-
Call Now
1800-102-2727
KMnO4 Preparation - Discovery, Structure, Preparation, Uses, Practice Problems, FAQs
Researchers have long been interested in water filtration methods. In fact, it is one of the main forces behind scientific progress.
Therefore, it should not be surprising that civilization has developed a variety of strategies to deal with such challenges. Would you like to discover what the oldest techniques for purifying water were?
Permanganate of potassium is the answer (KMnO4).
The substance has other uses in addition to being used to clean water. This reagent is a major component in many organic syntheses. Used as a major oxidizing agent.

Your chemistry lab may have had beakers filled with a deep purple liquid solution when you first walked in. Teachers ask you to perform titrations using this purple solution because it is so attractive to look at. Researchers have long been enthralled by this substance because of its diversity and prospective uses.
We'll learn more about this coloured chemical, how this is prepared and examine its potential applications in chemistry.
TABLE OF CONTENT
- Discovery of KMnO4
- Structure of KMnO4
- Preparation of KMnO4
- Uses of KMnO4
- Practice Problems
- Frequently asked questions-FAQs
Discovery of KMnO4 :
The first signs of KMnO4 were found when a German-Dutch chemist named Johann Rudolf Glauber started his research by combining two compounds: the mineral pyrolusite (MnO2), which contains manganese dioxide, and potassium carbonate. He dissolved the mixture in water to create a green solution. This was manganese potassium. The solution's colour gradually transitioned from blue to violet to scarlet. The first time KMnO4 was made in this experiment.

Structure of KMnO4:
K+ and ions make up the inorganic ionic compound KMnO4. In the permanganate anion , the manganese atom is joined to four oxygen atoms by three double bonds and one single bond. The oxidation state of the manganese in this salt is +7. ion hybridization results in d3s (orbitals utilized are
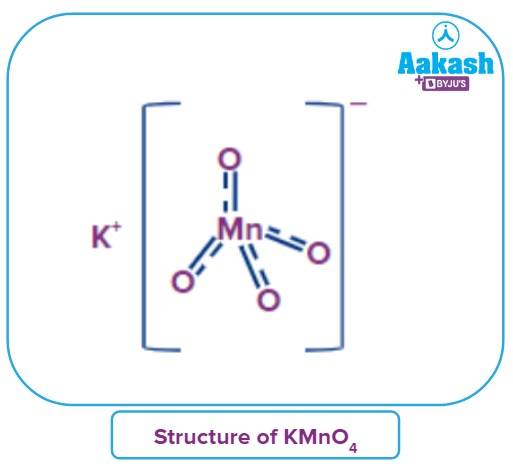
The partial double bond nature of the bonds between the oxygen atoms and the Mn atoms in the KMnO4 structure shown above is due to resonance.
Preparation of KMnO4:
- The dark green potassium manganate (K2MnO4) is produced commercially by oxidative fusion of MnO2 in an alkaline atmosphere with an oxidizing agent like O2 or KClO3. In a neutral or acidic media, this molecule disproportionates to produce the permanganate ion ().
- Peroxodisulphate oxidizes a manganese (II) ion salt to permanganate in the laboratory.
- Potassium manganate can be oxidized with chlorine or disproportionated in acidic environments. Chlorine oxidation is a chemical reaction that occurs in the human body.
Uses of KMnO4:
- Both analytical and preparative organic chemistry use potassium permanganate as a common oxidant.
- Its strong oxidizing ability is what allows it to bleach wool, cotton, silk, and other textile fibers, as well as lighten oils.
- Potassium permanganate is used to treat wounds, dermatitis, impetigo, pemphigus, and tropical ulcers.
- It is employed as a regeneration chemical to take iron and H2S out of well water. It was formerly used as a water sterilizer.
- It's employed to treat bacterial infections. But before applying potassium permanganate to your skin, you must dilute it with water. The majority of medical applications need for a dilution of 1 part to 10 when using a potassium permanganate solution that is 0.1%.
- Using potassium permanganate as a point-of-entry treatment, dissolved iron, manganese, and hydrogen sulphide are transformed into solid particles that may be removed from the water by filtration. It can also be used to stop iron bacteria from growing in wells.
Practice Problems:
Q1. What is the coefficient of water when sulphite reacts with permanganate ions?
A. 6
B. 5
C. 4
D. 3
Answer: D
Solution: Sulphurous acid or sulphite is converted to sulphate or sulphuric acid with a reaction with permanganate ion. Below is the required balanced reaction:
Q2. Potassium permanganate is used as a self-indicator because of __________.
A. It behaves as a strong oxidizing agent
B. It breaks down into ions easily
C. It gets disproportionated in acidic medium
D. It undergoes a chemical change resulting in colour difference.
Answer: D
Solution: Potassium permanganate has a rich purple colour that is extremely visible to the human eye, and when it is titrated, there is a noticeable loss of colour.
Q3. Pyrolusite on treated with KOH in presence of KNO3 give _________ color melt of manganate ions.
A. Black
B. green
C. yellow
D. pink
Answer: B
Solution: Pyrolusite on treated with KOH in presence of KNO3 give green colour melt of manganate ions.
Below is the flow of the above-mentioned statement.
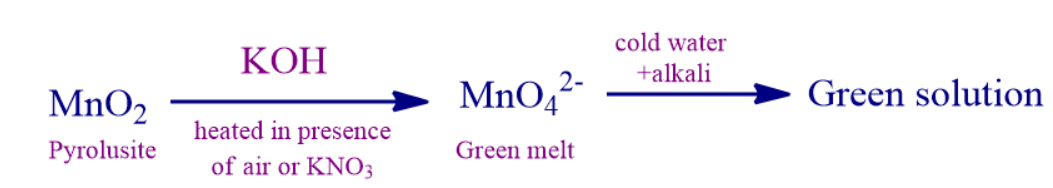
Q4. Predict the hydrogen ion coefficient in the reaction performed in the acidic medium in which KMnO4 acts as an oxidising agent.
A. 4
B. 16
C. 8
D. 7
Answer: C
Solution: The following equation can be used to represent the oxidising activity of KMnO4 in an acidic medium:
In the above reaction, coefficient of H+ is 8.
Frequently Asked Questions - FAQs:
Q1. What justifies standardizing KMnO4 on the same day?
Answer: We standardize potassium permanganate because using a standard solution of oxalic acid, potassium permanganate was standardized in order to be tested for strength. The oxidation and reduction theories can be studied using this reaction.
Q2. What occurs when water is added to potassium permanganate?
Answer: Even without stirring, the water in the entire beaker becomes purple when a potassium permanganate crystal is placed at the bottom of the liquid.
Q3. What does the term "standard solution" mean?
Answer: A standard solution is a liquid with a precisely known concentration that is made from a primary standard, which is a substance that is stable, highly pure, highly soluble in water, and has a high molar mass to enable precise weighing. The primary standard is weighed precisely and is made up to a specific volume.
Q4. Is potassium permanganate safe to eat?
Answer: No, it is not safe to consume potassium permanganate. The upper gastrointestinal system may be damaged if potassium permanganate is consumed. It can also produce systemic side effects including adult respiratory distress syndrome, coagulopathy, hepatic-renal failure, pancreatitis, and even death in extreme situations


