-
Call Now
1800-102-2727
Preparation of Alkanes and Alkenes, Practice Problems & Frequently asked questions-FAQs
For a long time, hydrocarbons have played an important role in our daily lives. Saturated hydrocarbons make up a significant portion of these compounds.
Can you name some saturated hydrocarbons found in our environment? I'll give you a hint!
CNG, which we use as a transportation fuel, is primarily composed of alkanes methane and ethane. Similarly, LPG or cooking gas is primarily composed of propane and butane.

So, in this article, you will gain a thorough understanding of alkanes & alkenes, their preparation, and their properties.
TABLE OF CONTENT
- Alkanes
- Preparation of Alkanes
- Preparation of alkanes from alkenes and alkynes
- Production of ethane from ethene
- Preparation of alkanes from alkyl halides
- Preparation of Alkanes from Carboxylic Acids
- Alkene
- Preparation of Alkenes
- Hydrogenation of Alkynes
- Dehydrohalogenation of Alkanes
- Dehalogenation of vicinal dihalides
- Dehydration of Alcohols
- Practice Problems
- Frequently Asked Questions
Alkanes:
Alkanes are saturated aliphatic hydrocarbons. CnH2n+2 is the general formula for an alkane, where n denotes the number of carbon atoms.
Examples: CH4, C2H6,C3H8,C4H10,C5H12,C10H22, etc.

The most basic alkane is methane, which is a key component of CNG. Alkanes are also known as paraffins due to their low affinity for many reagents. In this case, low reactivity does not imply low reactivity with all reagents in general.
CNG, for example, is derived from alkanes. It exhibits a violent reaction to heat.
Preparation of Alkanes:
Preparation of alkanes from alkenes and alkynes:
The addition of H2 in the presence of a catalyst (Ni/Pt/Pd) is required for catalytic hydrogenation of alkenes and alkynes. In this case, H2 acts as a reducing agent.
Catalyst: A reagent that increases the rate of a reaction without being consumed by the chemical reaction. However, in some cases, a catalyst can reduce the rate of a reaction.
At room temperature, the catalysts Pt and Pd catalyze the reaction. However, catalyst Ni necessitates a higher temperature and pressure.
A syn addition reaction is catalytic hydrogen addition.
Examples:
- When ethane is reduced with H2 in the presence of a metal catalyst (Ni/Pt/Pd), it produces ethane.
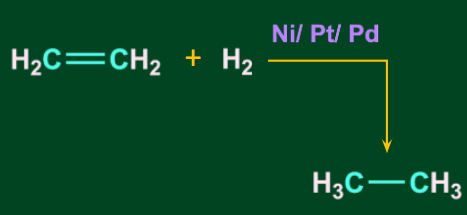
![]()
- When prop-1-yne is reduced with H2 in the presence of a metal catalyst (Ni/Pt/Pd), it produces propane.
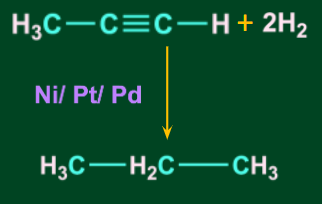
![]()
Production of ethane from ethene:
Hydrogen (H2) and alkene are first adsorbed on the metal catalyst's surface. The bonds are then partially formed and broken (the bond of alkene, bond of hydrogen, H2, breaks partially, while the new C–H bonds are partially formed in the transition state), resulting in a four-membered cyclic transition state. Syn addition occurs when hydrogens are added to the same side of a double bond. Finally, the alkane is formed, which is desorbed from the metal catalyst's surface.

![]()
Preparation of alkanes from alkyl halides:
Alkyl halides can be treated with Zn or dil. HCl or Na, dry ether to produce alkane.
- By reduction of an alkyl halide with Zn/dil. HCl:
The product contains the same number of carbon atoms as the reactant (alkyl halide). The halogen atoms (Cl, Br, and I) are substituted by H in this reaction.
Because C–F bonds are so strong, alkyl fluorides (R–F) cannot be reduced with Zn, dil. HCl.
Examples:
- On reduction of methyl chloride by Zn/dil HCl, Methane and zinc chloride are produced.

![]()
- When n-propyl chloride is reduced with Zn/dil HCl, propane and zinc chloride are formed.
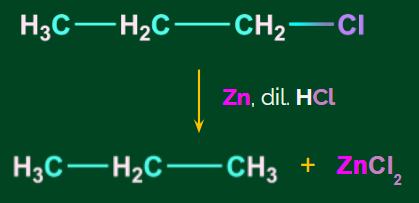

- Using Na, dry ether to reduce alkyl halides (Wurtz reaction):
Wurtz reaction: The Wurtz reaction is an organic chemical coupling reaction in which sodium metal reacts with two molecules of alkyl halides in a dry ether solution to produce a higher alkane and a compound sodium halide.
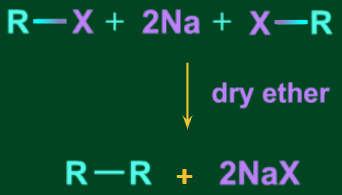
![]()
Example:
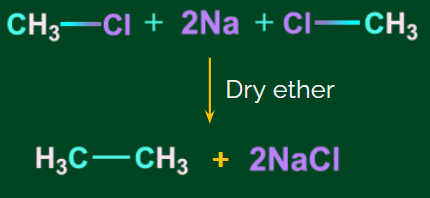
![]()
Preparation of Alkanes from Carboxylic Acids:
There are two ways to make alkanes from carboxylic acids:
Decarboxylation by soda-lime:
The process of removing CO2 is known as decarboxylation. The carboxylic acid also loses a CO2 molecule during the soda-lime process.
Reagents employed: CaO + NaOH (caustic soda) (Quick lime)
When carboxylic acids or their salts are heated with a 1:3 mixture of CaO and NaOH, i.e., soda lime, an alkane is formed as a result of CO2 removal. Decarboxylation is the process of removing carbon dioxide from a carboxylic acid.
The alkanes produced have one less C atom than the carboxylate ion. Decarboxylation is therefore a step-down reaction.
Reaction Involved:

![]()
Mechanism of the reaction:
- The electrophilic carbon of the C=O is attacked by the hydroxide ion (OH-) in the first step, and the electron density moves towards the oxygen atom from the C=O pi bond, resulting in the appearance of a negative charge on the oxygen atom.
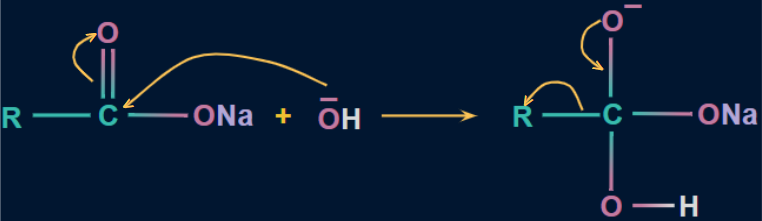
![]()
- The lone pair of oxygen forms a double bond with carbon again in the second step, allowing the alkyl substituent to leave in the form of R- (carbanion).
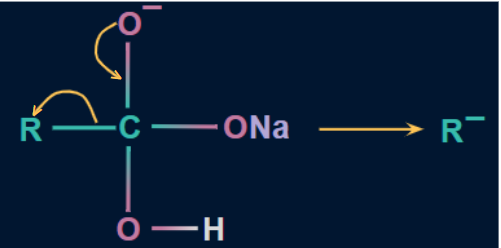
![]()
- R- reacts with the H of sodium bicarbonate to produce alkane as the end product.
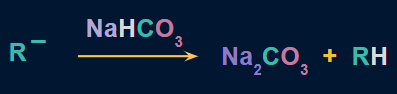
![]()
Decarboxylation rate by soda lime: The carboxylate ion's alpha carbon acquires a negative charge during decarboxylation. The higher the carbanion's stability, the faster the rate of decarboxylation.
|
Decarboxylation rate by soda lime ∝ Stability of carbanion |
Kolbe’s electrolysis:
At the anode, electrolysis of an aqueous solution of the sodium/potassium salt of a saturated carboxylic acid yields an alkane with an even number of C atoms. The following is the general chemical reaction:
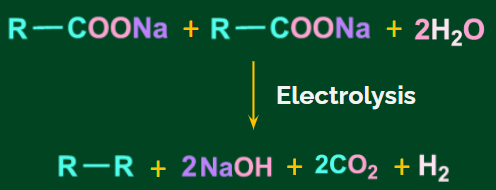
![]()
Mechanism:
- The reaction is thought to take the following steps:
RCOO-Na+ +RCOO-Na+ 2RCOO- +2Na+
- At anode (Oxidation):
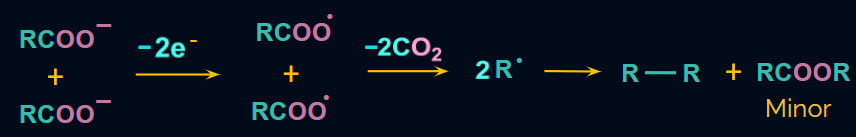
![]()
- At the cathode (Reduction):
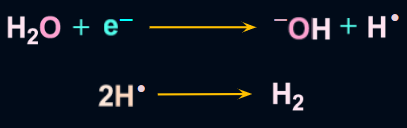
![]()
Alkene:
- Unsaturated hydrocarbons with at least one double bond are known as alkenes.
- CnH2n is the general formula for alkenes.
- Olefins are another name for alkenes (oil-forming).

Preparation of Alkenes:
In general, alkenes can be prepared using one of four reagents:
- Alkynes
- Alkyl halides
- Vicinal dihalides
- Alcohols
Hydrogenation of Alkynes:
- Lindlar’s Catalyst
H2/Pd, CaCO3, and quinoline used as reagents.
Poisoned palladium catalyst: It is made of powdered calcium carbonate coated with palladium and poisoned with quinoline to reduce its catalytic activity and prevent complete alkyne reduction. Lindlar's catalyst is used to perform partial alkyne-alkene reduction. Poisoning reduces the catalytic activity to some extent, limiting the reduction of the alkyne to the formation of an alkene.
Example:
Only cis alkenes are formed in the presence of Lindlar's reagent.

![]()
Reduction of Alkynes using Lindlar’s Catalyst:

![]()
- Birch Reduction:
The Birch reduction is the conversion of alkyne to alkene using Na/liquid NH3 in the presence of EtOH. This is a trans addition reaction.
In the Birch reduction, alkynes give trans alkenes.
Example:
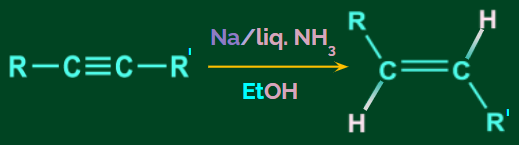
![]()
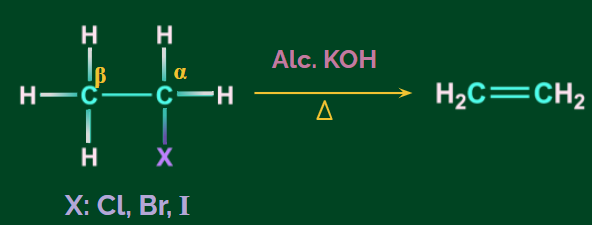
Dehydrohalogenation of Alkanes:
- Dehydrohalogenation, or the removal of HX, can be used to produce alkenes from alkyl halides.
- Alcoholic KOH is the reagent used.
- The β-elimination reaction is named after the removal of β-hydrogen.
- The hydrogen atom opposite the halogen atom attached to the β-carbon (carbon atom next to the halogen atom) is removed. As a result, it is referred to as anti-elimination.
Example:
![]()
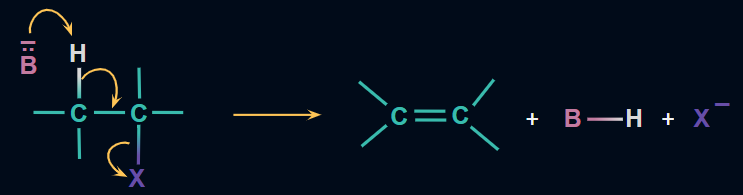
Mechanism:
The alkoxide ion derived from alcoholic KOH functions as a strong base. It attacks and separates the β-H atom from the alkyl halide molecule, which is slightly acidic in nature. The electrons shared by the broken hydrogen carbon bond are drawn to the ⍺-carbon atom, which has a slight electron deficit due to its attachment to the halogen atom. The halogen atom breaks as these electrons approach the ⍺-carbon atom, resulting in the formation of the double bond.
![]()
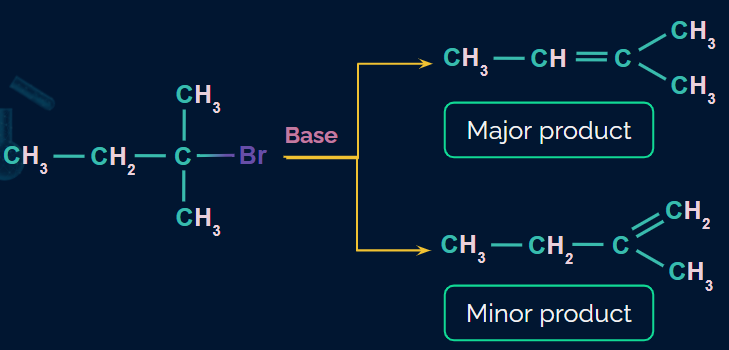
Saytzeff or Zaitsev rule:
In most elimination reactions with two or more possible products, the one with the highly substituted double bond will be the dominant and major product.
Example:
![]()
Dehydrohalogenation of RX:
- Reactivity: R−F < R−Cl < R−Br < R−I
- Because of its large size, an iodide is a better-leaving group.
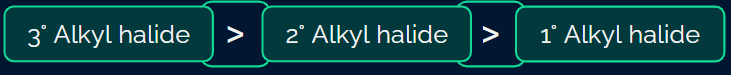
Rate of elimination:
- The rate of the elimination reaction is directly proportional to the stability of the alkene.
- Rate: Primary alkyl halide < Secondary alkyl halide < Tertiary alkyl halide
![]()
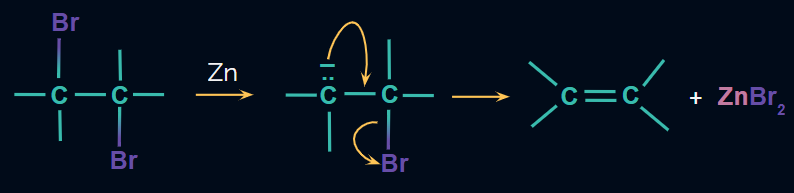
Dehalogenation of vicinal dihalides:
- Dehalogenation, or the removal of X2, is used to produce alkenes from vicinal dihalides.
- NaI in acetone or Zn in the presence of acetic acid, ethanol, or Zn/Δ are the reagents used.
- This reaction is carried out via the E2 mechanism.
Reaction:
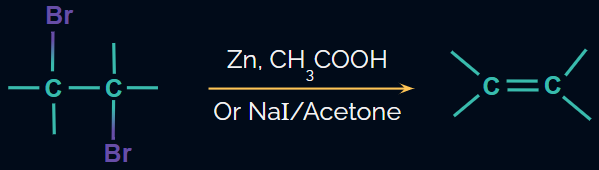
![]()
Mechanism:
![]()
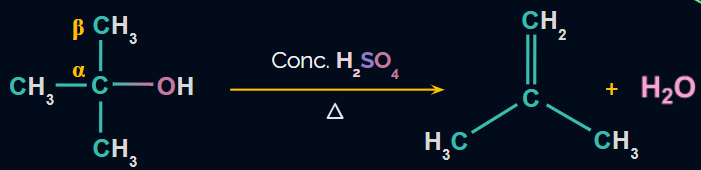
Dehydration of Alcohols:
- Acidic dehydration, or the elimination of H2O in the presence of acid, can be used to produce alkenes from alcohol.
- Conc. H2SO4 and heat is used as the reagents.
- Because β-H is removed, this reaction is known as the β-elimination reaction.
- The reaction is carried out via the E1 mechanism.
- Because the reaction involves the formation of a carbocation, rearrangement is possible.
Reaction:
![]()
Mechanism:
Step 1: Protonated alcohol formation
- Alcohols are weak bases due to the presence of two lone pairs of electrons on oxygen.
- As a result, they combine with strong mineral acids (H2SO4) to form protonated alcohol.
- Alcoholic oxygen protonation aids in the elimination of water molecules.
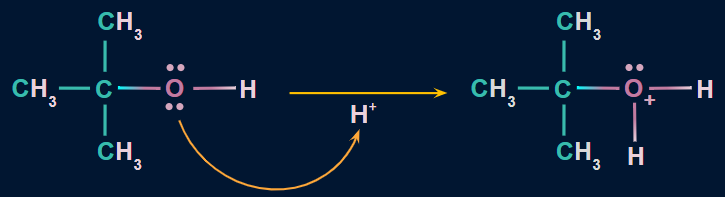

Step 2: Carbocation formation
- The C−O bond is broken with the elimination of a water molecule in this step to form a carbocation.
- This is the slow step. As a result, it is regarded as the rate-determining step.
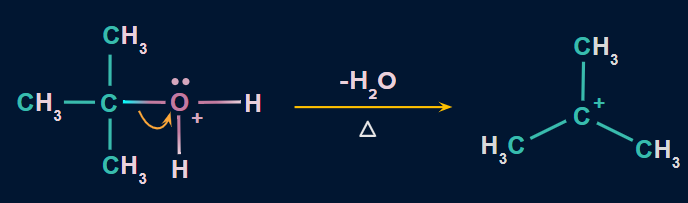
![]()
Step 3: Formation of alkene as a product
- Water attacks the proton of the carbon atom next to the carbocation, breaking the existing C−H bond and forming C=C.
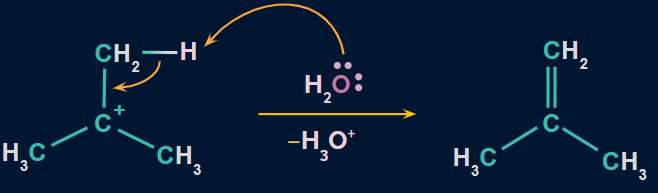

Order of reactivity of alcohols towards dehydration:
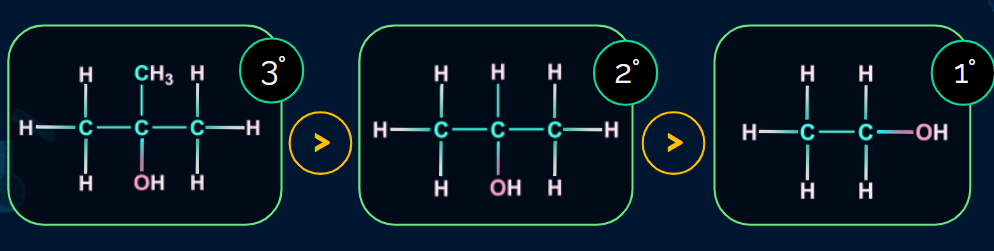

Practice Problems:
Q1. Which of the following compounds will show the highest heat of combustion?
A. 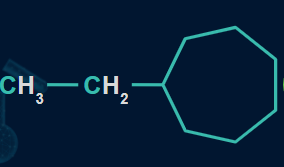
B. 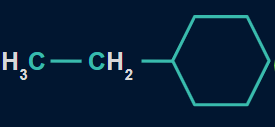
C. 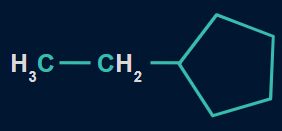
D. None of the above
Answer: A
Solution:
In the given structures, the ethyl group is common but the ring size varies from the seven-membered ring in structure (I), the six-membered ring in structure (II), to the five-membered ring in structure (III).
As the number of carbon atoms decreases, the heat of combustion also decreases.
So, option A is the correct answer.
Q2. The salt of an unknown carboxylic acid forms cyclobutane on Kolbe’s electrolysis. What can be the carboxylic acid?
- Hexanoic acid
- Succinic acid
- Adipic acid
- Fumaric acid
Answer: C
Solution:
A carboxylic acid having six carbon atoms is known as adipic acid or hexanedioic acid that forms cyclobutane by Kolbe’s electrolysis method.
Starting from the salt of adipic acid,
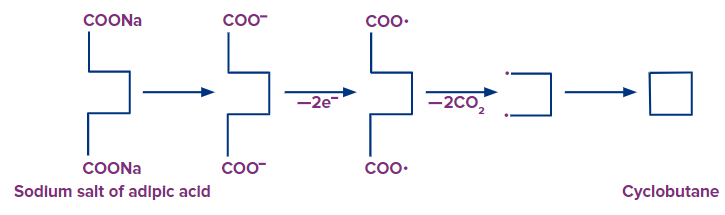
Hence, option (C) is the correct answer.
Q3. Iodoethane and iodopropane are allowed to undergo the Wurtz reaction. Find the alkane that can not be obtained in this reaction?
- Butane
- Propane
- Pentane
- Hexane
Answer: B
Solution:
Wurtz reaction takes place when two moles of alkyl halide react with Na in the presence of dry ether to give alkane.
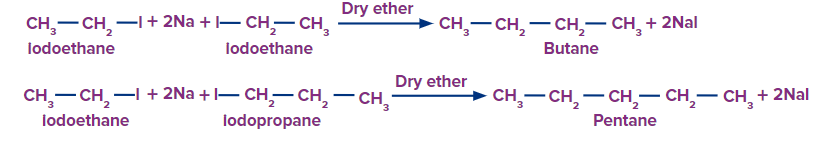

Therefore, option (B) is the correct answer.
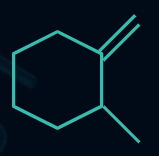
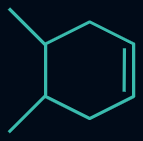
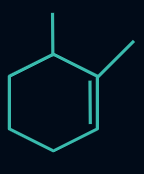
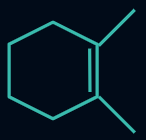
Q4. Which of the following compounds will evolve the most heat upon the catalytic hydrogenation with H2 ?
Answer: A
Compounds (I), (II), (III), and (IV) have three, four, six, and ten α-hydrogens, respectively. The more the α-hydrogens, the more will be the number of hyperconjugative structures, and hence more stable will be the alkene. Thus, less heat of hydrogenation will be evolved.
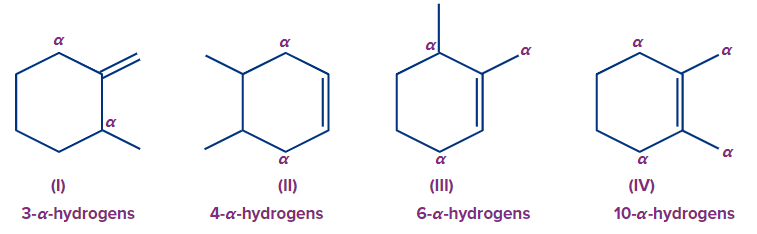
So, the order of heat evolved will be as follows:
(I) > (II) > (III) > (IV)
Frequently Asked Questions-FAQs:
1. What are the main sources of alkanes?
Answer: Petrol, Diesel, and LPG are obtained from the fractional distillation of crude oil, and all of these fuels are primarily composed of alkanes.
2. Write any three methods of preparation of alkanes.
Answer: There are various methods of preparation of alkanes. Some of them are the Wurtz reaction, Decarboxylation, and Kolbe’s electrolysis method.
3. Name a catalyst used in the hydrogenation of alkene.
Answer: Mainly Nickel is used as a catalyst but there are some other metals such as platinum, palladium, and rhodium that can also be used for hydrogenation.
4. Write any three methods of preparation of alkenes.
Answer: There are various methods of preparation of alkenes. Some of them are the Dehydration of alcohols, Dehydrohalogenation, and the Dehalogenation method