-
Call Now
1800-102-2727
Ferrous Oxide - Introduction, Preparation, Properties, Structure, Uses, Practice Problems & FAQs
In the modern world, we are modernizing ourselves. Isn’t it? We are adapting the western culture, which has been observed around your surroundings. Do you know why I am telling you such things?
Today, we will study one of the chemical compounds available in chemistry which is now giving a fascinating modern culture of inking tattoos. Many youngsters are now passionate about tattoos. Many celebrities you found have ink tattoos on their hands or other body parts. The media and famous people greatly influence young people and their lifestyles. Many youngsters today are curious to know what renowned people get up to.

Do you know what chemical is present in the tattoo that Indian cricketer Virat Kohli has on his hand? Even not only Virat but every tattoo which is inked has a chemical compound known as ferrous oxide which is mixed with other chemicals.
Iron is a very versatile compound in chemistry and ferrous oxide is one of the famous compounds of iron.
Let’s learn more about its preparation, properties and uses in way more detail!
TABLE OF CONTENT
- What is ferrous oxide?
- Preparation of ferrous oxide
- Properties of ferrous oxide
- Structure of ferrous oxide
- Uses of ferrous oxide
- Occurence of ferrous oxide
- Practice Problems
- Frequently asked questions
What is ferrous oxide?
Iron (II) Oxide Formula, commonly known as Ferrous Oxide or Iron Monoxide. One oxygen atom and one iron atom make up this inorganic molecule. FeO is the chemical or molecular formula for iron oxide. Wustite is the mineral form of it. It is a black-coloured powder that is one of the various iron oxides.

Preparation of ferrous oxide:
The thermal breakdown of iron(II) oxalate produces FeO.

To avoid the development of iron (III) oxide, this technique is carried out in an inert environment of Fe2O3.
Properties of ferrous oxide:
- Below 575 °C, FeO is thermodynamically unstable and tends to decompose to metal and Fe3O4.

- FeO is totally basic and dissolves in acids.
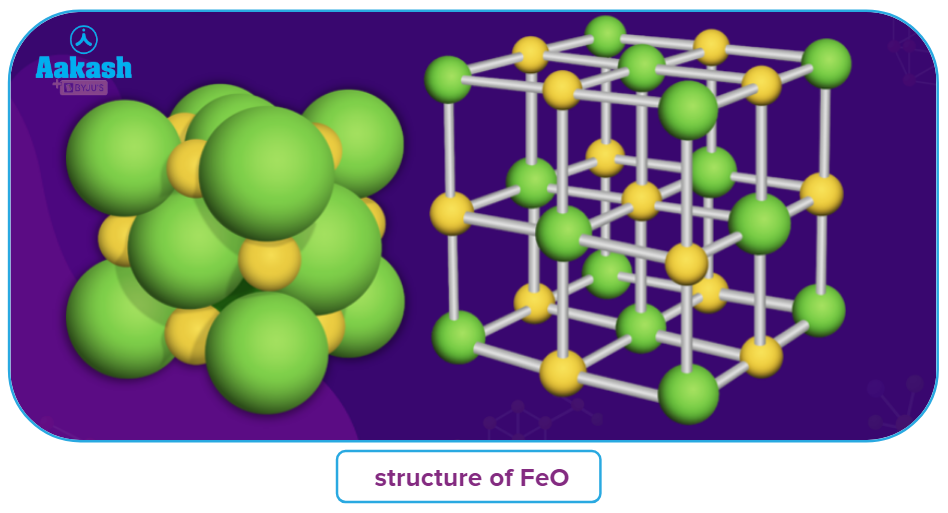
- It has a structure similar to sodium chloride lattice, with Fe2+ ions occupying all of the octahedral holes in a cubic close-packed arrangement of O2- ions.
- Fe(OH)2 is a white solid that forms from Fe (+II) solutions, but it quickly absorbs O2 from the air and turns dark green, then brown. This is due to the fact that it first oxidizes to a mixture of Fe(OH)2 and Fe(OH)3.
Structure of ferrous oxide:
The cubic, rock salt structure of iron(II) oxide is made up of iron atoms that are octahedrally coordinated by oxygen atoms and oxygen atoms that are octahedrally coordinated by iron atoms. Due to the simplicity of oxidizing Fe(II) to Fe(III), a little part of Fe(II) is effectively replaced by two-thirds as many Fe(III), which occupy tetrahedral places in the densely packed oxide lattice.
Uses of Ferrous oxide:
- It's used in cosmetics, and certain tattoos inks contain it.
- It can also be used to eliminate phosphate from home aquariums.
- Synthetic iron oxide has been permitted for use as a pigment in food, cosmetics, contact lenses, and pharmaceuticals.
Occurrence of ferrous oxide:
About 9% of the Earth's mantle is made up of iron(II) oxide. It's feasible that the mantle is electrically conductive inside, which would account for rotational disturbances that aren't explained by current theories of the mantle's characteristics. It has been reported that in India, Wustite(FeO) is found near Jharkhand.
Practice Problems:
Q1. What is the oxidation state of iron in FeO?
A. +2
B. +3
C. +1
D. 0
Answer: A
Solution: Let us assume the oxidation state of Fe in FeO be x. The oxidation state of oxygen is -2 because it is in oxide form. Solving this,
Hence, +2 is the oxidation state of Fe in FeO
Q2. Slag formation takes place when FeO reacts with ___________.
A. Cu2O
B. SiO2
C. O2
D. O3
Answer: B
Solution: The slag is a complex mixture of silicates and oxides that forms as a molten liquid melt and solidifies after cooling. Waste material removed from metal during smelting is known as slag.
Q3. When FeO reacts with Carbon _______ gas is released.
A. CO2
B. CO
C. CO2 and CO
D. CH4
Answer: B
Solution:In order to comprehend the fundamental processes occurring during the reduction process of smelting, it has been researched how FeO reacts with carbon in reduction reactions. Carbon remove any dissolved O2 and reduce FeO while acting as deoxidizers. Carbon monoxide is released in the process. Below is the mentioned reaction;
Q4. FeO has _________ kind of structure.
A. Fluorite
B. Wurtzite
C. Antifluorite
D. Rock salt
Answer: D
Solution: The cubic, rock salt structure of iron(II) oxide is made up of iron atoms that are octahedrally coordinated by oxygen atoms and oxygen atoms that are octahedrally coordinated by iron atoms.
Frequently Asked Questions - FAQs:
Q1. What kind of a bond does FeO has?
Answer: Iron oxide has an oxidation state of +3 and +2. The difference in electronegativity between the two atoms is what causes the bond to form between iron and oxygen. The bond between oxygen and iron is ionic because iron is metal and oxygen is non-metal.
Q2. Does FeO can be called rust?
Answer: Rust is an iron oxide, a typically reddish-brown oxide created when iron and oxygen react catalytically with moisture from the air or water. Rust is often linked to the corrosion of refined iron and is composed of hydrous iron(III) oxides.
Q3. Is FeO acts as an electrolyte or not?
Answer: In contrast to the majority of other metal electrowinning (recovering metals from its electrolytes) electrolytes, the dissociated ferrous oxide FeO becomes the main contributor to the electrical conductivity as its concentration increases.
Q4. What is the molar mass of FeO?
Answer: The molar mass of FeO can be calculated with the help of the below-mentioned information:
Molar mass of
Molar mass of
Molar mass of FeO comes out to be


