-
Call Now
1800-102-2727
Crystal Defects-Nonstoichiometric: Non Stochiometric Defect, Metal Excess Defect, Metal Deficiency Defect, Definition of Impurity Defect, Calculation of Cation Vacancies, Practice Problems and FAQs:
Have you heard of odd man out phrase? It indicates some unconnected outsider present in a group of like people.. This odd man may either add some new property or entirely change the nature of the group.

Similarly,r some defects may change the stoichiometry of the crystal formula and nature. These defects are called nonstoichiometric defects.
Have you ever tasted a salt mixed with a bit of sugar? The taste would be entirely different and hence sugar will be treated as an impurity in salt.
In a similar manner impurity defects arises when foreign atoms are present at the lattice site in place of host atom or at the vacant interstitial site (just like sugar granule in place of salt).
Technically We can understand this like in case of steel, which is carbide of iron. C atom are present at the interstitial sites in the lattice of Fe atoms.
Table of content
- Non-Stoichiometric defect
- Metal excess defect
- Metal deficiency defect
- Impurity defect
- Frequently Asked Questions
Defects in crystal
Any departure from a perfectly ordered arrangement of constituent particles in a crystal is called imperfection or defect.
Any change or deviation from the expected perfect crystal structure made by the constituent atoms, ions or molecules is considered as a defect in the crystalline structure.
There are two types of defects
- Point defect
- Line defect
Point defect
When deviations or irregularities exist from the ideal arrangement around a lattice point of the constituent atom/ion/molecule in a crystalline substance, the defect is called point defect.
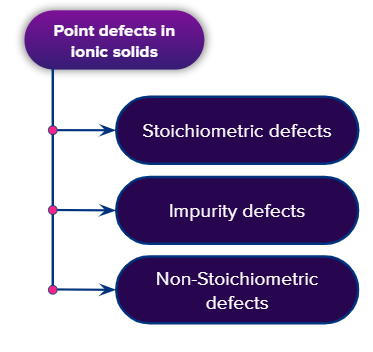
Point defects in a crystal may be considered in the following three categories
- Stoichiometric defects
- Non-stoichiometric defects
- Impurity defects
Non-Stoichiometric Defects
If, the imperfections in the crystal, results in the change of the ratio of the cations to the anions and hence the ideal chemical formula, the defects are named as non-stoichiometric defects.
The non-stoichiometric effect may be caused by metal excess or metal deficiency or presence of foreign material called as impurity.
-
Metal excess defect:
Metal-nonmetal ratio may become larger under two circumstances, One is due to the preferential loss of anions or by the excess addition of cations.
|
By anion vacancy |
By the presence of extra cations |
|
A hole is created due to the missing of a anion. Now the hole is occupied by an electron to maintain electrical neutrality. The sites containing the electron thus trapped in the anion vacancies are F-centres. They are responsible for imparting colour to the crystal. |
An extra cation occupying the interstitial site may also contribute to metal excess. Electrical neutrality is maintained by an electron that is present in another interstitial site. Cations more than the stoichiometric amounts and present in the interstitial crystal sites result in metal excess defect.. The additional cation charges will be nullified by the presence of additional electrons in some other interstitial sites of the crystal structure. |
|
Example: when NaCl is heated with sodium vapour, deposition of excess of Na atoms occurs on the surface of NaCl crystal. When heated with sodium vapour, NaCl, deposition of excess of Na atoms gets deposited on the surface of NaCl crystal. Diffusion of Cl- ions occurs to the surface where they combine with Na atoms which become ionised by losing electrons. These electrons absorb the energy of the white light, giving yellow colour to NaCl. Na atoms who ionize by losing electrons Energy is adsorbed and a complimentary yellow colour is released by these electrons. |
Example: when ZnOis heated, it loses oxygen and turns yellow On heating, oxygen is released from ZnO giving a yellow colour to the crystal. ZnOZn2++12O2+2e- Zinc ions and the electrons occupy the interstitial sites and thus making the crystal still neutral the excess of Zn2+ ions this formed gat trapped into the vacant interstitial sites and the electrons in the neighbouring interstitial sites. |
|
In crystals with Schottky defects, this defect can be found. |
In crystals with Schottky defects, this defect can be found. |
-
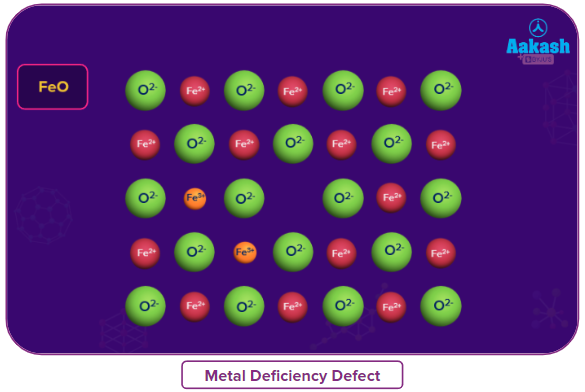
Metal deficiency defect
This defect occurs when the metal shows variable valency. This defect usually occurs due to the missing of a cation having higher charge in adjacent lattice site.
Examples: FeO, FeS and NiO. Due to metal deficiency defect, the compounds are non-stoichiometric.
- FeO is found, mostly, with a composition of Fe0.95O, loss of some Fe2+ions is compensated by presence of required number of Fe3+ions
-
Impurity defect
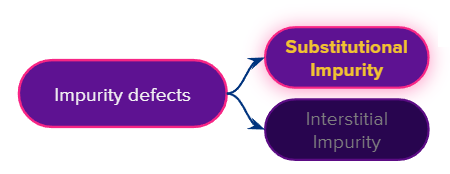
An impurity defect is a crystal lattice distortion caused by an impurity (foreign atom/ion) occupying an interstitial site in the lattice or replacing the parent atom/ion in the regular sites.
Impurity defect can be classified into the following type that is
- Substitutional impurity
- Interstitial impurity
Substitutional impurity
Defects in ionic crystals, can be introduced by adding impurities. Similar sized cation substitute the existing cation of ionic crystal.
Example: When molten NaCl is crystallized having a small amount of SrCl2, Some Na+ ions’ locations are occupied by Sr2+ ions Each Sr2+ replaces two Na+ sites by occupying a site of one Na+ and other site remaining vacant.
When molten NaCl is crystallized having a small amount of SrCl2, some Na+ ion’lattice points are replaced with Sr2+ ions. Each divalent Sr2+ ion empties two Na+ sites. One such empty site will be occupied by strontium ion leaving the other site vacant.


Number of cationic vacancies generated = Number of Sr2+in the crystal
Other example:solid solution of CdCl2 and AgCl.
Interstitial Impurity Defect
Here, some small foreign atoms (like B, C, N, H) are trapped in interstitial voids of the lattice without any chemical reaction.
Doping is the addition of impurities to a crystalline substance in order to alter its properties.
1. Introducing impurity defect in ionic solids
- In this case the impurities are introduced by adding ions as impurity.. Vacancies are produced when the impurity ions are in a different valence state than the host ions.
Presence of ions other than that of the crystal ions will create impurity defect in ionic solids. If the other ions are of different valency from the crystal ions , then they will create vacancies related to the difference in valency in the crystal
- If a molten NaCl ,containing a little SrCl2 as impurity is allowed to cool, in the crystals of NaCl formed, at some lattice sites, Na+ ions are substituted by Sr2+ ion. For every Sr2+ion thus introduced, two Na+ions are removed to maintain electrical neutrality. One of these lattice sites is occupied by Sr2+and remains vacant. These vacancies will increase electrical conductivity of the solid. Similar defect and behavior is observed when CdCl2is added to AgCl.
2. Introducing impurity defect in covalent solids
In case of covalent solids such as silicon germanium (group 14 elements) which have 4 valence electron in their outermost shell, the impurities added may be of the elements which may have more than 4 valence electrons (for example Group 15 elements like P or As which have five valence electrons) or of the elements which have less than 4 valence electrons (for example Group 13 elements like Ga which have three valence electrons).
Thus, the impurities added may be electron rich or deficit. The defects thus introduced in the crystals are called electronic defects.
a) Doping with electron rich impurities-
- Group 14 elements like silicon or germanium have four electrons in outermost orbit. Hence, it normally forms four covalent bonds with the neighboring atom.
- When the crystal is added with any of the group 15 elements , the silicon or germanium atoms at a lattice sites are substituted by atoms of P or As. Now, as these atoms have 5 electrons in the valance shell after forming normal four covalent bonds with the neighboring silicon atoms, the fifth extra electron and gets delocalized. These delocalized electrons increase the conductivity of silicon or germanium.
Group 14 elements have four electrons in their outermost orbit. Consider, a group 14 element in a crystal structure is replaced by any of the elements of 15 group which have five electrons in their outermost orbit . Group 15 element will use only four of its electrons for bonding and the fifth electrons is completely free to move and conduct electricity of the group14 elements of silicon and germanium.
- As the increase in conductivity is due to negatively charged electrons, the silicon or germanium crystals doped with electron rich impurities are called n-type conductors. higher electron containing elemental impurities are known as n-type conductors.
b) Doping with electron deficit impurities-
- When group 14 element like Si or Ge is doped with group 13 elements like B, Al, or Ga, the Si or Ge atom at some lattice are substituted by those of B, Al, or Ga.
- Now, as Group 13 elements have only 3 valence electrons, they can form three covalent bonds with the neighboring silicon atoms.
- Thus, a hole is created at the site where fourth electron is missing lost. This is called electron hole or electron vacancy. The vacancy is known as electron vacancy or a hole
- An electron with neighboring atom jump to fill up this electrons hole but then an electron hole is created at the site from where electrons jumped.
- As it continues, the electron holes will move in a direction opposite to that flow of the electrons.
As this jumping continues, electrons and holes will be moving in opposite directions
- Now, when an electric field is applied, the electrons move towards the positively charged plate and electron holes move towards the negatively charged plate as if they carry positive charge,
will move towards the cathode like a cation
Hence, silicon and germanium doped with electron-deficit impurities are called p-type semiconductors.
Frequently Asked Questions
Q1. Can we say that all crystals have some defects?
Answer: There are defects in all crystals. Metals' mechanical characteristics are influenced by defects. In reality, calling these features "defects" is a misnomer because they are frequently employed to modify a material's mechanical properties.
Q2. Why isn't FeO (s) created in a stoichiometric composition?
Answer: as Fe2+ ions in FeO (s) are replaced by Fe3+ions. As 3 Fe2+ions are replaced by 2 Fe3+ions to maintain electrical neutrality, there’s less amount of metal as compared to stoichiometric proportion.
Hence, FeO (s) is not formed in stoichiometric composition.
Q3. Why does table salt (NaCl) occasionally turn yellow?
Answer: Yellow colour of NaCl is due to metal excess defect as some cl ions leave their lattice and are occupied by an unpaired electrons. These sites are celled F-centres. These electrons absorb energy from visible region for excitation so that complementary colour is yellow.
Table salt occasionally turn yellow. Give reasons.
Chloride ions in the table salt sometimes may leaves s chlorine leaving behind the electrons. This electrons are trapped in interstitial sites and absorb white light emitting a yellow colour
Q4. Is there any benefits of having defects in crystals?
Answer: Crystal defects have a variety of effects on the stress–strain diagram, including increasing tensile strength as the density of imperfections rises or partitioning of the crystal structure due to their shape. Imperfections play a role in work hardening and strain hardening.
Q5. How does crystal defects influence the properties of of materials?
Answer: Line defects degrade the structure in a one-dimensional space, and the type and density of the defects alter the solids' mechanical properties.


