-
Call Now
1800-102-2727
Physisorption vs Chemisorption: Definition, Types of Adsorption, Characteristics and Difference Between Physisorption and Chemisorption, Practice Problems & FAQs
You want to go to work quickly.
You sigh as you see your car windows are all frozen after hitting the snooze button a couple of too many times.
You complain about having to scrape the windows and consider the source of their frost.
Well, there are water molecules in the air, which is why your windows accumulate a thin layer of frost on cold or rainy days. The adsorption of water molecules to the glass is an example of physisorption. These molecules adhere to the glass's surface after being drawn to it by the glass. Water molecules are constantly present, but when the temperature drops too low, they freeze, causing your windows to frost. Adsorption is the process by which molecules adhere to the surface of a solid.
On this page, we will study more about adsorption, types and their characteristics.
Table of Contents
- Definition of Adsorption
- Types of Adsorption
- Characteristics of Physisorption
- Characteristics of Chemisorption
- Differences between Physisorption and Chemisorption
- Practice Problems
- Frequently Asked Questions(FAQs)
Definition of Adsorption:
The act of atoms/ions/molecules of one substance adhering to the surface of another is known as adsorption. The phenomenon of adsorption of getting attracted and accumulating onto a liquid surface or a solid surface, resulting in a larger concentration of the molecules on the surface.
Adsorbate: The material that absorbs onto any surface is referred to as an adsorbate. The term "adsorbate" refers to a gas that has been adsorbed onto a solid surface, for example.
Adsorbent: An adsorbent is material on whose surface adsorption occurs. Adsorbents come in both solid and liquid forms. The most often utilised adsorbents are metal powders, charcoal powder, and silica powder. Due to their huge surface areas, alumina, silica, and activated charcoal are all very efficient adsorbents.
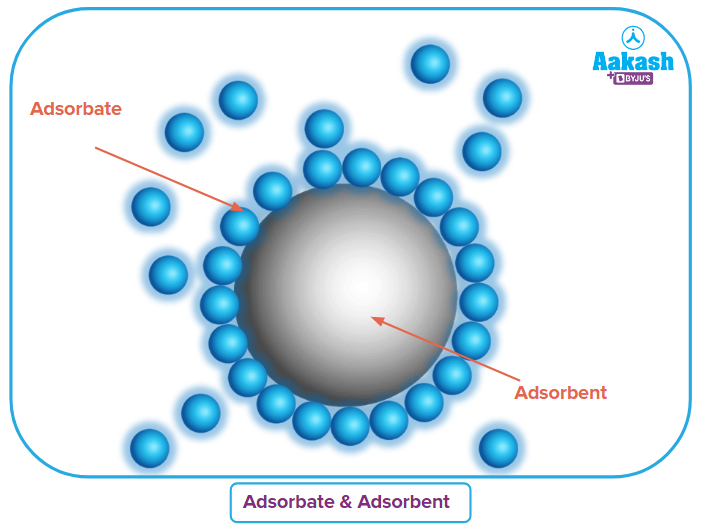
Types of Adsorption:
Absorption is categorised into two forms based on the nature of the force that exists between the adsorbate and adsorbent molecules:
1. Physisorption or physical adsorption and
2. Chemisorption or chemical adsorption.
Physisorption or physical adsorption: Van der Waals adsorption, physical adsorption, or physisorption are all terms used to describe the absorption process. The adsorbate is kept on the surface of the adsorbent by weak forces of the van der Waals force.
Example1: Gas adsorption on animal charcoal.
Example2: Water vapour adsorption on silica gel.
Chemisorption or chemical adsorption: Adsorption is referred to as chemical adsorption or chemisorption if the adsorbate is kept on the surface of the absorbent by a strong chemical interaction.
Example: Iron rusting.
Characteristics of Physisorption:
No chemical bonds are created in this. It is only a physical change. As a result, it is referred to as physical adsorption.
Physisorption is non-specific because van der Waals forces are general and not related to any specific type of substance.
Stronger van der Waals forces of attraction exist for gases that are easily liquefiable.
Multilayers of adsorption are possible in physisorption.
In most cases, physisorption is reversible. It can be reversed by either raising the temperature or lowering the pressure.
Although the process of physisorption is exothermic, the heat of adsorption involved is comparatively small, ranging between 20 and 40 kJ/mol.
Van der Waals forces and physisorption's non-specific nature cause it to be multi-layered.
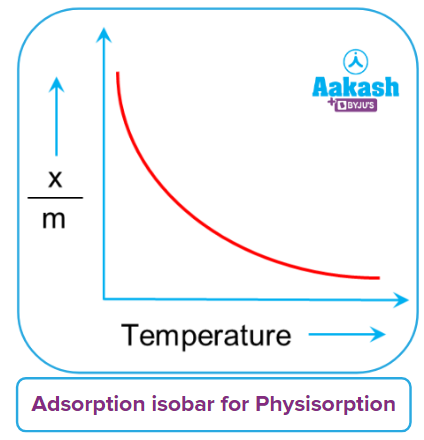
The temperature has a direct effect on absorption, but only for physical adsorption. Adsorption decreases with increasing temperature. Adsorption is inversely proportional to temperature.
Adsorbent (s) + Adsorbate (g) ⇌ [Adsorbent---Adsorbate] + ΔH
The adsorption isobar curve can be used to express how the amount of gas absorbed by the adsorbent changes with temperature. It illustrates the relationship between xm and temperature under a constant pressure. Here, x is the mass of the adsorbed gas, m is the mass of the adsorbent, and xm is the mass of the adsorbed gas as a percentage of its mass.
With increasing temperatures, physical adsorption gradually decreases or the gas is removed from the surface.
Characteristics of Chemisorption:
Between the adsorbent and the adsorbate, a chemical bond is created in this process.
Chemisorption is extremely specific since it only takes place when a chemical bond can form between the adsorbent and the adsorbate.
Adsorbate should be capable of forming a chemical bond with the adsorbent.
Chemisorption is often irreversible due to irreversible chemical change in most situations.
Chemisorption is an exothermic process with an adsorption heat of 40-400 kJ/mol.
A unimolecular layer is formed as a result of chemisorption. The chemical bond is established only when the layer of molecules comes into direct contact with the adsorbent's surface.
The adsorption isobar curve can be used to express how the amount of gas absorbed by the adsorbent changes with temperature. It illustrates the relationship between xm and temperature under a constant pressure. Here, x is the mass of the adsorbed gas, m is the mass of the adsorbent, and xm is the mass of the adsorbed gas as a percentage of its mass.
Any chemical reaction needs the reactant to have the energy to be greater than its activation energy to form products. Initial heating helps the molecules to acquire the activation energy necessary for the reaction. So the reaction and hence the chemisorption increases with increasing temperature, initially. Since these chemisorptions are highly exothermic, according to Lechatlier’s principle, further increases in temperature decrease the chemisorption. Hence, chemical adsorption initially reaches a maximum and then steadily decreases. The heat that is delivered acts as activation energy during the initial increase in chemisorption, and as more and more adsorbate molecules gather energy, they eventually surpass the activation energy. As a result, adsorption decreases with increasing temperature.
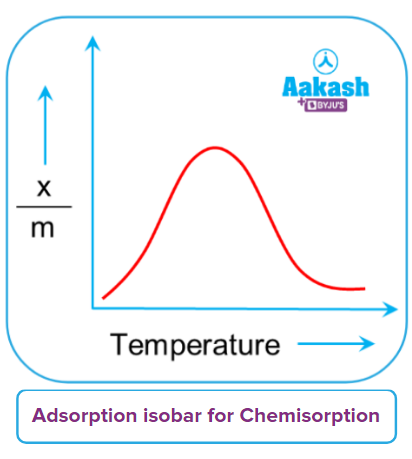
Chemisorption increases as pressure increases. At greater pressures, the gas molecules approach the adsorbent's surface and support the creation of chemical bonds.
Differences between physisorption and Chemisorption:
The forces that interact between the adsorbent and the adsorbate divide adsorption into two categories.
|
Parameter |
Physical adsorption |
Chemical adsorption |
|
Forces |
The adsorbate and adsorbent are held together by weak van der Waals forces. |
The adsorbate and adsorbent are held together by strong chemical bonds of ionic or covalent or hydrogen bonds |
|
Strength |
In comparison to chemical adsorption, physisorption is a weak phenomenon. |
Chemical adsorption is an extremely strong phenomenon. |
|
Layer |
This process involves multiple layers. |
This process involves a single layer. |
|
Occurrence |
Physical adsorption is non-specific and happens everywhere on the adsorbent. |
Chemisorption is particularly specific and happens at reaction centres on the adsorbent. |
|
Factors affecting |
Pressure, surface area, temperature and adsorbate type affect Physisorption |
Chemisorption is influenced by surface area, temperature, and the type of adsorbate. |
|
Activation energy |
Low energy (20–40 kJ/mol) is required for activation. |
The activation energy ranges from 40 to 400 kJ/mol. |
|
Reversibility |
Easily reversible |
Irreversible |
|
Example |
Gas adsorption on animal charcoal. Water vapour adsorption on silica gel. |
Adsorption of hydrogen, nitrogen, oxygen and other gases on metals. |
Practice Problems:
Q1. Adsorption is always
(A) Exothermic
(B) Endothermic
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: There is always a decrease in the surface energy, or residual forces, which are clearly visible as heat, as a result of adsorption. Heat is thus released during adsorption (exothermic process). This means that adsorption is always exothermic(H <0).
Q2. For the process of adsorption change in entropy is
(A) Less than zero
(B) Greater than zero
(C) Equal to zero
(D) Greater than or equal to zero
Answer: (A)
Solution: With adsorption, the amount of randomness in the adsorbate particles reduces as they are bonded to the surface or when their freedom is restricted. Entropy decreases as a result (S <0).
Q3. The term "sorption" is used when
(A) Absorption takes place
(B) Adsorption takes place
(C) Both adsorption and absorption take place
(D) Desorption takes place
Answer: (C)
Solution: When both adsorption and absorption occur, the term "sorption" is used. Although the material is evenly distributed throughout the majority of the solid, its concentration is higher near the surface than it is throughout.
For example: In fabric dyeing, the colours on clothes will undoubtedly fade after a certain number of washes if they are only superficially absorbed. Both the adsorption and absorption processes take place at the same time when dying fabrics to avoid this problem.
Q4. Which one of the following forces primarily contributes to chemical adsorption?
(A) Van der Waals forces
(B) Chemical bonds
(C) Gravitational force
(D) Electromagnetic force
Answer: (B)
Solution: When chemical forces of attraction or chemical bonds are present between the adsorbent and the adsorbate, the process of chemical adsorption, also known as chemisorption, takes place.
Frequently Asked Questions(FAQs):
Q1. Describe the differences between adsorption and absorption.
Answer:
|
Parameter |
Adsorption |
Absorption |
|
Definition |
Adsorption is the process through which material accumulates on a surface in molecular species at greater concentrations. |
Absorption is the total incorporation of one material into the bulk of another. |
|
Phenomena |
It is a surface phenomenon. |
It is a bulk phenomenon. |
|
Molecular Interaction |
When compared to absorption, the contact between molecules during the adsorption process is comparatively low. |
The interactions between molecules is relatively significant during the absorption process. |
|
Example |
When placed in a watery environment, silica gel absorbs the water. There is a large concentration of water on the surface of the silica gel. |
Calcium chloride, on the other hand, absorbs water when exposed to moisture. The liquid is evenly distributed. |
Q2. Define Freundlich adsorption isotherm.
Answer: The extent of adsorption is correlated with pressure at a constant temperature according to the Freundlich adsorption isotherm.
Where
K & n=adsorption constant
The major drawback of the Freundlich adsorption isotherm is that it fails at high pressure. It was unable to explain the multi-layered adsorption mechanism.
Q3. Why is physical adsorption not specific in nature?
Answer: Physical adsorption, also known as physisorption, is the term for adsorption in which the adsorbate particles are held to the surface of the adsorbent by physical forces such as van der Waals forces.
The molecules of the adsorbent and adsorbate are weakly attracted to one another. According to Le-principle, Chatelier's physical adsorption is more common at low temperatures and less common at higher ones. As the temperature rises, the gas molecules' kinetic energy increases, causing them to leave the surface of the adsorbent.
Physisorption lacks specificity since it is non-specific because all gases are equally adsorbed on all solids. Adsorbent does not exhibit any specific preference for an adsorbate, which can be explained by the fact that van der Waals forces are present everywhere and do not favour any specific gas on any given surface of an adsorbent.
Q4. Define Enthalpy of adsorption (∆HAdsorption).
Answer: Enthalpy of adsorption (∆HAdsorption) is the amount of heat absorbed when 1 mol of an adsorbate is adsorbed on a certain adsorbent at equilibrium with adsorption. It depends on the properties of the adsorbent and adsorbate. Adsorption is the process of attracting molecules of adsorbate to the adsorbent's surface. As a result, energy is released, and the heat of adsorption is negative, implying that adsorption is always exothermic.


