-
Call Now
1800-102-2727
Irreversible Processes - Definition, Difference Between Reversible and Irreversible Processes, Practice Problems and FAQs
Have you seen the movie “3 idiots”? I am sure most of you have seen it. This movie is one of the highest-grossing Bollywood movies made ever. Rajkumar Hirani directed this movie, starring Aamir khan (as Rancho), R.Madhavan (as Farhan Qureshi), Kareena Kapoor (as Pia) and Sharman Joshi (as Raju Rastogi) in lead roles.

Do you remember one of the scenes from this movie when Farhan tells Raju that nothing is impossible, Raju squeezes toothpaste into Farhan's hand and instructs him to put it back into the tube. One of the most hilarious scenes of this movie, right?
The point to highlight here is that it is impossible to put back the toothpaste into the tube. Now, my question is, can we say that this process is irreversible? The answer is Yes!!
Let’s understand in detail about the irreversible process and the thermodynamic aspect of it.
Table of Contents
- What is an Irreversible Process?
- Graphical Representation of an Irreversible Process
- Examples of Irreversible Processes
- Difference between Reversible and Irreversible Processes
- Practice Problems
- Frequently Asked Questions - FAQs
What is an Irreversible Process?
An irreversible process can be defined as a process in which the system and the surroundings do not return to their initial state once the process is started or the initial state is reached but not through the same path. In other words, in an irreversible process, if the system and surroundings return to their original state after taking certain paths, there will be some net change which occurs in the universe. Consider the case of a vehicle which has travelled some distance using gasoline. The fuel burns to generate energy for the engine, converting it into smoke and heat energy in the process. The energy which is lost will never be recovered making it an irreversible process.
Some features and reasons for the irreversibility of processes are:
- Friction is the process of converting the energy into heat energy and which is a loss of heat, hence friction process is always an irreversible process.
- The unregulated expansion of the fluid prevents the fuel from returning to its original form. The reversal of heat transfer through a finite temperature is not feasible because the forward process is spontaneous in this scenario.
- Intermixing two different substances is an irreversible process since the intermixing process is spontaneous in nature, and the opposite isn't possible.
- The fast processes are irreversible processes.
Illustration for an irreversible process:
Consider an ideal gas enclosed in a cylinder at 10 atm. At any point in time, there are two forces
acting on the piston.


Upward force: It acts due to the collisions of the gas particles with the piston i.e., the internal pressure of the gas. .
Downward force: It acts due to the atmospheric pressure i.e., the external pressure.
The piston is moved upward quickly to a new position such that the volume enclosed by the piston becomes double the initial volume. During this expansion process, a new zone is created near the piston where the gas particles are not uniformly distributed. The pressure at any two points in the new volume is different, or we can say that the pressure is not defined for the system in this intermediate stage because the system is not in thermodynamic equilibrium. After some time, let's assume when the gas particles are equally distributed in the cylinder, then, according to Boyle's law, the pressure of the final state is 5 atm. Thus, the pressure can now be defined for this final state, and now the system is in thermodynamic equilibrium. During this whole process of expansion, the pressure of the system is defined only when it is in thermodynamic equilibrium and not in an intermediate stage.
So, it can be considered an irreversible process that this process is a fast process and it can not be reversed without affecting the system or surroundings. Moreover, the final pressure of the gas is equal to the external pressure i.e., pfinal=pexternal.
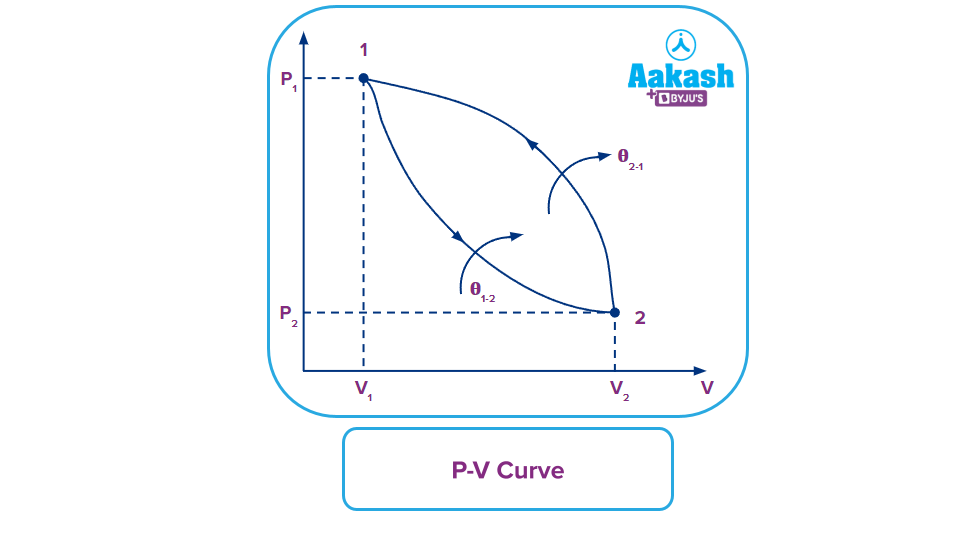
Graphical Representation of an Irreversible Process
Let’s consider two different thermodynamic states 1 and 2. During the thermodynamic process 1-2, some amount of heat is released by the system and this process is an irreversible process because it can not be reversed and even if it is reversed as shown in the figure below i.e., process 2-1, different amounts of heat is added to the system. So, for an irreversible process, the process cannot be reversed by not affecting the surrounding, there will always be a loss of energy during the process and back.
Examples of Irreversible Processes
- Movement of objects experiencing friction from the surface.
- Throttling of a gas from a small hole.
- Heat transfer from one object to another.
- Diffusion/effusion of gasses.
Difference Between Reversible and Irreversible Process
|
Reversible Process |
Irreversible Process |
|
Reversible processes are ideal processes. |
Irreversible processes are real processes. |
|
The reversible process is a slow process. |
An irreversible process is a fast process. |
|
A reversible process can be reversed without affecting the universe. |
An irreversible process cannot be reversed without affecting the universe. |
|
In a reversible process, changes that occur in the system are infinite. |
In an irreversible process, changes that occur in the system are finite. |
|
The reversible process takes infinite time for the completion. |
The irreversible process takes a finite time for the completion. |
|
In the case of a reversible process, there is always an equilibrium condition established between the initial state and the final state of a system. |
In the case of an irreversible process, the initial state and the final state of a system are never in equilibrium. The equilibrium is only achieved at the end of the process. |
|
In the case of a reversible process, Pint= Pext throughout the process. |
In the case of an irreversible process, Pint≠ Pext during the process. Internal pressure is only equal to external pressure when the equilibrium is achieved that is at the end of the process. |
|
Examples:
|
Examples:
|
Practice Problems
Q. Which of the following is true for a thermodynamically irreversible compression process?
().
- This process is a slow process.
- All the above options are incorrect.
Answer: (D)
Solution:
In the case of an irreversible process, during the process. Internal pressure is only equal to external pressure when the equilibrium is achieved that is at the end of the process. So, . Irreversible process is a fast process. All three options (A), (B) and (C) are incorrect. So, the correct answer is option (D).
Q. Select the correct statement from the following.
- Throttling of a gas from a small hole is an example of a reversible process.
- Heat transfer from one object to another is an example of an irreversible process.
- Effusion of gas is an example of a reversible process.
- Infinitesimally slow isothermal compression or expansion of gases are irreversible processes.
Answer: (C)
Solution:
The throttling of a gas from a small hole (like effusion) is an example of an irreversible process. Heat transfer from one object to another is an example of an irreversible process. Infinitesimally slow isothermal compression or expansion of gases are reversible processes. So, option (B) is the correct answer.
Q. Consider a thermodynamically irreversible expansion process. The initial internal pressure of the gas inside the cylinder is 3 atm. The external pressure outside the cyllinder 1.5 atm. The final pressure (Pfinal) of the gas would be:
- Insufficient data to find out the final pressure of the gas
Answer: (C)
Solution:
An irreversible process is a fast process and the internal gas pressure is never in equilibrium with the external pressure during the process until it reaches the final equilibrium stage. So, finally, once the system expanded completely and reaches the equilibrium state, the final pressure of the gas will be equal to the external pressure.
So,
Q. Consider a thermodynamically irreversible expansion process. During the process (from state 1 to state 2), Q1-2=20 J energy is produced by the system. Which of the following is true for the compression process from state 2 to state 1?
- Q2-1=20 J
- Q2-1 ≠ 20 J
- Throughout the process, Pinternal= Pexternal
Answer: (B)
Solution:
Irreversible process cannot be reversed without affecting the universe. So,
In the case of an irreversible process, Pint≠ Pext throughout the process. Internal pressure is only equal to external pressure when the equilibrium is achieved that is at the end of the process. So, the correct answer is option (B).
Frequently Asked Questions – FAQs
Q. What is a quasi-static process?
Answer: A quasi-static process is a process which occurs infinitesimally slow such that at every stage of the process thermodynamic equilibrium is maintained. A reversible process is said to be a quasi-static process, where the change is likely to be occurring at a highly slow rate.
Q. Why friction is known as an irreversible process?
Answer: During friction between surfaces, the heat energy is lost or dissipated which can never be recovered by reversing the process. Due to this loss of heat energy, the friction process is an irreversible process.
Q. What is the reason for heat transfer being an irreversible process?
Answer: There is a finite temperature difference between the two bodies which are interacting. The energy transfer is an irreversible process because of the loss of energy as the interaction is not completely true.
Q. Is speaking a reversible or irreversible process?
Answer: While speaking, air gushes out through the larynx and throttling of air occurs. This throttling process is a fast process and there is friction involved between air and vocal chord inside the larynx. So, speaking is an irreversible process.


