-
Call Now
1800-102-2727
Simple Titration - Definitions, Examples, Practice Problems & FAQs
If you have an unknown solution, how can you find the normality of an unknown solution?
The normality of an unknown solution is calculated by titration against a known standard solution. Take unknown solution in a conical flask and standard solution in a burette and add drop by drop to a conical flask. The endpoint of the process is indicated by a suitable indicator.
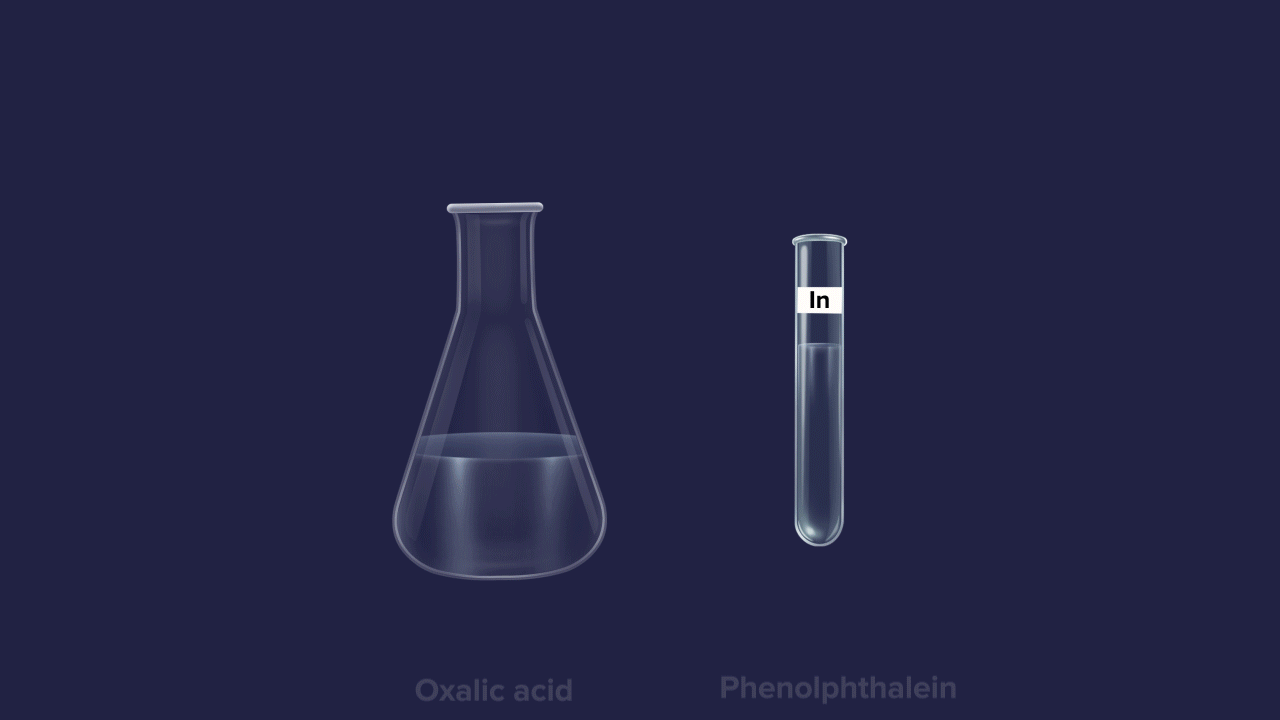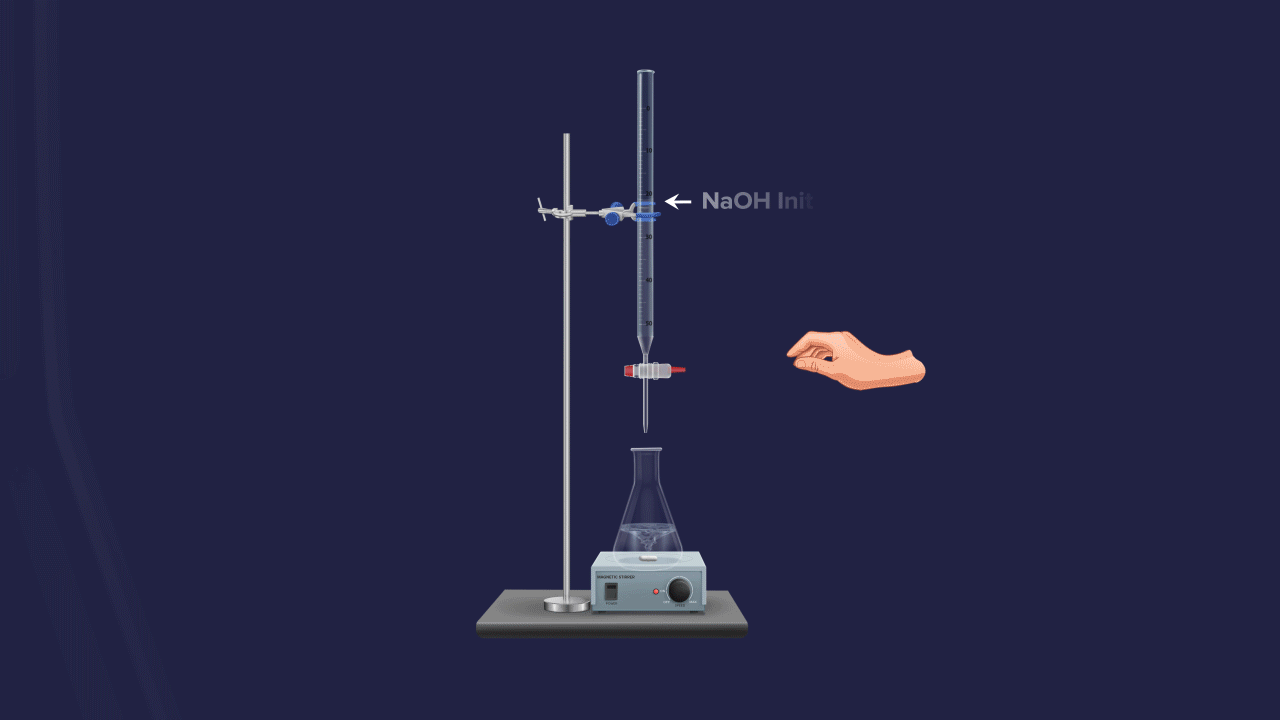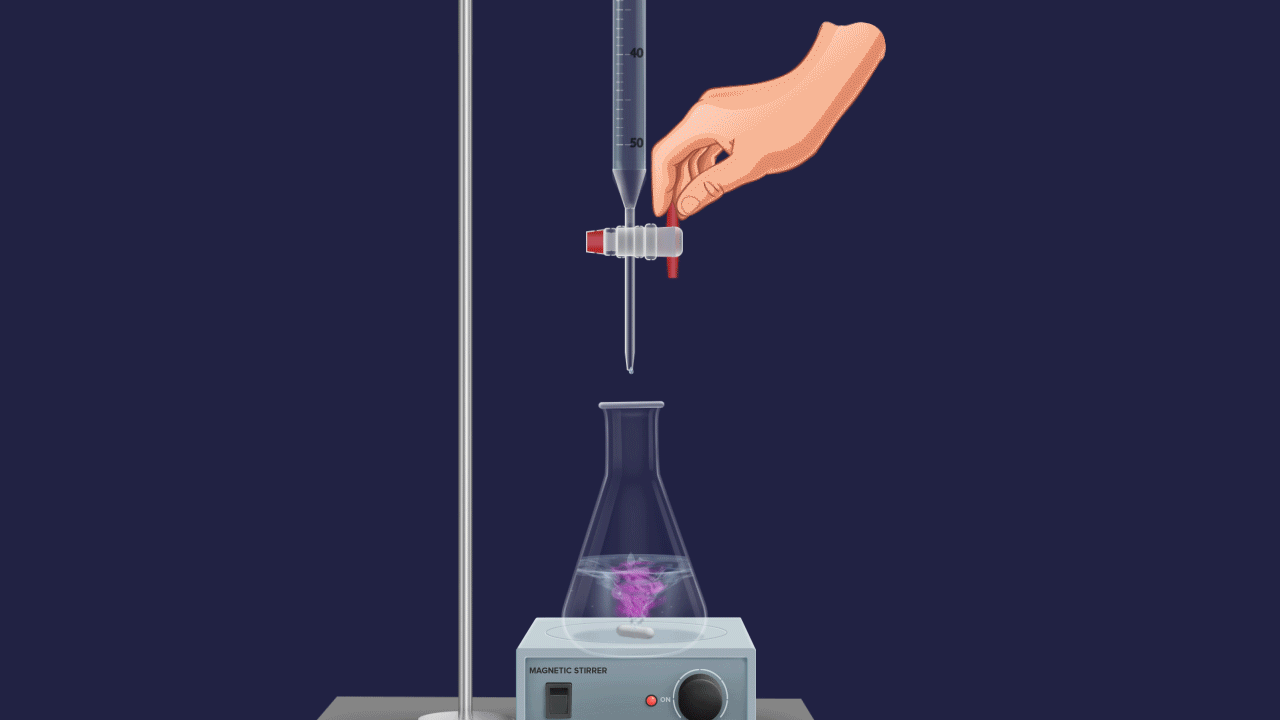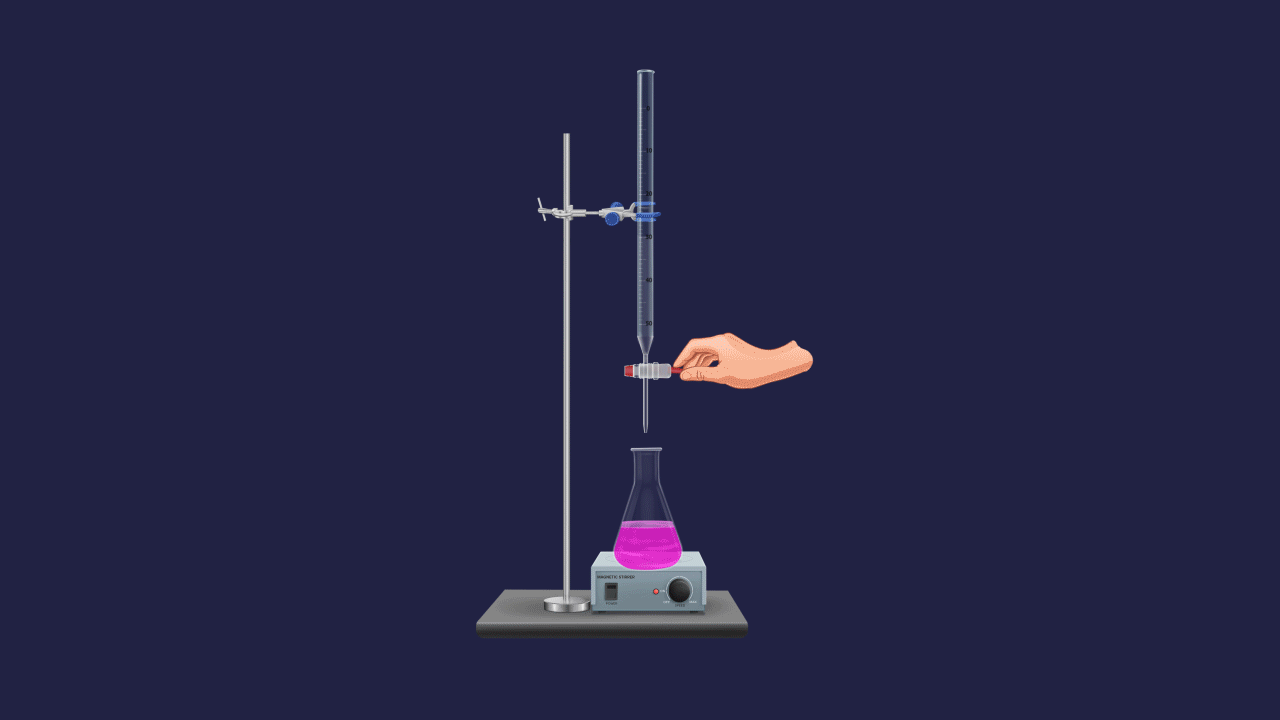

Table of contents:
- Titration
- Standard solution
- Equivalence point
- Indicators
- Acid-base titration
- Redox titration
- Practice problems
- Frequently asked questions-FAQs
Titration
Titration is the steady addition of a known concentration solution (known as a titrant) to a known volume of an unknown concentration solution (known as the analyte) until the reaction reaches the end point, which is often indicated by a change of colour.
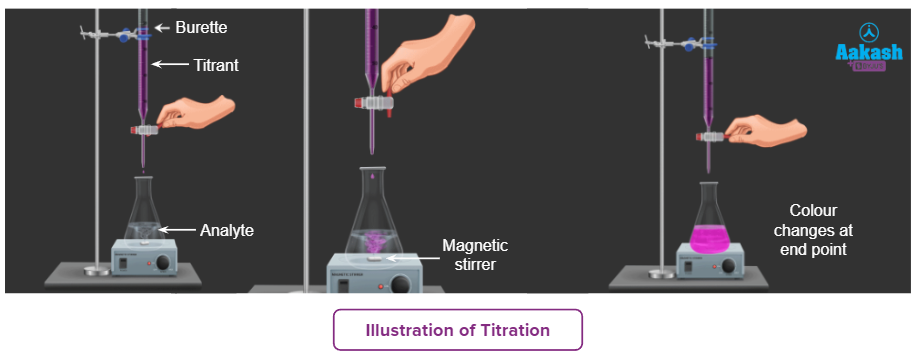
Standard solution
It is defined as a solution whose concentration is known. Generally, it is taken in a burette.
Type of standard solution:
- Primary standard solution
- Secondary standard solution
Primary standard solution:
The reagents that can be accurately weighed and their solution need not be standardized before use as these reagents do not react with air or moisture.
Examples: Oxalic acid (H2C2O4), Potassium dichromate (K2Cr2O7), Silver nitrate (AgNO3), Copper sulfate (CuSO4), ferrous ammonium sulfate (Mohr’s salt), Hypo solutions (Na2S2O3), etc.
Secondary standard solution:
The reagents that cannot be accurately weighed and their solution needs to be standardized before use as these reagents can easily react with air or moisture.
Examples: Sodium hydroxide (NaOH), Potassium hydroxide (KOH), Hydrochloric acid(HCl), Sulphuric acid (H2SO4), I2 solution, Potassium permanganate (KMnO4), etc.
Equivalence point
Number of equivalents of the titre =
Analyte: It is the solution with an unknown concentration.
The titrant is the solution with a known concentration (standard solution).
The normality of a solution can be calculated as:number of equivalent of solutevolume of solution (L)
Number of equivalent =
At the equivalence point,
Number of the equivalent of analyte = number of the equivalent of titrant
Endpoint
The endpoint is defined as the point during titration at which the indicator changes its colour.
Types of titration:
- Acid-base titration
- Redox titration
Indicators:
Indicators are generally organic weak acids or weak bases which show different colours in ionized and unionized forms or different colours in different pH ranges.
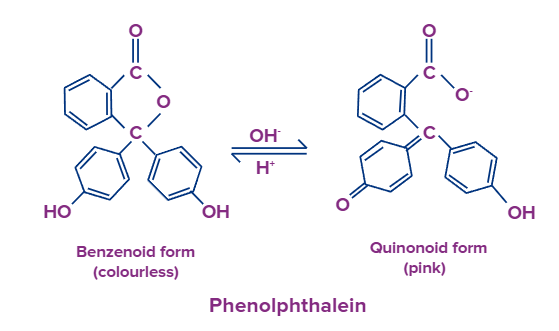
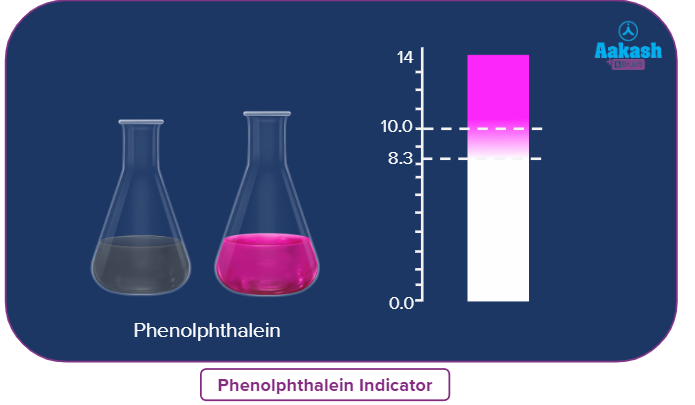
E.g-, Phenolphthalein is an organic weak acid (phenolphthalein ionized form is pink coloured and unionized form is colourless)
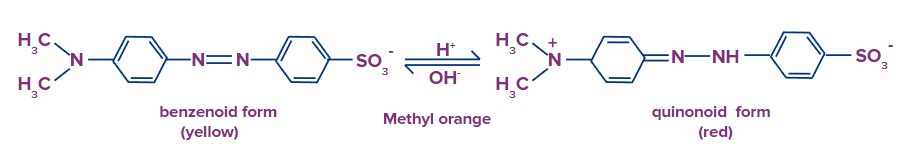
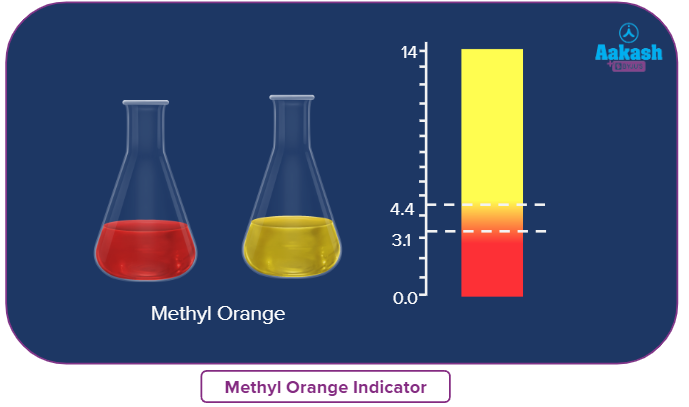
Methyl orange (MeOH) is a weak organic base (the ionized form is red and the unionized form is yellow).
List of some indicators and their colour according to pH
|
Indicator |
pH Range |
Acid |
Base |
|
Phenolphthalein |
8.0-10.0 |
colourless |
pink |
|
Thymol Blue |
1.2-2.8 |
red |
yellow |
|
Tetrabromphenol blue |
3.0-4.6 |
yellow |
blue |
|
Methyl red |
4.4-6.2 |
red |
yellow |
|
Rosolic acid |
6.8-8.0 |
yellow |
red |
|
Thymolphthalein |
9.4-10.6 |
colourless |
blue |
|
Tropeolin O |
11.0-13.0 |
yellow |
orange-brown |
Generally, in acid-base titration phenolphthalein and methyl orange used as indicators.
Acid-base titration
It is the method of finding the concentration of an unknown acid/base by titrating it against a base/acid with a known concentration.
Redox titration
In redox titration, the oxidizing agent (O.A.) reacts with the reducing agent (R.A.) to give products. In redox titrations, electrons are transferred between the titre and the titrant.
At the equivalence point,
Equivalent of oxidizing agent = equivalent of reducing agent
Practice problems:
Q 1. Phenolphthalein changes colour in presence of
a. NaOH
b. NaCl
c. NaHCO3
d. HCl
Answer: (C)
Solution: Phenolphthalein changes colour in presence of NaHCO3, weakly basic medium. Phenolphthalein changes colour in pH range 8.0 to 10.0 (colourless to pink).
Q 2. Methyl orange changes colour in presence of
a. NaOH
b. H2CO3
c. NaHCO3
d. HCl
Answer: (B)
Solution: Methyl orange changes colour in presence of H2CO3, weakly acidic medium. Methyl orange changes colour in the pH range from 3.1 to 4.4 (red to yellow).
Q 3. What is the normality of a solution of sulphuric acid, 15 mL of solution neutralizes 20 mL of 0.2 N NaOH solution?
a. 0.1 N
b. 0.267 N
c. 0.54 N
d. None of these
Answer: (B)
Normality of NaOH= 0.2 N
Volume of
Volume of
At the equivalence point,
Number of the equivalent of H2SO4 = number of the equivalent of NaOH
Q 4. Solution of 20 mL of 0.2 M ferrous sulfate titrated with potassium permanganate in an acidic medium. Calculate the total amount of potassium permanganate used
a. 10 mL of 0.2 M
b. 10 mL of 0.3 M
c. 20 mL of 0.2 M
d. 1 mL of 0.8 M
Answer: (D)
In acidic medium,
|
Element |
Initial oxidation state |
Final oxidation state |
|
Fe |
+2 |
+3 |
|
Mn |
+7 |
+2 |
Molarity of FeSO4= 0.2 M
Volume of FeSO4= 20 mL
At the equivalence point,
Number of the equivalent of KMnO4 = number of the equivalent of FeSO4
Frequently asked questions-FAQs
Q. What is the difference between endpoint and equivalence point titration?
Answer: The endpoint mode simulates the traditional titration technique, in which the titrant is added until the reaction is completed, as shown by a colour change in an indicator. The sample is titrated with an automatic titrator until a predetermined value is obtained.
The equivalency point is defined as the point where the analyte and reagent are present in equal amounts. In most circumstances, it's almost identical to the titration curve's inflexion point, such as titration curves generated from acid/base titrations. The associated pH or potential (mV) value and titrant consumption establish the curve's inflexion point (mL). The equivalency point is determined by the intake of a known concentration of titrant. The amount of substance that has reacted with the sample is calculated as the product of titrant concentration and titrant consumption. The measured points in an autotitrator are evaluated using certain mathematical processes, resulting in an evaluated titration curve. This evaluated curve is then used to calculate the equivalence point.
Q. In which industries or segments is titration used?
Answer: Titration is an extremely valuable analytical tool to check, primarily the purity of samples. This method has its used in a variety of fields such as electronics, Electroplating, Energy, Explosives, Machinery, Coal products, Coating, Textile, Food & Beverage, Paper&Pulp, and Petroleum, to name a few.
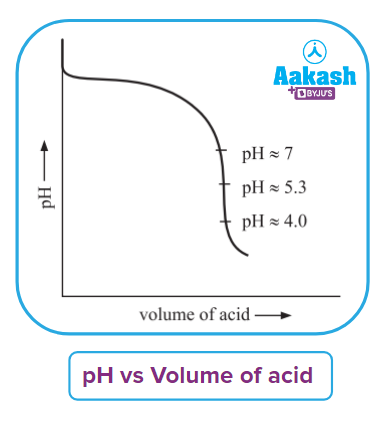
Q. Define titration curve.
Answer: The titration curve is plotted between pH of solution vs volume of titrant added, with the help of obtained curve we can decide which indicator is suitable for alarming the completion of the titration process.
Q. What are the different types of titrations?
Answer: The following is a list of several types of titrations such as
1. Titration of acid and base
2. Titration using complexometry
3. Redox Titration
4. Titration using iodine is called iodometry and iodimetry
5. Titration through precipitation
6. Titration using permanganate ion


