-
Call Now
1800-102-2727
Molecular Mass of Polymers - Degree of Polymerisation, Stereochemistry of Polymers, Practice Problems and FAQs
Do you ever observe that when we go to market and ask for carry bags the shopkeeper packs the goods according to the purchase.
But why is it so?
As different bags have different strengths though the polymer material used in making these bags are the same. We know that monomers unit combine together to form polymer through the process known as polymerisation. Different polymers have different chain lengths and different strengths. These physicochemical properties depend upon important factors such as the molecular mass of a polymer and the degree of polymerisation. Both these parameters play a very vital role in the fields of pharmaceutical, biotech, and petroleum industries to name a few.
Let’s understand the definition of molecular mass of the polymer and the degree of polymerisation.

Table of Contents
- Molecular Mass of Polymer
- Polydispersity Index
- Degree of Polymerisation
- Stereochemistry of Polymers
- Practice Problems
- Frequently Asked Problems-FAQs
Molecular Mass of Polymers
The molecular mass of polymers can be expressed as number-average molecular mass (Mn) and mass-average molecular mass (Mm).
Number average molecular mass (Mn):
The number average molecular mass of the polymer is the arithmetic mean, which represents the total mass of the polymer molecules divided by the total number of molecules.
Number average molecular mass in a unit mass counts the total number of molecules of a polymer with out taking shape or size into consideration. All type of moilecules are considered indentical.
Colligative properties such as depression in freezing point, osmotic pressure and elevation in boiling point depend on the number of molecules present in the polymer and yield number average molecular mass.
Mathematically, it can be represented as:
![]()
If N1, N2, .........Ni represent the number of macromolecules with molecular masses M1, M2,.........Mi respectively.
Mass average molecular mass (Mw):
Contrary to number-average molecular mass, mass-average molecular mass is determined by methods that depend on the mass of individual molecules, viz., sedimentation, ultra-centrifuge and light scattering.
Mathematically, it can be represented as:
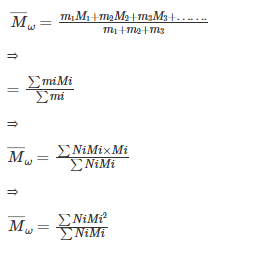
Here, m1,m2,m3....mi represent the mass of the molecules with molecular masses M1, M2, M3.........Mi respectively.
Now, it can be written that:
mi=NiMi
So, the formula of (Mw) can be written as:
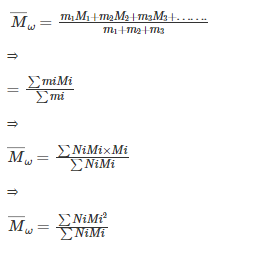
Where, N1, N2, N3 .........Ni represent the number of macromolecules.
Polydispersity Index (PDI)
The ratio of mass-average molecular mass to the number-average molecular mass is called polydispersity index (PDI). Polydispersity index is used to determine the broadness of molecular weight distribution. The larger the PDI, the broader the molecular weight distribution. As the PDI value rises, there will be increased variation in chain length, cross-linking, network formation, and branching. PDI = 1 denotes a monodisperse polymer. However, producing comparable Mw or Mn polymer chains is typically difficult.
The PDI for natural polymers is unity (Mw=Mn) since natural polymers are homogeneous (monodispersed). For polymers having PDI value equal to 1, the polymer chains in the polymer are of almost same chain length and same molecular weight.
The PDI for synthetic polymers, which are heterogeneous (polydispersed), is greater than unity as Mw is always greater than Mn.
Degree of Polymerisation
Degree of polymerisation is a measure of the number of monomeric units present in a polymer. It is the ratio of molecular mass of the polymer to the molecular mass of the monomer (or) the repeating unit.
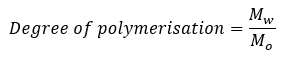
Where, Mw is the molecular mass of the polymer
Mo is the molecular mass of the monomer (or) the repeating unit
The degree of polymerization, which determines the length of the chains and is affected by the strength of the polymer. Higher degrees of polymerization correspond to stronger polymer qualities (such as mechanical strength and melting point), and these properties get stronger as the polymer's chain length gets longer.
Let us consider an example, the chain length of polyethylene, which is determined by the polymer's molecular weight, is directly correlated with the material's tensile strength.
Stereochemistry of Polymers
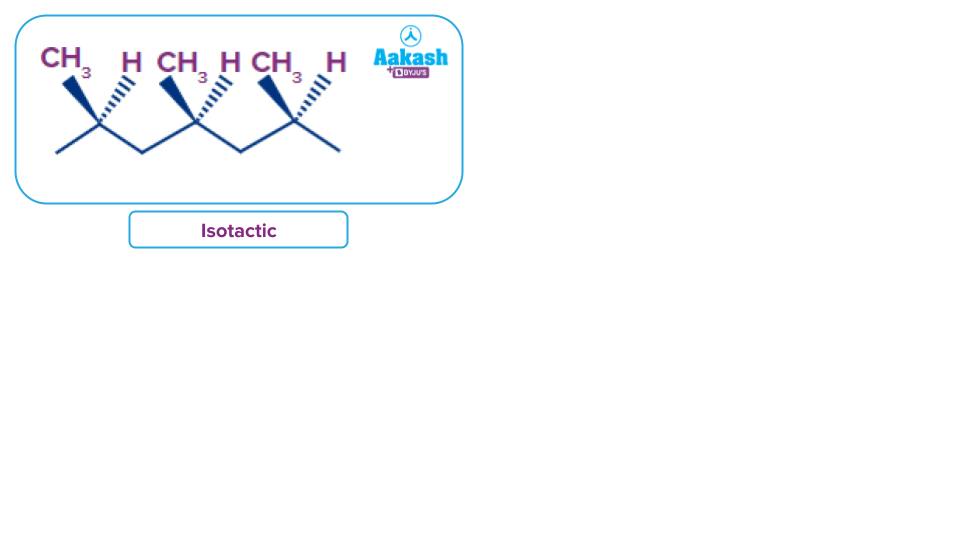
In addition to the influence of nature of atoms present in the polymeric chain, the size and cross-linking of chains also influence the physical properties of a polymer. For example, polyisoprene polymerises in any of the three below-given forms.
Isotactic: All the methyl groups of polypropylene exist on one side of the extended chain. It forms fibres as it is highly crystalline with a high melting point.
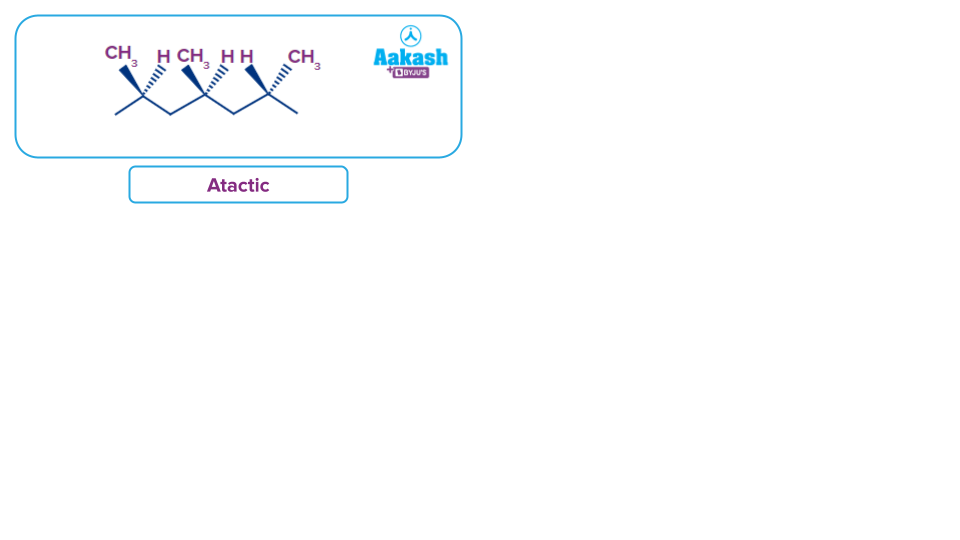
Syndiotactic: The methyl groups exist on the alternative sides of the extended chain.
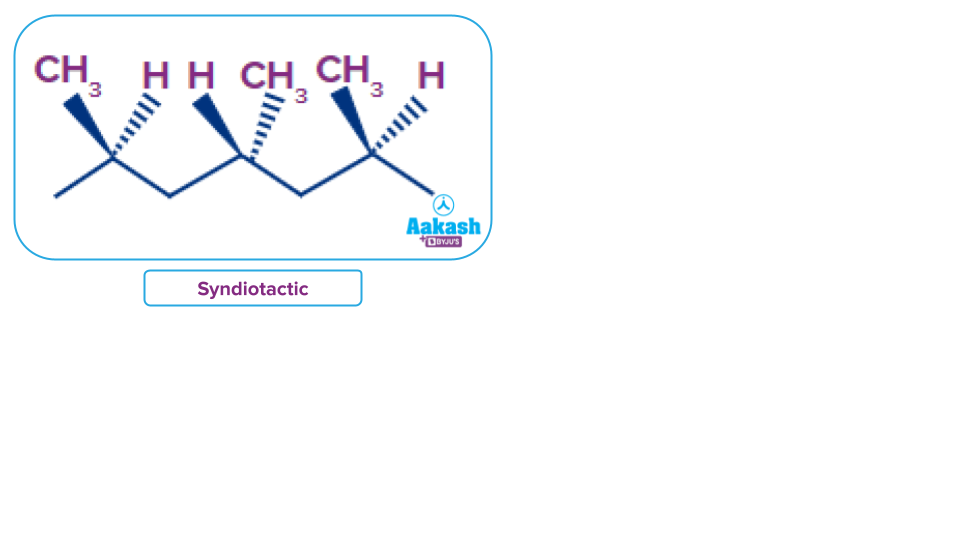
Atactic: The methyl groups exist randomly on the extended chain. It is soft, elastic and rubbery.
Practice Problems
Q. Match the following:
|
Column I |
Column II |
||
|
A) |
Number-average molecular mass |
P) |
PDI = 1 |
|
B) |
Mass-average molecular mass |
Q) |
Osmotic pressure method |
|
C) |
PDI of natural polymer |
R) |
PDI > 1 |
|
D) |
PDI of synthetic polymer |
S) |
Ultra-centrifuge and light scattering methods |
Answer: A) → Q) ; B) → S) ; C) → P) ; D) → R)
Solution:
|
Column I |
Column II |
||
|
A) |
Number-average molecular mass |
Q) |
Osmotic pressure method |
|
B) |
Mass-average molecular mass |
S) |
Ultra-centrifuge and light scattering methods |
|
C) |
PDI of natural polymer |
P) |
PDI = 1 |
|
D) |
PDI of synthetic polymer |
R) |
PDI > 1 |
Colligative properties such as depression in freezing point, osmotic pressure and elevation in boiling point depend on the number of molecules present in the polymer and yield number average molecular mass.
Mass-average molecular mass is determined by methods that depend on the mass of individual molecules, viz., sedimentation, ultra-centrifuge and light scattering.
The ratio of mass-average molecular mass (Mw) to the number-average molecular mass(Mn) is called polydispersity index (PDI).
The PDI for natural polymers is unity (Mw=Mn) since natural polymers are homogeneous(dispersed).
The PDI for synthetic polymers, which are heterogeneous (polydispersed), is greater than unity as Mw is always greater than Mn.
Q: The number-average and mass-average molecular mass of a polymer sample in which 30% molecules have a molecular mass of 20000, 40% have 30000 and the rest have 60000, respectively are:
- 36000, 43333.33
- 39000, 43333.33
- 43333.33, 36000
- 39000, 46333.33
Answer: (A)
Solution:
The formula for calculating the number-average molecular mass of polymers is:
The formula for calculating the mass-average molecular mass of polymers is:
So, option (A) is the correct answer.
Q. Syndiotactic polypropylene is the polymer in which the methyl groups are:
- Present on one side of the extended chain.
- Present on alternative sides of the extended chain.
- Present randomly on the extended chain.
- None of the above
Answer: (B)
Solution: In syndiotactic polypropylene polymer, the methyl groups are present on the alternative sides of the extended chain.
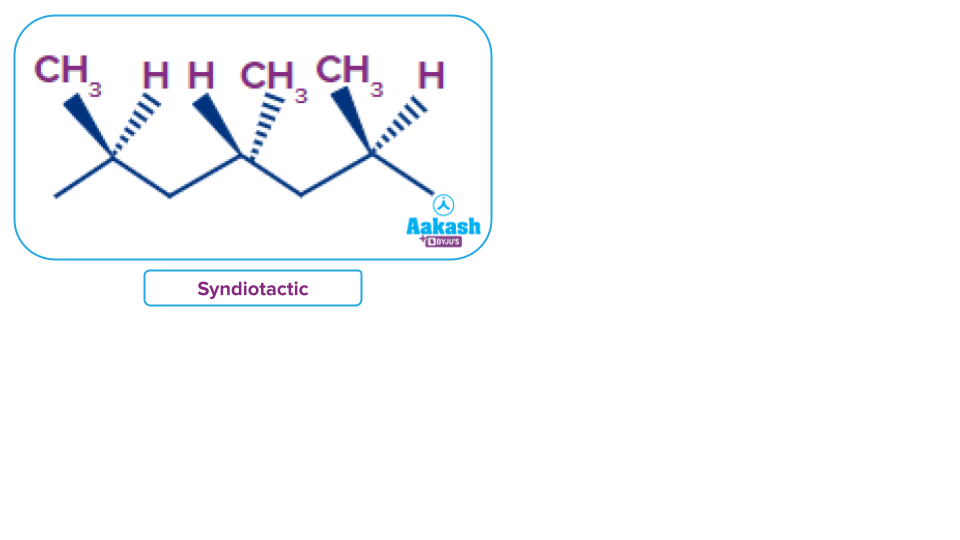
So, option B is the correct answer.
Q. Mass average molecular mass is determined by methods that depend on the __________.
- number of particles
- mass of the polymer
- mass of individual molecules
- All the above
Answer: (C)
Solution: Mass average molecular mass is determined by methods that depend on the mass of individual molecules, viz., sedimentation, ultra-centrifuge and light scattering.
So, option C is the correct answer.
Q. Number-average molecular mass is determined by methods that depend on the __________.
- number of molecules
- mass of individual molecules
- mass of the polymer
- All the above.
Answer: (A)
Solution: Colligative properties such as elevation in boiling point, depression in freezing point and osmotic pressure depend on the number of molecules present and determines number-average molecular mass. So, option A is the correct answer.
Q. Degree of polymerisation is given by the formula:
- Mw✕ Mo
- (Mw✕ Mo)/Mw
- Mw/ Mo
- Mo/ Mw
Answer: (C)
Solution: It is the ratio of molecular mass of the polymer to the molecular mass of the monomer (or) the repeating unit.
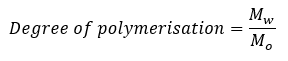
Where, Mw is the molecular mass of the polymer
Mo is the molecular mass of the monomer (or) the repeating unit
So, option C is the correct answer.
Frequently Asked Questions - FAQs
Q. What will happen when the degree of polymerisation increases?
Answer: Degree of polymerisation relates to the mechanical strength of polymer. Higher the degree of polymerisation represents higher strength properties of polymer (like higher mechanical strength, higher melting temperature) which increases with increase in the chain length of the polymer.
Q. How can we control the molecular mass of polymer in free radical polymerisation?
Answer: Controlling the molecular mass in free radical polymerisation can be done by either of the following ways:
- Increasing the amount of initiator relative to the monomer.
- Using a chain transfer agent like mercaptan or thiol.
Q. What does it signify if the PDI value is greater than 1?
Answer: PDI value greater than 1 signifies polymer has a wide range of chain length and weight. With the increase in the value of PDI, there will be increased variation in chain length, cross-linking, network formation, and branching.
Q. What is the most suitable method to determine the molecular mass of a polymer?
Answer: The most suitable method to determine the molecular mass of the polymer is the osmotic pressure method. There are some other techniques as well like gel permeation, NMR etc. but the best suited technique is the osmotic pressure method.


