-
Call Now
1800-102-2727
Ozonolysis of Alkynes- Ozonolysis, Mechanisms, Types, Practice Problems and FAQs
Cancer is the highest human killer of today and effective anticancer drugs are desperately being developed.. Taxol, from a bark of a Yew tree contains a diketone, is a highly effective anticancer medication used to treat solid tumour malignancies of the breast, ovary, lung, bladder, prostate, melanoma, oesophagus, and other organs.
Let's talk about the process known as ozonolysis, which allows us to convert an alkyne molecule into a diketone molecule.
Table of Contents
- Ozonolysis Reaction
- Alkynes ozonolysis
- Mechanism
- Practice Problems
- Frequently Asked Questions
Ozonolysis Reaction
Ozone is an extremely reactive isotope of oxygen. In the presence of ozone molecules, unsaturated substances undergo oxidative cleavage. An intermediate complex known as ozonide is created when the compound's unsaturated bonds are switched out for C-O bonds. To eliminate oxygen, zinc dust is applied to the intermediate ozonide molecule in case of reductive ozonolysis. Oxygen being taken out of the ozonide causes a reductive process that creates carbonyl molecules.
Alkynes ozonolysis
Alkynes having two pi bonds are subjected to ozonolysis, with the end result being either an acid anhydride or a diketone. In this reaction, the fragmentation is not fully complete (alkenes undergo complete fragmentation). No reducing agents are needed because a straightforward aqueous workup can be used. If water is present during the reaction, the acid anhydride goes through hydrolyzation and yields two carboxylic acids. Another application of ozonolysis is to locate the triple bond in an unknown alkyne.
Let's examine the ozonolysis reaction mechanism that occurs in an alkyne.
Mechanism of ozonolysis reaction of alkyne
The reaction mechanism of ozonolysis involves three steps.
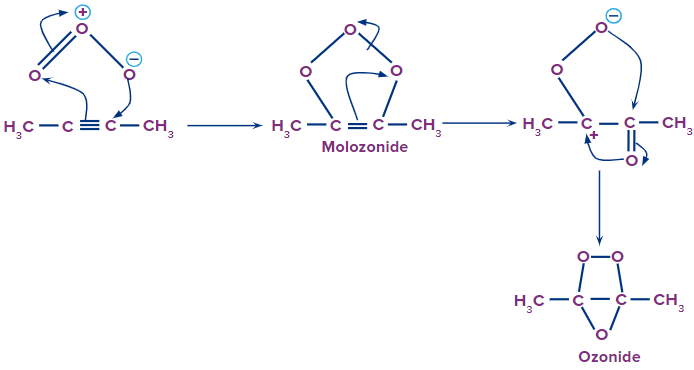
Step 1: Attack of the ozone molecule
There are three oxygen atoms in the ozone molecule. Both of the oxygen atoms change their partial charges, one becoming partially negative and the other partially positive. The triple-bonded carbon atom is attacked by the negatively charged oxygen ion. The reactant picks up the ozone molecule. Within the structure, there is a rearrangement that produces the molozonide, an intermediary structure.
Step 2: Formation of ozonide intermediate
A stable ozonide intermediate is created as a result of further self-rearrangement of the molozonide intermediate. We can observe that the triple bond is broken down into a single bond in ozonide. Later, a new carbonyl molecule is produced when the single bond between two carbon atoms also breaks.

Step 3: Formation of carbonyl compounds
It can be performed by two types of mechanisms, which are given as follows:
- Reductive ozonolysis
- Oxidative ozonolysis
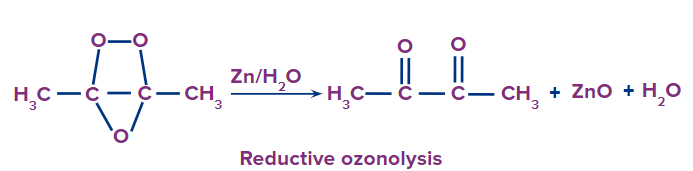
Reductive ozonolysis
This type of ozonolysis of alkyne involves the addition of an ozone molecule to an alkyne to form an ozonide. Ozonide is unstable and decomposes easily by reduction into , -diketones. The reagent that is used in reductive ozonolysis is given as follows:
(i) Zn and H2O
(ii) (CH3)2S and H2O
(iii) CH3COOH
The formation of , -diketones can be given as:
Oxidative Ozonolysis
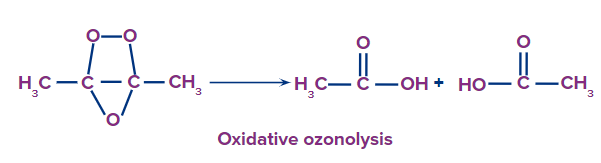
In this kind of ozonolysis, an ozone molecule is added to an alkyne to create an ozonide, which is then oxidized to produce smaller oxidized molecules like acid. The following is a list of the reagent used in oxidative ozonolysis:
(i) H2O
(ii) H2O2
The formation of carboxylic acids can be given as:
In oxidative ozonolysis, if formic acid is formed, it will further decompose into carbon dioxide and water
HCOOHCO2+H2O
Practice Problems
1. What should result from treating cyclopentyne with ozone and then treated with hydrogen peroxide and (dimethyl sulphide, and water) respectively?
- Cyclopentane-1,2-dione, Cyclopentane-1,5-dioic acid
- Cyclopentane-1,5-dioic acid, Cyclopentane-1,2-dione
- Cyclopentane-1,2-dione, Cyclopentane-1,2-dione
- Cyclopentane-1,5-dioic acid, Cyclopentane-1,5-dioic acid
Solution: When cyclopentyne is treated with ozone in the presence of Hydrogen peroxide (O3 and H2O2), this is oxidative ozonolysis which involves the addition of an ozone molecule to an alkyne to form an ozonide, followed by the oxidation to form a carboxylic acid. So, the product should be Cyclopentane-1,5-dioic acid.
When cyclopentyne is treated with ozone in the presence of dimethyl sulphide and water ((CH3)2S and H2O), this is reductive ozonolysis which involves the addition of an ozone molecule to alkyne to form an ozonide, decomposes easily by reduction into , -diketones. So, the product should be Cyclopentane-1,2-dione.
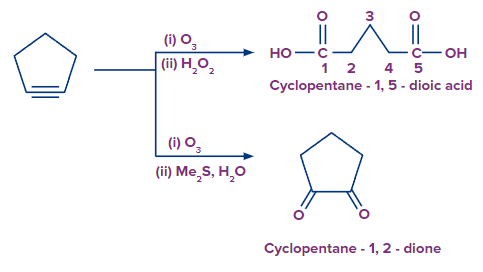
So, the correct answer should be (B).
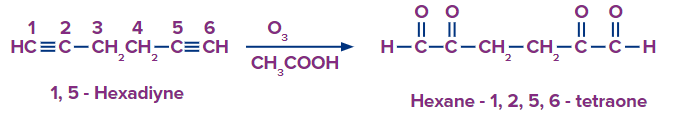
Q2. Hexane-1,2,5,6-tetraone is formed from a reagent X treated with ozone and acetic acid. What is the possible IUPAC name of X?
- 1,3-Hexadiyne
- 1,4-Hexadiyne
- 2,4-Hexadiyne
- 1,5-Hexadiyne
Solution: This type of alkyne ozonolysis involves adding an ozone molecule to an alkyne to form an ozonide. Ozonide is unstable and quickly decomposes with acetic acid by reduction into -diketones. Here, when reagent X is treated with O3 and CH3COOH, the product formed is Hexane-1,2,5,6-tetraone. The reagent should be 1,5-Hexadiyne. Hence, the correct answer is option (D).
Q3. Compounds A, B, and C are created during the ozonolysis of a chemical in a laboratory. When sodium bicarbonate reacts with molecule A, which has the chemical formula C3H6O2 , it produces the colourless gas D with effervescence. B and D are the same compounds. Decide on the beginning reagent's IUPAC name and the type of ozonolysis.
- But-1-yne, oxidative ozonolysis
- Propa-1,2-diene, reductive ozonolysis
- Propyne, oxidative ozonolysis
- Propyne, reductive ozonolysis
Solution: A colourless gas D with effervescence is produced by treating chemical A, which has the molecular formula C3H6O2 , with sodium bicarbonate. This is a test to analyse if the carboxylic acid group is present. Therefore, propanoic acid (CH3CH2COOH) should be compound A . Carbon dioxide is the gas released during this test. Therefore, substances B and D are CO2. If the end result is a carboxylic acid, ozonolysis must be an oxidative process. Since chemical B is CO2, formic acids are required to produce it. The proposed system is 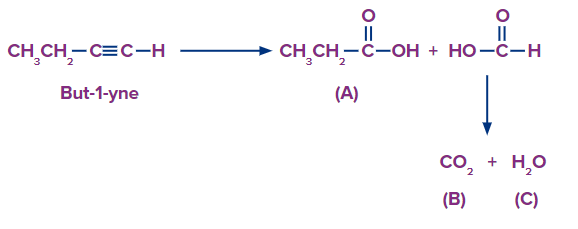
So, the correct answer is an option (A).
4. Predict the IUPAC name of starting reagent and the nature of ozonolysis while doing ozonolysis of a compound, compounds P, Q and R are formed. The compound P with molecular formula C2H4O2, when treated by basic salt gives a colourless gas, S with effervescence. The compounds Q and S are the same.
- But-1-yne, oxidative ozonolysis
- Propa-1,2-diene, reductive ozonolysis
- Propyne, oxidative ozonolysis
- Propyne, reductive ozonolysis
Solution: The compound P with molecular and formula C2H4O2 is treated by basic salt that must be sodium bicarbonate test to give a colourless gas Q with effervescence. This is a test for the presence of the carboxylic acid group. So, compound P should be Acetic acid (CH3COOH). The gas released during this test is carbon dioxide. Hence the compounds Q and Sare CO2. If the product is a carboxylic acid, then the nature of ozonolysis must be oxidative. The compound Q is CO2, then it must be formed from formic acids. The suggested mechanism is
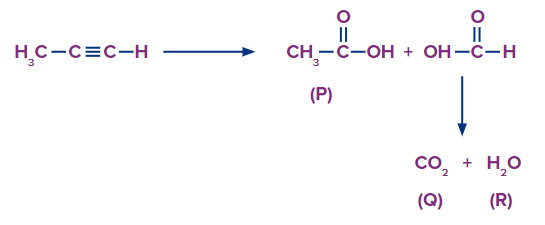
So, the correct answer is an option (C).
Frequently Asked Questions
1. What chemical uses does ozonolysis have?
Answer: The following are the applications of ozonolysis: In organic chemistry, ozonolysis is used to locate double and triple bonds in alkenes and alkynes. Long alkenes and alkynes can be structurally analysed via ozonolysis.
2. Is alkyne basic or acidic?
Answer: Alkynes are acidic because they have the ability to liberate hydrogen atoms to create alkyne ions. The alkyne is therefore employed as Brönsted-Lowry acid. Alkynes, as previously mentioned, have a sp-hybrid carbon atom, which is triple-bonded.
3. What are the industrial uses of ozonolysis?
Answer: Ozonolysis is used in bleaching. Water treatment processes employ ozonolysis. Alcohols, carboxylic acids, aldehydes, and ketones are also created through ozonolysis.
4. What function does zinc perform in ozonolysis?
Answer: In ozonolysis, zinc dust is employed to stop the compound from further oxidation. The reaction is put on hold by Zn because it stops the molecule from making more oxygen bonds.
5. What do you mean by ozone crack?
Answer: The ozone attack on the elastomer, which is present in tiny levels in the atmosphere, causes fissures. The ozone here attacks the double bond in the rubber chain. Ozone cracks appear if the rubber product is under tension. Since the cracks are placed at a right angle to the axis of strain, they can develop all the way around a bent rubber tube. When they develop in fuel pipelines and grow inward from the exposed outer surface, these cracks are quite harmful. Even gasoline leaks and fires may result from this.


