-
Call Now
1800-102-2727
Law of Reciprocal Proportion – History, Definition and Examples of Law of Reciprocal Proportion, Practice Problems and FAQ
The cornerstones of our judicial system are lawyers. They are the representatives of all enacted laws that make up a healthy and lawful community. We need lawyers since they are fully knowledgeable about the national laws that govern a country. They are well-informed on India's constitution and judicial system. They can therefore provide people with substantial legal support thanks to their knowledge.
The four types of legal systems are mixed law, common law, customary law, and religious law. Using them, the judge makes the ultimate determination of what constitutes justice as the prosecutor oversees each case in the courtroom.

Similarly, chemistry also has some crucial rules known as the Laws of Chemical Combination that govern the formation of every chemical substance. One of these laws is the Law of Reciprocal Proportion.
In this article, we will understand the law of reciprocal proportions with some examples.
TABLE OF CONTENTS
- History of Law of Reciprocal Proportion
- Law of Reciprocal Proportion
- Practice Problems
- Frequently Asked Questions – FAQ
History of Law of Reciprocal Proportion
After calculating the neutralisation ratios of metals with acids, Richter essentially introduced the law of reciprocal proportions. Early in the 19th century, Berzelius looked into it and came up with the following formulation: The ratio of the quantities C and D that saturate the same amount of A is the same as the ratio between the quantities C and D that saturate the same amount of B, when two substances, A and B, have an affinity for two others, C and D. Later, Jean Stas proved that the stoichiometric rules were accurate within the bounds of experimental error.
Law of Reciprocal Proportion
The law of reciprocal proportions was given by Jeremias Ritcher in the year 1792.
According to this law, the ratio of the masses in which two different elements combine separately with a fixed mass of a third element, is either the same or a simple multiple of the ratio of the masses in which they combine with each other.
Example–1:
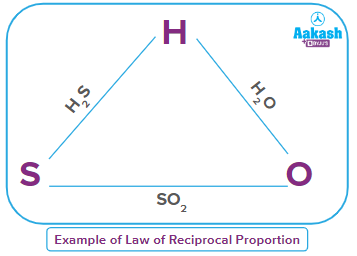
In H2O, the ratio of masses of hydrogen and oxygen is 2:16, i.e 1:8
In SO2, the ratio of masses of sulphur and oxygen is 32:32, i.e 1:1
Therefore, the ratio of masses of hydrogen and sulphur which combine with a fixed mass of oxygen (for example, 8 parts) will be 1:8. … … … (1)
When hydrogen and sulphur combine, they form H2S in the mass ratio of 2:32, i.e,. 1:16 … … … (2)
The ratio of (1) and (2) are related as 2:1, i.e., they are a whole number multiple of each other. Therefore, the ratio of masses of hydrogen and sulphur that combine with a fixed mass of oxygen is a whole number multiple of the ratio in which hydrogen and sulphur combine. Hence, it proves the law of reciprocal proportion.
Example–2:
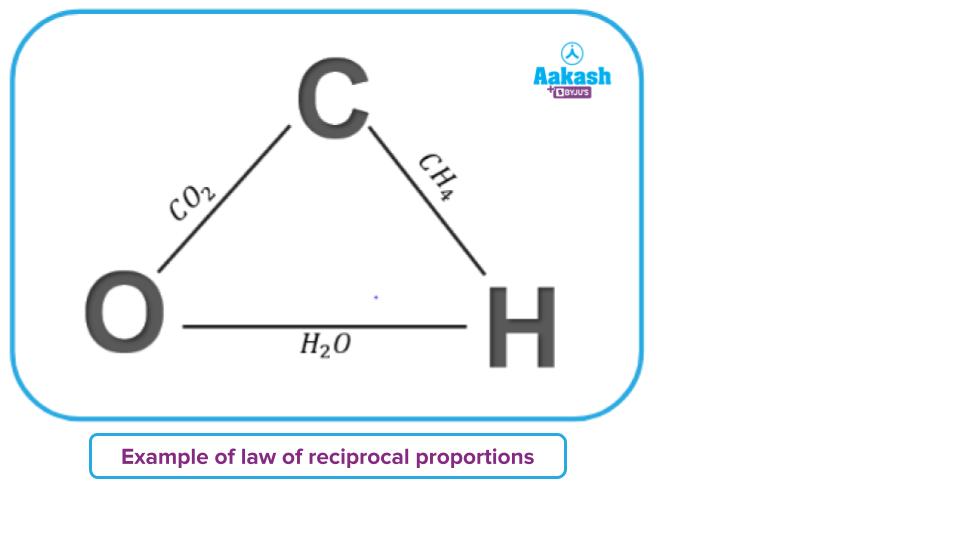
In CH4, the ratio of masses of carbon and hydrogen is 12:4, i.e 3:1
In H2O, the ratio of masses of oxygen and hydrogen is 16:2, i.e 8:1
Therefore, the ratio of masses of carbon and oxygen which combine with a fixed amount of hydrogen (for example, 1 part) will be 3:8. … … … (3)
When carbon and oxygen combine, they form CO2 in the mass ratio of 12:32, i.e,. 3:8 … … … (4)
The ratio of (3) and (4) are the same, hence proving the law of reciprocal proportion.
When carbon and oxygen combine, they form CO2 in the mass ratio of 12:16, i.e,. 3:4 … … … (5)
The ratio of (4) and (5) are related as 2:1, i.e., they are a whole number multiple of each other, thus proving the law of reciprocal proportion.
Practice Problems
1. Which of the following scientists introduced the law of reciprocal proportion?
a. Jeremias Ritcher
b. Neils Bohr
c. John Dalton
d. Avogadro
Answer: A
Solution: The law of reciprocal proportions was introduced by Jeremias Ritcher in the year 1792. After calculating the neutralisation ratios of metals with acids, Richter essentially introduced the law of reciprocal proportions.
So, option A is the correct answer.
2. What made the law of reciprocal proportions significant?
a. We now understand stoichiometry as a result of it.
b. It assisted researchers in finding novel compounds.
c. It assisted scientists in determining compound sizes.
d. The periodic table was made possible by it.
Answer: A
Solution: One of the fundamental principles of stoichiometry is the law of reciprocal proportions, often known as the law of equivalent proportions or the law of permanent ratios. It relates the ratios in which components combine across a variety of elements. Hence it is helpful in defining reaction stoichiometry.
So, option A is the correct answer.
3. Which of the following compounds describes the law of reciprocal proportion?
a. N2O4, N2O3, N2O5
b. NaBr, NaI, NaCl
c. P2O3, PH3, P2O5
d. CS2, CO2, SO2
Answer: D
Solution: The ratio of the masses in which two elements combine independently with a fixed mass of a third element, are either the same or a simple multiple of the ratio of the masses in which they combine with each other, according to the law of reciprocal proportion.
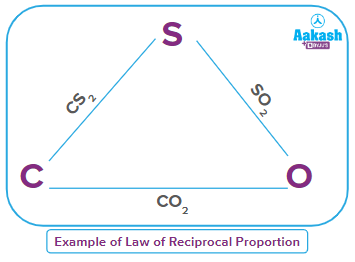
In CS2, the ratio of masses of carbon and sulphur is 12:64
In CO2, the ratio of masses of carbon and oxygen is 12:32
Therefore, the ratio of masses of sulphur and oxygen which combine with a fixed mass of oxygen (for example, 12 parts) will be 64:32 i.e., 2:1. … … … (6)
When sulphur and oxygen combine, they form SO2 in the mass ratio of 32:32, i.e,. 1:1 … … … (7)
The ratio of (6) and (7) are related as 2:1, i.e., they are a whole number multiple of each other, hence proving the law of reciprocal proportion.
So, option D is the correct answer.
4. Why is the law of reciprocal proportions also called law of equivalent proportions?
Answer: The ratio of the two components' masses when they combine independently with a third element of fixed mass is either the same or a whole number multiple of the ratio in which they mix. Only when the masses of the combining elements are combined in proportion to their equal masses, is the simple whole-number ratio established. The law of reciprocal proportion is therefore often referred to as the law of equal proportion.
Frequently Asked Questions – FAQ
1. How can the Law of reciprocal proportion be explained in terms of Dalton’s atomic theory?
Answer: According to Dalton's atomic theory, compounds are created by mixing atoms of various elements in a straightforward ratio of whole numbers. All atoms of the same element are said to be identical. As a result, when the component weights are combined with a fixed weight of another element, the component weights should have a straightforward ratio to the combined component weights. The reciprocal proportion rule is made clearer by this.
2. What are the limitations of this law of reciprocal proportion?
Answer: Differences similar to those seen in the law of constant proportions are produced by the element's isotopes. The synthesis of a number of chemicals should thus use the same isotope or a combination of isotopes. The law only applies to a small subset of products that exhibit the disputed feature because there are only a finite number of elements that will combine with the third element and also with one another.
3. Is the law of multiple proportions and the law of reciprocal proportions the same?
Answer: No, they are different. While the two distinct elements mix independently with the same weight of the third element in terms of the law of multiple proportions, the two different elements combine separately with the same weight of the third element, in the case of the law of reciprocal proportions.
4. Why is the name given to this law ‘reciprocal proportions’?
Answer: ‘Reciprocal’ means involving two or more parties who consent to lend a hand to one another or act in a similar manner toward one another. Let's say a third element has a fixed mass and two unique elements combine separately. The ratio of the masses they combine must be either the same or a simple multiple of the ratios of the masses they combine, according to the law. Hence this law is termed as ‘Law of reciprocal proportions’


