-
Call Now
1800-102-2727
Simple Distillation: Definition of Distillation, Simple Distillation, LImitations, Types of Distillation, Applications of Distillation, Practice Problems & Frequently Asked Questions
You must have seen big batteries in automobiles or inside inverters in your house to get electric power. These batteries also get weakened and need top-ups. I am not talking about your phone top-ups, but about distilling water top-ups of these batteries. These batteries lose water over a long time of use and have been replenished with “distilled not ordinary water” to bring back the density of sulphuric acid inside the battery. If not, you will lose your battery and have to spend again to buy a new one.
But, what is the difference between distilled water and( ordinary) water?
The word ‘distil’ refers to a process of distillation by which this distilled water is prepared.
You will come to know about the distillation and its variation along with its application in this text.
Table of Contents:
- Definition of Distillation
- Simple Distillation
- Limitations of Simple Distillation
- Types of Distillation
- Applications of Distillation
- Practice Problems
- Frequently Asked Questions(FAQs)
Definition of Distillation:
Distillation is a method of separating pure liquid components of a liquid mixture by way of heating and cooling. The distillation involves heating a liquid mixture and cooling(or condensing) and collecting the vapours.
The condensed liquid—known as the "distillate"—dribbles into a reservoir away from the original liquid.
Simple Distillation:
Simple distillation involves heating a liquid mixture in a "round bottom or distillation flask" and cooling(or condensing) the vapours above the liquid by passing the vapours through a condenser cooled by cold water and collecting the cooled liquid in a separate receiver container.
The technique is frequently employed with liquids that have non-volatile contaminants and non-interacting liquids or pure components with a boiling point difference of 25° C or more. The liquids also should be stable at their boiling points.
Principle:
Molecules at the surface of all substances try to get into the space above as vapours and create a vapour pressure. The amount of vapour formation changes with the state of the substance and the interactions existing in the solution. With heating, vapour formation and the vapour pressure increases. When the vapour pressure matches the atmospheric pressure, the liquid boils at a constant temperature known as its boiling point.
Liquids containing solid or high boiling contaminants will be left behind on distillation facilitating the separation of low boiling liquids. For instance, simple distillation can be used to purify simple organic liquids like chloroform, aniline, carbon tetrachloride, toluene, xylene, benzene, ethanol, acetone, etc.
Similarly, noninteracting liquids mixture having a sufficient (minimum 25oC) difference in their boiling points (like acetone and water) can be separated and purified using simple distillation. The liquid mixture's distinct components separate at their respective boiling points. The more volatile component distils first during a simple distillation, then follows the less volatile component. The two liquids are collected separately once the vapours condense. For instance, through distillation, chloroform (CHCl3) and aniline (C6H5NH2) with boiling temperatures of 334K and 462 K, respectively, can be separated. Chloroform that is more volatile evaporates first and is gathered in the receiver. The distillation flask still contains aniline.
Procedure:
Following is the general sequence of steps in the simple distillation.
Step-1: The liquid mixture is taken in a distillation flask halfway.
Step 2: The flask is fitted with a delivery tube, condenser and receiver flask as shown in the gif below
Step 3: Pass cold water through the condenser tube and gradually heat the mixture.
Step-4: The low boiling liquid evaporates faster, comes out of the delivery tube, get liquified in the
Condenser and collect in the receiver.
Observation: Solid and high boiling contaminants remain in the flask.
Example: Acetone boils at 329K while the water boils at 373K. When an acetone-water mixture is heated, acetone evaporates more quickly than water and gets collected in the receiver leaving water in the distillation flask.
Limitations of Simple Distillation:
Simple distillation cannot be used for the separation of pure components, under the following conditions,
- When the liquids have close boiling points
- If they are unstable and undergo chemical changes at their boiling points
- If they have too high boiling points and
- If there is an interaction between the component liquids.
In such cases, improved distillation techniques are used.
Types of Distillation:
There are four different distillation techniques, such as
1. Fractional distillation
2. Steam distillation
3. Vacuum distillation or distillation under reduced pressure
4. Azeotropic distillation
Fractional distillation:
Fractional distillation is a kind of distillation used to separate miscible liquids, and have close boiling points. The vapour component of any liquid mixture often varies from the liquid component. When the combination is heated, the liquid with the lower boiling point boils and forms vapour. The more volatile component spends more time in the vapour state than the liquid component does. In fractional distillation, more obstructions are placed in the vapour flow so that the less boiling fractions condense and fall back allowing the more boiling to go up and get separated. Multiple condensations and distillations are performed during the process, and the mixture is often broken down into its constituent elements. 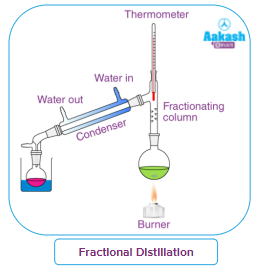
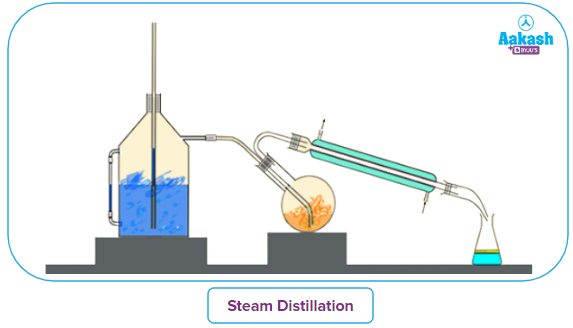
Steam distillation:
This technique is used to separate chemicals that may decompose at their boiling points and are insoluble in water. This process involves passing steam from steam generators to pass through the liquid to be distilled. The volatile organic chemical and steam mixture is condensed and collected in the receiver. A separating funnel is used to separate the chemical from the water.
On heating, each of the liquid and water would exhibit their individual vapour pressure independent of the other, and the mixture would start to boil at a temperature when the sum of the vapour pressures of the organic liquid (P1) and water (P2) equals atmospheric pressure (P).
P= P1 + P2
Vacuum distillation or distillation under reduced pressure:
Liquids with extremely high boiling points and those that disintegrate at or below those boiling points can both be purified using this method. By lowering the pressure on the surface, such liquids are made to boil at a temperature lower than their typical boiling point. The temperature at which a liquid's vapour pressure equals the outside pressure is known as the boiling point of the substance. To lower the pressure, a water pump or vacuum pump is employed. In the soap industry, glycerol is frequently separated from spent lye using this technique.
Applications of Distillation:
Scientific Use:
- It is used for recovery of volatile solvents after use.
- Purify low boiling liquids from contaminants
- Preparation of distilled water which is free from dissolved oxygen and other minerals. It is a pure form of water that can be used in pharmaceuticals, food preparations and heavy-duty batteries.
Practice Problems:
Q1. What is the core of the distillation process?
(A) Difference in boiling point
(B) Difference in melting poin
(C) Difference in temperature
(D) None of the above
Answer: (A)
Solution: The difference between the liquid boiling points provides the basis for the distillation process. Because various liquids evaporate at different temperatures due to their different boiling points Thus, the vapours that have cooled and the liquid that has formed can be collected separately.
Q2. Choose the chemicals that were separated using the simple distillation process.
(A) Aniline-water mixture
(B) Aniline-chloroform mixture
(C) Crude oil in the petroleum industry
(D) Glycerol sprint lye in the soap industry
Answer: (A)
Solution: Simple distillation is used to separate the mixture of aniline and chloroform. When there is a sufficient difference between the mixture's boiling points, a simple distillation process is used. The boiling point difference between aniline (462 K) and chloroform (334K) is 128 K so Aniline-chloroform mixture separation is achieved using this technique. A mixture of water and aniline uses steam distillation, a mixture of glycerol and spent lye uses reduced pressure distillation, and fractional distillation is used to separate crude oil.
Q3. When two liquids have boiling points close to each other, they are separated by:
(A) Steam distillation
(B) Fractional distillation
(C) Simple distillation
(D) None of the above
Answer: (B)
Solution: Fractional distillation is the method for separating two liquids when the difference in boiling points is small or close to each other. Such liquids create their vapours in the same temperature range and condense at the same time.
Q4. Among the following, which vaporises more quickly?
(A) Chloroform
(B) Aniline
(C) Water
(D) None of these
Answer: (A)
Solution: Since chloroform is a volatile and colourless liquid, it will vaporise more quickly than water and aniline.
Frequently Asked Questions(FAQs):
Q1. What is the process of industrial distillation?
Answer: Fractional distillation is one of the separation techniques that is frequently used in many industries. Although the scale of the distillation is increased, the fundamental concept behind the process does not change.
Industrial distillation is frequently performed in substantial, vertical cylindrical columns known as "distillation or fractionation towers" or "distillation columns," which can be up to 60 metres or taller and 0.65 to 6 metres in diameter. In these industrial towers, reflux is utilised to make sure the mixtures are completely separated.
Q2. Why does pressure affect a liquid's boiling point?
Answer: A liquid's vapour pressure equals atmospheric pressure when it reaches its boiling point. As a result, as the atmospheric pressure on a liquid's surface rises, the liquids boil at a higher temperature to produce more vapour pressure.
Q3. How do you separate a mixture of kerosene and petrol (whose boiling points vary by more than 250C) when they are miscible with one another?
Answer: Given that kerosene and gasoline are miscible with one another and have boiling points that vary by more than 250C, which is a significant difference, they can be separated using a simple distillation method.
Kerosene and petrol will be combined, and the distillation flask will be warmed. Due to its lower boiling point, petrol will initially start to evaporate and produce vapours as the mixture's temperature rises. Utilising a condenser, the petrol vapours are condensed and gathered through the condenser outlet. Higher boiling point kerosene will condense in the distillation flask and be left behind.
Q4. What advantages does fractional distillation provide over simple distillation?
Answer: Obstructions like glass beads in the fractionating column operate as "theoretical plates" on which the vapours can condense, re-evaporate, and then condense again, effectively distilling the compound numerous times. Fractional distillation produces a more effective separation than simple distillation.


