-
Call Now
1800-102-2727
Gas Electrode: Electrodes, Types of Electrodes, Gas Electrodes, Standard Hydrogen Electrode, Chlorine Gas Electrode, Oxygen Gas Electrode, Practice Problems, FAQs
You must have been to a bank. Even if you haven’t, I believe you must be aware of the basic operations of a bank.
In simple terms, we can say that bank is a an institution which collects money from the public and gives some rate of interest for the time they keep their money with them.
But apart from just keeping money in our account, it also gives money to people and organisations in terms of loans. Nowadays they do multiple other activities like selling insurance policies etc.

We can conclude our definition as the organisation which allows people or other organisations to save, borrow or exchange money or assets is called the bank.
Similarly in electrochemistry electrodes are referred to as a point where either electron enters or leaves the electrolyte.
Sometimes, electrodes don’t even participate in this process yet they provide the required surface for the oxidation and reduction process to take place. These electrodes are called inert electrodes.
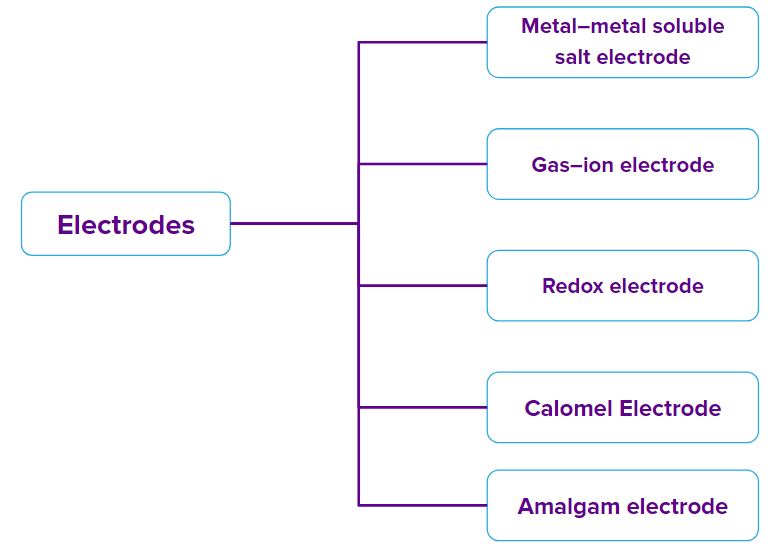
There are different types of electrodes which are generally used in electrochemical processes. Such as Metal–metal soluble salt electrode, Gas–ion electrode, Redox electrode, Metal–metal insoluble salt electrode, Calomel Electrode, and Amalgam electrode.
Let’s understand gas electrode, their type and working principle.
Table of content
- Electrodes
- Types of Electrodes
- Gas Electrode
- Standard Hydrogen Electrode (SHE)
- Chlorine Gas Electrode
- Oxygen Gas Electrode
- Practice Problem
- Frequently Asked Questions (FAQs)
Electrodes
An electrode is a type of electrical conductor that makes contact with nonmetallic circuit components such as an electrolyte and, gas. Electrodes can be referred to as either an anode or a cathode depending on their ability to pass on or receive electrons in an electrochemical cell. The anode is an electrode where electrons leave the cell into the circuit and oxidation occurs. Electrons enter the cell through the cathode and undergo a reduction process.
An electrode of metal is not permanently fixed as an anode or cathode because it can act as an anode or a cathode depending on its electrode potential in relation to the other electrode.
Types of Electrodes
On the basis of the constitution of an electrode, electrodes are classified into the following types:
1. Metal–metal soluble salt electrode
2. Gas–ion electrode
3. Redox electrode
4. Metal–metal insoluble salt electrode
5. Calomel Electrode
6. Amalgam electrode
Gas Electrode
A gas electrode is made up of a gas such as H which is in contact with a solution that contains the gas's ions, such as . The concentration of its ions in the solution and the pressure of a gas affect the gas electrode's potential.
A gas electrode is made out of gas that is bubbled around an inert metal wire (a platinum electrode) and submerged in a solution that contains ions, with which gas is irreversibly reacted. Platinum is utilised to absorb the gas and as a conductor. for example, Standard hydrogen electrode.
Standard Hydrogen Electrode (SHE)
Let us consider a hydrogen gas electrode, operating as an anode. It can be represented as
and the anodic half-cell reaction is
Application of Platinum (Pt) in Standard Hydrogen Electrode (SHE)
The Standard Hydrogen Electrode uses platinum for the following reasons:
- Platinum is a somewhat inert metal that resists corrosion well.
- The proton reduction reaction is promoted by the catalytic properties of platinum.
- Platinum black, a fine powder made of platinum, can be used to cover the surface of platinum. A platinized platinum electrode is the name given to this kind of platinum electrode.
- Platinum enhances the kinetics of the reaction by adhering hydrogen at the interface.
Construction of SHE
Below is a list of the components of a standard hydrogen electrode.
- An electrode made of platinum that is covered in finely ground platinum black (platinized platinum electrode).
- a flow of hydrogen.
- a solution of acid with a molar concentration of H+ of 1 mole per cubic decimeter.
- A hydroseal, intended to stop oxygen interference, is also a component of SHE.

The hydrogen electrode is called a standard hydrogen electrode when the concentration of H +ion
is 1 M and the pressure of H2 a gas is 1 atm.
The standard hydrogen electrode (SHE) functions as a reference electrode and is used for the measurement of standard reduction potentials of other half-cells or couples. The standard reduction potential values obtained in this manner are arranged in decreasing order to give electrochemical series. The reduction potential of the hydrogen electrode is zero.
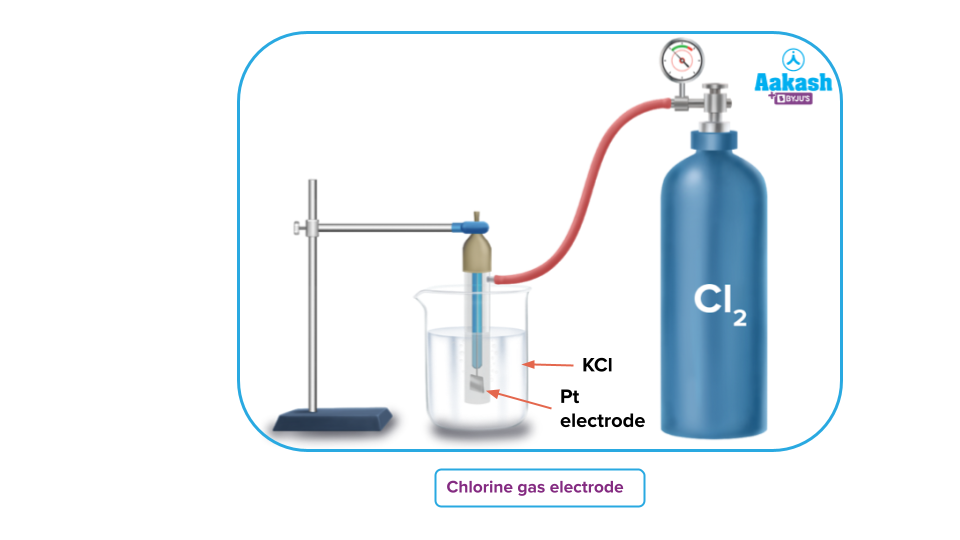
Chlorine Gas Electrode
A chlorine gas electrode is made up of Cl2 gas which is in contact with a solution that contains the Cl- ions. The concentration of its ions in the solution and the pressure of a gas affect the gas electrode's potential.
The chlorine gas electrode when functions as an anode can be represented as
and the anodic half-cell reaction is
The reduction potential of oxygen is 1.23 V.
Oxygen Gas Electrode
An oxygen gas electrode is made up of O2 gas which is in contact with a solution that contains the OH- ions. The concentration of its ions in the solution and the pressure of a gas affect the gas electrode's potential.
When acting as an anode, the oxygen gas electrode is represented by
and the anodic half-cell reaction is
When acting as the cathode, the gas-gas ion half-cell can be represented as
Gas ion (c M) | Gas (P atm) | Pt.
Consider a hydrogen half-cell that serves as the cathode. It is represented as
and the cathodic half-cell reaction is
The reduction potential of chlorine is 1.396 V .
Practice Problems
Q1. The hydrogen electrode is called the standard hydrogen electrode when
A. H +ion is 1 M and the pressure of H2 gas is 1 atm
B. H +ion is 10 M and the pressure of H2 gas is 1 atm.
C. H +ion is 1 M and the pressure of H2 gas is 10 atm.
D. H +ion is 10 M and the pressure of H2 gas is 10 atm.
Answer: (A)
Solution: The hydrogen electrode is called standard hydrogen electrode when the concentration of H +ion is 1 M and the pressure of H2 gas is 1 atm.
Q2. SHE (standard hydrogen electrode) is represented as
A. H2 (P atm)|Pt | H+ (c M)
B. Pt|H2 (P atm) | H+ (c M)
C. Pt|H+ (c M)| H2 (P atm)
D. Pt|H2 (P atm) | H2 (c M)
Answer: (B)
Solution: SHE has represented as
The half cell reactions are as follows
Anode (oxidation):
Cathode (reduction):
Q3. Chlorine gas electrode can be represented as
A. Pt|Cl2(P atm) | Cl2 (c M)
B. Cl2(P atm)| Pt | Cl- (c M)
C. Pt|Cl2(P atm) | Cl- (c M)
D. Pt| Cl- (c M)| Cl2(P atm)
Answer: (C)
Solution:
Chlorine gas electrode can be represented as
The half-reactions are as follows
Anode (oxidation):
Cathode (reduction):
Q4. The pressure of hydrogen gas needed to reduce the potential of the H2-electrode in pure water to zero at 298 K is
A. PH2=10-14 atm
B. PH2=1014 atm
C. PH2=10-10 atm
D. PH2=1010 atm
Answer: (A)
Solution:
The concentration of H+ ion in H2O is
According to Nernst equation
Putting the values of R, T , F and n
We get
As we know log 0=1
Hence, The pressure of hydrogen gas needed to reduce the potential of the H2 electrode in pure water to zero at 298 K is 10-7 atm
Frequently Asked Questions (FAQs)
Q1. Which is the most widely used reference electrode?
Answer: The most common type of reference electrode is a silver/silver chloride reference electrode. Because of its low half-cell potential of about +222 mV (SHE), low impedance, and lower toxicity than the calomel electrode containing mercury, silver/silver chloride is a popular choice of biological electrodes.
Q2. Can we consider a platinum electrode as a reference electrode?
Answer: No, Platinum electrodes are used as inert electrodes. Another example of an inert electrode is a graphite electrode. But Pt can be used as a reference electrode in situations where traditional reference electrodes are ineffective.
Q3 What do you understand by pH electrode?
Answer: pH electrodes are analytical sensors used to measure hydrogen potential (pH), which is the negative logarithm of hydrogen ion activity in solution. The pH of a substance is proportional to the concentrations of hydrogen ion [H+] and hydroxyl ion [OH-].
Q4. Why is platinum used as an electrode?
Answer: Platinum serves as an oxidation and reduction reaction surface or base rather than participating in the cells' process, making it an inert catalyst. Platinum is utilised in the operation of cells because hydrogen may be easily adsorbed and because it is an inert metal that does not take part in redox reactions.


