-
Call Now
1800-102-2727
Anomalous Properties of Fluorine: Introduction, Physical Properties, Anomalous Properties, Anomalous Chemical Properties, Applications, Practice Problems & FAQs
Do you know that an adult needs around 4mg of fluorine as fluoride daily. If you are a regular eater of sweet dishes and chocolates, definitely you could have ended with a dentist, who would have given a fluoride treatment or at least suggested a fluoride toothpaste.
It has been demonstrated that fluoride guards against tooth decay.
When a person consumes sweet foods, oral bacteria grows in the mouth and release acid. By removing minerals from the tooth's surface, this acid weakens the tooth and raises the risk of cavities. The tooth's enamel surface is strengthened and rebuilt with the aid of fluoride. Fluoridation of water prevents tooth decay by exposing people to little amounts of fluoride on a regular basis. Fluoride can even rebuild the tooth's surface by keeping the tooth robust and solid and preventing the development of cavities.

In contrast to utilising them infrequently at large concentrations, it is typically advised to utilise these fluorides more regularly at small concentrations. When apatite crystals are formed, fluoride ions (F-) take the place of hydroxyl groups (OH-) to produce a stronger, fluoridated tooth mineral (fluorapatite).
Fluorapatite is more resistant to subsequent demineralization when acid is created by the bacteria because it is less soluble than hydroxyapatite, even in acidic conditions.
Table of Content:
- Introduction of Fluorine
- Physical Properties of Fluorine
- Anomalous Properties of Fluorine
- Anomalous Chemical Properties of Fluorine
- Applications of Fluorine
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Fluorine:
Halogen gas, fluorine, is a member of the periodic table's 17th group. The element atomic number is 9. Being the most electronegative element in the periodic table fluorine is most distinguishing quality. Under normal circumstances, it appears as an extremely poisonous pale yellow diatomic gas. Fluorine has the highest electronegativity of any element, measuring 3.98 on the Pauling scale.
Fluorine has a valency of one as it is a halogen. It lacks one electron and is an electron receiver. These substances are oxidising agents because they accept electrons.
Fluorine is the greatest oxidising agent among the halogens due to its high electronegativity. Fluorine has the electronic structure 1s2 2s2 2p5. We can plainly see from the element's electrical structure that it is missing one electron. It receives an electron gladly since it is an electrophile.
Physical Properties of Fluorine:
- Fluorine is the lightest of all the halogens, with an estimated atomic mass of 19..
- In its natural state, fluorine is found as a gas. It is a lightweight gas with a pale yellow colour.
- Fluorine has a melting point of 53.4 K and a boiling point of 85 K.
- The atomic radius of fluorine, a very light element, is extremely small. A fluorine atom has a very strong nuclear force as a result of this. The smallest halogen atom is fluorine, which has an atomic radius of only 147 pm.
- The powerful attraction force between the nucleus and the electrons caused by their small size results in a very high energy requirement to extract an electron. Fluorine has a first ionisation energy of 1680.6 KJ mol-1. Thus, one mole of fluorine requires 1680.6 KJ of energy to remove one electron.
- Fluorine has the highest standard electrode reduction potential of 2.87 V. Compared to all other elements this is the highest.
- Fluorine has the highest electronegativity of any element, measuring 3.98
- There is just one stable isotope of fluorine, which is fluorine 19.
Anomalous Properties of Fluorine:
Compared to other family members, fluorine exhibits abnormal behaviour due of its-
- Incredibly tiny atomic radius.
- High ionisation energy and electronegativity values.
- Due to the lack of vacant d- orbitals.
- Low dissociation energy of the F-F bond.
- Strong ability for hydrogen bonding to form.
Abnormal Chemical Properties of Fluorine:
1. Polyhalide ions: Fluorine is not prone to generate polyhalide ions, but other halogens do, because it has the lowest size and no d-orbitals in its valence shell.
2. Electronegativity: Since fluorine is the strongest electronegative element in the periodic table, it is the most powerful oxidizer and accepts electrons from other elements. Fluorine possesses five electrons in its 2P shell, making it the most electronegative element. Since Fluorine is so close to the ideal electron configuration for the 2P orbital, which has six electrons, the electrons are bound very securely to the nucleus. Because of its high electronegativity, fluorine has a narrow radius, which is explained by the strong attraction of the positive protons to the negative electrons, which holds them closer to the nucleus than the more massive and less electronegative elements.
3. Hydrogen bonding: Of all the halogens, fluorine has the highest electronegativity. Because the H-F bond is quite polar in nature, fluorine makes strong hydrogen bonds in its hydride, in contrast to other halogens. As a result, unlike other hydrogen halides which are gases, HF is a liquid at ambient temperature.
4. Oxidation states: Fluorine has a single oxidation state of -1 since it is the smallest and most electronegative element, but other halogens have multiple oxidation states, including -1, +1, +3, +5, and +7.
5. Oxidising nature: Due to the strong electronegativity , fluorine has a higher probability to receive an electron. It is therefore a stronger oxidising agent than other halogen family members. Fluorine is a more potent oxidizer even though its electron gain enthalpy is less negative than that of chlorine. This is due to chlorine's smaller standard reduction potential
(1.36 V) and fluorine's greater standard reduction potential (+ 2.87 V).
6. Reactivity: Out of all the halogens, fluorine has the highest reactivity. This is brought on by the low F-F dissociation enthalpy of the F2 molecule. Other halogens can be replaced by it in salt solutions. In contrast to the other halogens, fluorine combines with hydrogen in the dark and at low temperatures and with gold and platinum.
When exposed to moisture, fluorine reacts violently and turns into hydrofluoric acid. If hydrogen fluoride (HF) is not neutralised, it can severely infiltrate biological tissues and cause further damage.
Fluorides are created when fluorine reacts directly with non-metals such as carbon, silicon, nitrogen, and others. On the other hand, the other halogens do not interact with these substances directly, which is the cause of fluorine's peculiar behaviour.
7. Low electron gain enthalpy than chlorine: Comparing chlorine to fluorine, the chlorine has a larger gain in enthalpy. This can be related to fluorine's small size, which causes its small 2p orbitals to have stronger inter-electronic repulsion and much less affinity to incoming electrons. Compared to fluorine, chlorine has a higher negative electron gain enthalpy. Chlorine, thus, has the most intense negative electron gain enthalpy of all the elements. It is a result of the fluorine atom's short size and diminished 2p subshell. The incoming electron experiences a greater degree of repulsion from the current electrons because of the fluorine particle's small size.
8. Formation of Hexafluoride: Fluorine rapidly converts sulphur into the a +6 oxidation state, generating the stable hexafluoride SF6, owing to its high electronegativity.
9. Covalency: Fluorine only exhibits a covalency of +1 (in the F-F molecule), but other halogens can display covalencies of up to +7 since they have unoccupied d-orbitals in their valence shells.
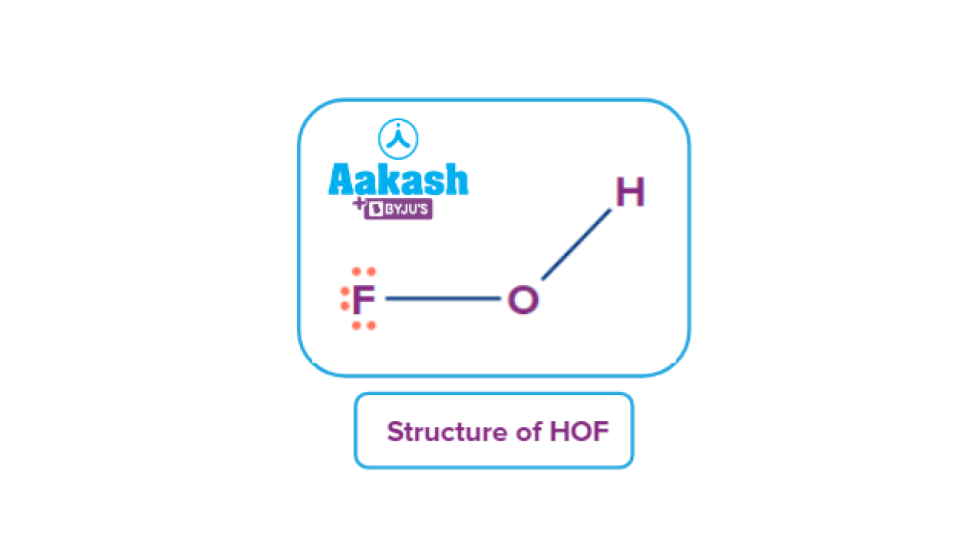
10. Formation of oxoacid: Because of its powerful electronegativity and compact size, fluorine only produces one oxoacid, known as HOF, also known as fluoric (I) acid or hypofluorous acid, unlike chlorine, bromine, and iodine, which all produce several oxoacids.
HOF is a colourless, flammable gas. In 1968, it was initially produced using the matrix separation technique. A crystalline nitrogen network that was non-reactive included F2 and H2O.
The production of the HOF was caused by the photolysis of the gases. Additionally trapped in solid nitrogen, it was unable to interact or collide with molecules such as water or oxygen. A product was consequently produced. Recently, F2 was converted into HOF by placing it in an ice bath at 273 K and extracting the finished product into a cold trap.
Structure of HOF:
Applications of Fluorine:
- Fluorine is used in toothpaste and potable water to prevent tooth decay.
- It can be found in ceramic clay.
- They can be found in the refrigerants known as chlorofluorocarbons.
- They serve as a source of nuclear power.
Practice Problems:
Q1. The name of the compound Na3AlF6 is
(A) Cryolite
(B) Feldspar
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: Fluorine can be obtained from nature or created in a laboratory. To synthesise pure Fluorine and other chemicals in a laboratory, substances like potassium fluoride are electrolyzed with hydrofluoric acid. It can be done using a variety of substances, most frequently ionic compounds containing fluorine and a metal. Various minerals and compounds that include fluorine can be found in nature. It can be found in Fluorspar (CaF2) and Cryolite ( Na3AlF6 ), which are the two major compounds.
Q2. Fluorine’s best oxidising agent is due to
(A) Highest electron affinity
(B) Largest reduction potential
(C) Largest oxidation potential
(D) None of the above
Answer: (B)
Solution: An element with such a higher electronegativity is fluorine. Fluorine has the largest reduction potential and the lowest bond enthalpy, giving it more potential to lose electrons. Additionally, fluorine is easily reduced. Thus, one of the best oxidising agents is fluorine.
Q3. Which of the following lists represents the bond dissociation enthalpy of halogen compounds correctly?
(A) F2 >Cl2>Br2 >I2
(B) Cl2 >Br2>F2 >I2
(C) I2 >Cl2>Br2 >I2
(D) I2 >Br2>Cl2 >F2
Answer: (B)
Solution: As atom size grows, the bond dissociation energy of the halogen family decreases down the group with fluorine as an exception.
Being the smallest molecule, fluorine atoms have the shortest radii and theoretically should have the strongest attraction between their nuclei and the shared pair of electrons, forming a covalent bond that is exceedingly difficult to break. Because of the strong effective nuclear charge and the strong nuclear attraction to the valence electrons, there is less shielding of the valence electrons, which results in interelectronic repulsions between the tiny F-F molecules. Because their valence electrons are closer to one another, the F-F bond is weak and requires very little dissociation energy, hence these molecules repel one another. Bond dissociation enthalpy should be in the following order: Cl2 >Br2 >F2 >I2 .
Q4. Hydrofluoric acid is
(A) Polar
(B) Non polar
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: Hydrofluoric acid (HF) is an extremely polar molecule. Electronegativity, a characteristic of all atoms, is the cause of the polarisation of the hydrofluoric acid (HF) covalent bond. When an atom is connected to another atom, there is a tendency for them to draw electrons to themselves in the periodic table (as in HF). Atoms with a tendency to aggressively draw electrons are said to have a high electronegativity in comparison to atoms with a tendency to attract electrons relatively low.
Fluorine has an electronegativity of 4.0 while hydrogen has a value of 2.2 in the molecule HF. This variation causes the hydrofluoric acid (HF) covalent bond to become significantly polarised.
Frequently Asked Questions(FAQs):
Q1. When proceeding from fluorine to iodine in the group of halogens, the colour of the halogens deepens. Explain why?
Answer: Because of their lower ionisation energy, halogens are coloured, which causes the electron to become excited by absorbing visible light. Thus, the molecule receives the complementary colour from the reflected light.
Proceeding down the group, the colour deepens as the atomic radii of the elements grow from fluorine to iodine. As a result, low energy light (higher wavelength i.e. toward red) is absorbed.
Q2. What exactly is abnormal behaviour?
Answer: As a result of their compact size, electronegativity, and absence of d-orbitals, the elements in a group might behave differently from other members of the family, which is known as abnormal behaviour. An individual, who exhibits abnormal behaviour is unique from the rest of the group. Anomalous properties are those that differ from what should exist for all members of a group of elements. Fluorine differs in some ways from other halogens. As a result, fluorine is said to behave abnormally.
Q3. Why is hydrofluoric acid the weakest of the halo acids?
Answer: Hydrofluoric acid is a weak acid rather than a strong one. Because it doesn't entirely dissociate in water, it is categorised as a weak acid. Essentially, it contradicts the concept of a strong acid. However, following dissociation, a powerful force binds the ions it forms to one another. When we look at fluorine, we typically see that it has a good attraction for hydrogen. Breaking the strong H-F bond in water requires a significant amount of energy. The substantial hydrogen bonding in HF prevents the release of hydrogen as H+ ions in aqueous solution because fluorine has a higher electronegativity.
Q4. Compared to chlorine, fluorine is more intensely reactive, why?
Answer: Because fluorine molecules have a low dissociation energy and fluorine ions have a high hydration energy, fluorine is more reactive than chlorine. As an element moves lower in the group, its electronegative property, which measures its capacity to draw a pair of bonding electrons, drops. When compared to chlorine, fluorine is more electronegative.


