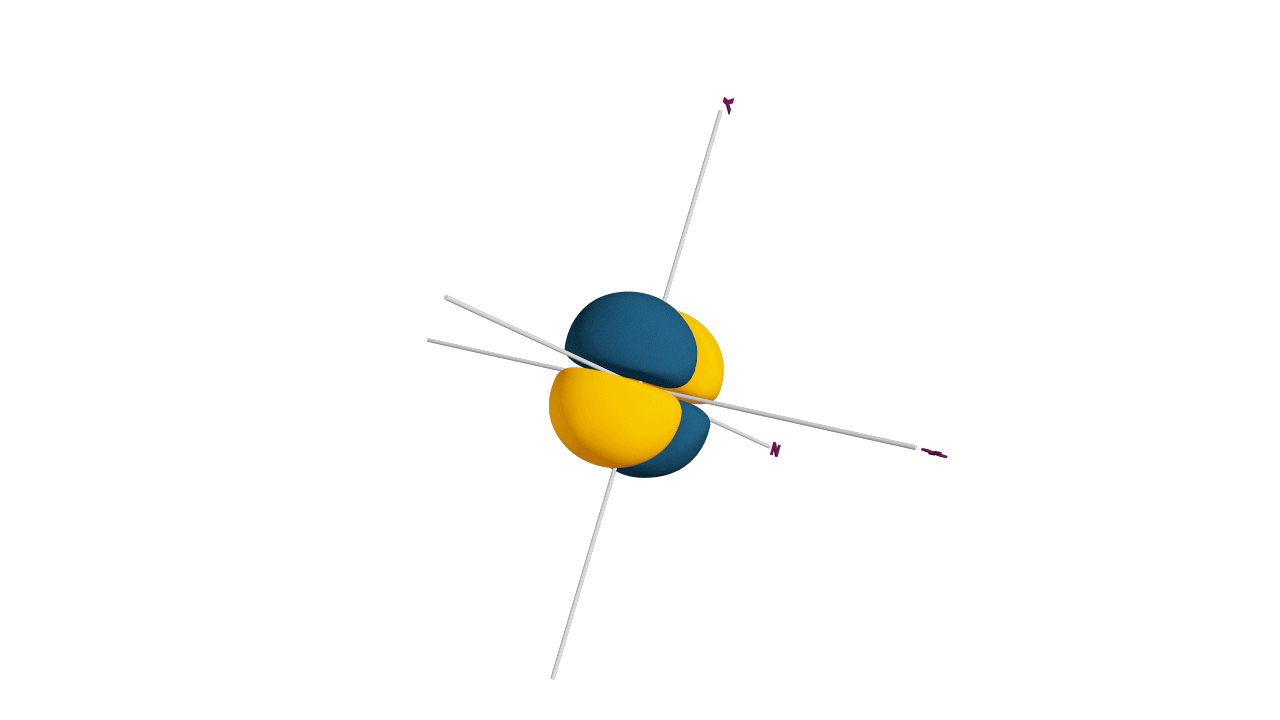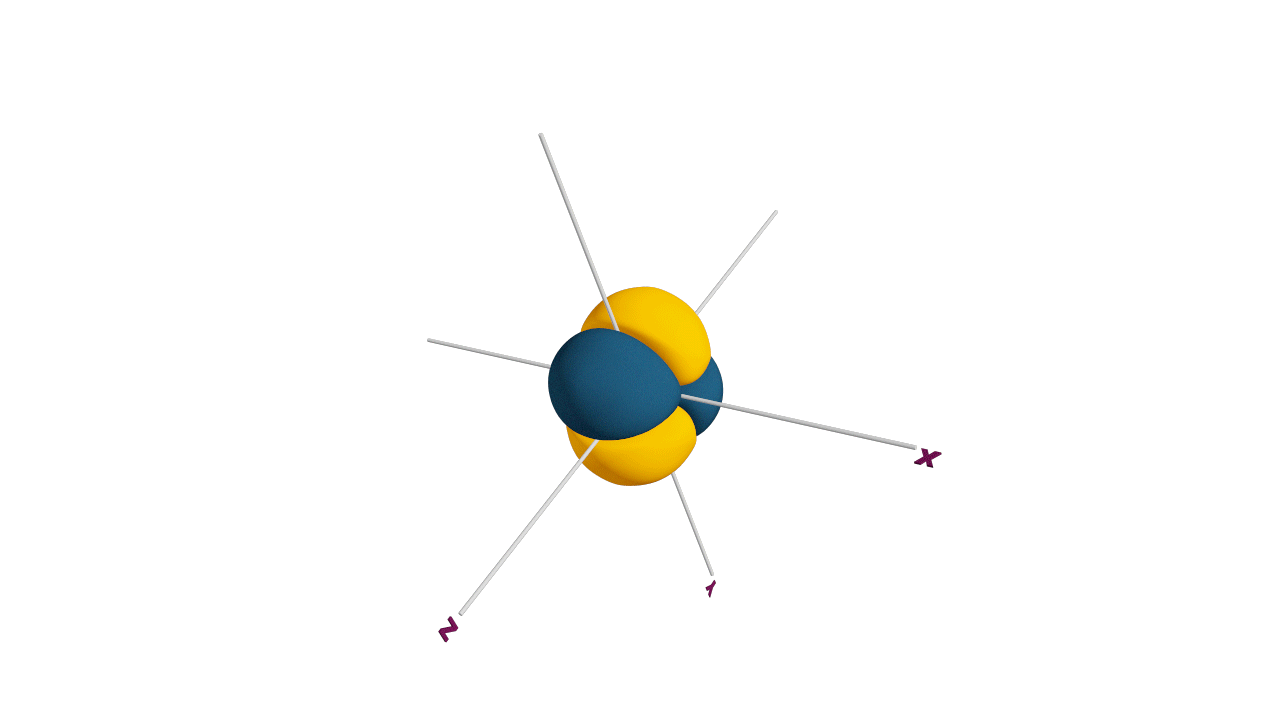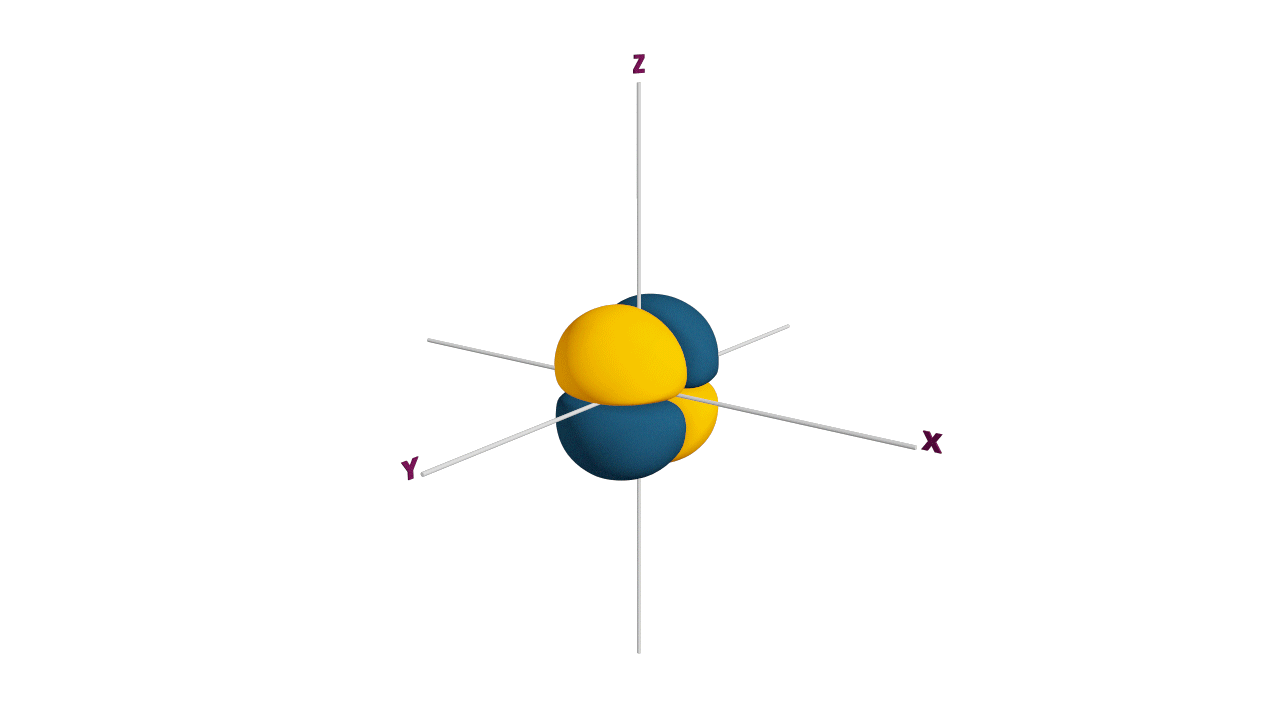

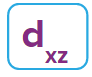
Practice problems:
Q 1. Calculate the number of maximum possible exchanges in configuration d5?
- 5
- 8
- 10
- 12
Answer: (A)
Solution:
d5 configuration:
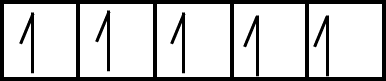
Number of electrons having same energy and spin (n) = 5
Minimum number of electrons required for exchange = 2
Q 2. Calculate the number of maximum possible exchanges in configuration d8?
- 5
- 10
- 13
- 15
Answer: (C)
Solution:
d8 configuration:

Case 1:
![]()
Number of electrons having same energy and spin (n) = 5
Minimum number of electrons required for exchange = 2
Case 2:
![]()
Number of electrons having same energy and spin (n) = 3
Minimum number of electrons required for exchange = 2
Total number of possible exchange = 10 + 3 = 13
Q 3. Calculate the number of maximum possible exchanges in configuration d10?
- 5
- 8
- 10
- 20
Answer: (D)
Solution:
d10 configuration:
![]()
Case 1:
![]()
Number of electrons having same energy and spin (n) = 5
Minimum number of electrons required for exchange = 2
Case 2:
![]()
Number of electrons having same energy and spin (n) = 5
Minimum number of electrons required for exchange = 2
Total number of possible exchange = 10 + 10 = 20
Q 4. which configurations are most stable?
- d3
- d5
- d8
- d10
Answer: (D)
Solution: d10 is the most stable configuration because of its highest exchange energy and due to symmetricity.
Frequently asked questions-FAQs
Q. What is spin multiplicity?
Answer: Maximum spin multiplicity = 2|S|+1
|S| = Modulus of the maximum spin of an atom
Generally, the higher the spin multiplicity of any configuration more the stable configurations.
Q. Which Orbital has maximum symmetry?
Answer: ‘s’ orbitals are the most symmetrical orbitals because of their spherical shape.

Q. Is it necessary to start filling any orbitals with an upward arrow?
Answer: No, you can start filling orbitals with upward or downward spin but make sure, no electron pairing takes place in the orbitals in a sub-shell until each orbital is occupied by one electron with parallel spin.
Q. What are different subshells?
Answer: A group of orbitals building up the orbit/shell of an atom is called subshells. E.g- s subshell, p subshell, d subshell, f subshell
NEET Related Links
JEE MAIN Related Links
JEE Main marks vs rank vs percentile
JEE Advanced Related Links
JEE Advanced Eligibility Criteria
JEE Advanced Chemistry Syllabus
JEE Advanced Registration Dates

