-
Call Now
1800-102-2727
Principle of Atomic Conservation (POAC): Definition, Solving Stoichiometric Problems Using POAC, Practice Problems & FAQs
Wanna have a cheese sandwich? But to have it first you have to prepare it! So, from where would you start. You need to know the ingredients to prepare the cheese sandwich.
What ingredients do you need to make the best cheese sandwiches?
Bread and cheese slices are the two main ingredients used to make this delicacy. To prepare a cheese sandwich we need two bread slices and one cheese slice. Now, if I ask you that the number of bread and cheese slices taken to prepare the sandwich is not equal to the number of bread and cheese slices in the sandwich after its preparation.
Would that be true?
The answer is no. The statement is incorrect. The total number of bread and cheese slices should be the same before and after the preparation of the cheese sandwich.
We can relate this context to a chemical reaction, in any chemical reaction the number of atoms of a particular element is the same in reactants and products. This principle is known as the Principle of Atomic Conservation (POAC). Let’s dive deeper into this principle and try to understand what exactly is POAC and how it is helpful in solving stoichiometric problems.
Table of Contents
- What is the Principle of Atomic Conservation (POAC)?
- Balancing a Chemical Reaction Using POAC Principle
- Solving Stoichiometric Problems Using POAC
- Practice Problems
- Frequently Asked Questions-FAQs
What is the Principle of Atomic Conservation (POAC)?
POAC is also based on the law of mass conservation, we know if atoms are conserved for a particular reaction which means the mass of atoms is also conserved. So, according to the principle of atomic conservation, the total number of atoms on the reactant side is always equal to the total number of atoms on the product side in a chemical reaction.
For example;
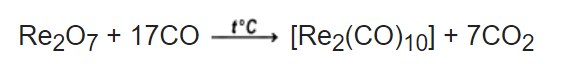
No. of atoms: Re = 2 C = 17 Re = 2 C = 7
O = 7 O = 17 C = 10 O = 14
O = 10
So, on the reactant side, total number of rhenium atoms is equal to 2 and same no. of atoms of rhenium are there at the product side. Similarly, there are 17 carbon atoms both on the reactant and product sides and 24 oxygen atoms on both sides of the chemical reaction. So, in this balanced chemical equation, the total number of atoms of rhenium, carbon and oxygen are balanced at both the reactant and product sides. It can be said that the total number of atoms of a particular element is the same in reactants and products.
Balancing a Chemical Reaction Using POAC Principle
One method to balance a chemical reaction is the Hit and Trial method, but this method is not feasible in the case where stoichiometric coefficients exceed 3-4. So, the more feasible method to balance a chemical reaction is using the principle of atomic conservation. Let’s understand this by taking a chemical reaction:
To balance the above chemical equation, let’s assume some stoichiometric coefficient variables for reactants and products.
Total number of calcium atoms in reactants = 3x
Total number of phosphorus atoms in reactants = 2x
Total number of carbon atoms in reactants = z
Total number of oxygen atoms in reactants = 8x+2y
Total number of silicon atoms in reactants = y
Total number of calcium atoms in products = a
Total number of phosphorus atoms in products = 4c
Total number of carbon atoms in products = b
Total number of oxygen atoms in products = 3a+b
Total number of silicon atoms in products = a
Now, according to the POAC principle;
Total number of calcium atoms in reactants = Total number of calcium atoms in products
i.e., 3x=a ...(i)
Total number of phosphorus atoms in reactants = Total number of phosphorus atoms in products
i.e., 2x=4c ...(ii)
Total number of carbon atoms in reactants = Total number of carbon atoms in products
i.e., z=b ...(iii)
Total number of oxygen atoms in reactants = Total number of oxygen atoms in products
i.e., 8x+2y=3a+b ...(iv)
Total number of silicon atoms in reactants = Total number of silicon atoms in products
i.e., y=a ...(v)
Let’s assume the value x=1.
By putting the value of x in equation (i), we get; a=3
By putting the value of x in equation (ii), we get; c=1/2
By putting the value of a in equation (v), we get; y=3
By putting the value of a, x and y in equation (iv), we get; b=5
By putting the value of b in equation (iii), we get; z=5
So, the chemical reaction becomes;
Furhter solving it, the balanced chemical equation is:
Solving Stoichiometric Problems Using POAC Principle
With the help of this method, we can solve stoichiometric problems without balancing the reactions. So, the number of moles of product formed can be calculated, and the number of moles of remaining reactant or consumed can also be calculated. Let us take the example of the disproportionation reaction of potassium permanganate.
KMnO4 K2MnO4+MnO2+O2 (unbalanced)
Applying principle of atomic conservation for potassium atom;
Moles of K atoms in reactant = Moles of K atoms in product
Moles of K atoms in KMnO4= Moles of K atoms in K2MnO4
We can observe, moles of K atoms in KMnO4= 1 mol atoms, moles of K atoms in K2MnO4= 2 mol atoms
Applying principle of atomic conservation for O atom;
Moles of O atoms in reactant = Moles of O atoms in product
Moles of O atoms in KMnO4= Moles of O atoms in K2MnO4 + Moles of O atoms in MnO2 +
Moles of O atoms in O2
We can observe, moles of O atoms in KMnO4= 4 mol atoms
moles of O atom in K2MnO4= 4 mol atoms
moles of O atom in MnO2= 2 mol atoms
moles of O atom in O2= 2 mol atoms
4 moles of KMnO4= (4 moles of K2MnO4) + (2 moles of MnO2) + (2 moles of O2)
Applying principle of atomic conservation for Mn atom;
Moles of Mn atoms in reactant = Moles of Mn atoms in product
Moles of Mn atoms in KMnO4= Moles of Mn atoms in K2MnO4 + Moles of Mn atoms in MnO2
We can observe, moles of Mn atoms in KMnO4= 1
Moles of Mn atoms in K2MnO4= 1
Moles of Mn atoms in MnO2= 1
Practice Problems
Q1. 3.37 g PtCl4 has gone through a number of chemical process treatments and finally all of chlorine is converted to a complex [Pt(en)2Cl2]SO4. The final weight of the product formed is:
Given: Molar mass of PtCl4 = 337 g mol-1, Molar mass of [Pt(en)2Cl2]Cl2 = 482 g mol-1
a. 9.64 g
b. 3.67 g
c. 7.98 g
d. Can’t be calculated
Answer: (A)
Solution: We are unaware of the type of chemical treatment provided to PtCl4 and intermediates formed but we know after completion of chemical treatment, all Cl atoms are converted to the final compound. So, we can conserve the Cl atoms in the reactant and product.
Applying the principle of atomic conservation for Cl atom.
moles of Cl atoms in reactant = moles of Cl atoms in product
moles of Cl atoms in PtCl4 = moles of Cl atoms in [Pt(en)2Cl2]SO4
We can observe, moles of Cl atoms in PtCl4= 4 mol
moles of Cl atoms in [Pt(en)2Cl2]SO4 = 2 mol
4 mass of PtCl4 molar mass of PtCl4 = 2 mass of [Pt(en)2Cl2]SO4 molar mass of[Pt(en)2Cl2]SO4
4 3.37 g337 g = 2 mass of [Pt(en)2Cl2]SO4 482 g
Q2. To produce 3 moles of NH3 using the Habers process, how many moles of N2 should be consumed?
a. 2.5 mol
b. 1.5 mol
c. 0.5 mol
d. 2 mol
Answer: (B)
Solution:
N2+H2---NH3 (unbalanced)
Applying principle of atomic conservation for N atom.
moles of N atoms in reactant = moles of N atoms in product
moles of N atoms in N2 = moles of N atoms in NH3
We can observe, moles of N atoms in N2= 2 mol
moles of N atoms in NH3= 1 mol
2 moles of N2 = 1 moles of NH3
2 moles of N2 = 1 3 mol
Q3. Find the mass of NH3 produced on the reaction of 2 moles of N2 with 6 moles of H2.
- 54 g
- 68 g
- 78 g
- 45 g
Answer: (B)
Solution:
N2+H2---NH3 (unbalanced)
Applying principle of atomic conservation for N atom.
moles of N atoms in reactant = moles of N atoms in product
moles of N atoms in N2 = moles of N atoms in NH3
We can observe, moles of N atoms in N2= 2 mol
moles of N atoms in NH3= 1 mol
2 moles of N2 = 1 moles of NH3
2 2 mol = 1 moles of NH3
moles of NH3= 4 mol
Mass of NH3= 4 17 g = 68 g
Q4. To produce 68.1 L of NH3at STP, how much volume of N2 and H2 is required respectively?
a. 67.2 L & 22.4 L
b. 67.2 L & 109.4 L
c. 33.6 L & 100.8 L
d. 34.05 L & 102.15 L
Answer: (D)
Solution:
Moles of NH3 at STP = 68.122.7mol= 3 mol
N2+H2---NH3 (unbalanced)
Applying principle of atomic conservation for N atom.
moles of N atoms in reactant = moles of N atoms in product
moles of N atoms in N2 = moles of N atoms in NH3
We can observe, moles of N atoms in N2= 2 mol
moles of N atoms in NH3= 1 mol
2 moles of N2 = 1 moles of NH3
2 moles of N2 = 1 3 mol
moles of N2 = 1.5 mol
Volume of N2 at STP = 1.5 22.7 L = 34.05 L
Applying principle of atomic conservation for H atom.
moles of H atoms in reactant = moles of H atoms in product
moles of H atoms in H2 = moles of H atoms in NH3
We can observe, moles of H atoms in H2= 2 mol
moles of H atoms in NH3= 3 mol
2 moles of H2 = 3 moles of NH3
2 moles of H2 = 33 mol
moles of H2 = 4.5 mol
Volume of H2 at STP = 4.5 22.7 L = 102.15 L
Frequently Asked Questions-FAQs
Q. What is the advantage of using POAC methodology?
Answer: Following are the advantages of using POAC methodology:
- Balancing of chemical equations is not required in the majority of problems as the method of balancing the chemical equation is based on the principle of conservation of atoms.
- To balance the chemical equation, which is difficult to balance using the Hit and Trial method.
Q. What are the benefits of POAC over mole-mole analysis?
Ans: No balancing of reaction is required and information is sufficient. Even if we are not able to write all products of a particular reaction, in those cases as well POAC can be applied to solve stoichiometric problems.
Q. How principle of atomic conservation ensures that mass is also conserved in a chemical reaction?
Answer: In a chemical reaction, no new atoms are created or taken away from the chemical reaction. The atoms which are there initially in the compounds taken as reactants are only rearranged to form products. Now, according to the principle of atomic conservation, the number of atoms of a particular element is the same in reactants and products which ensures the mass is also conserved.


