-
Call Now
1800-102-2727
Factors Affecting Vapour Pressure: Introduction, Factors Affecting, Applications of Vapour Pressure, Practice Problems & Frequently Asked Questions(FAQs)
Everyone is familiar with the LPG cylinder, which has become essential for our cooking.
Do you know which technique will use an LPG cylinder to supply gas to the burner?
The answer is its vapour pressure.

LPG stands for liquified petroleum gas. An LPG cylinder cannot provide fuel to the burner if there is insufficient vapour pressure. The valve opens when the burner knob is turned, increasing the vapour pressure. This enables the fuel vapours to move in the burner's path. The valve also closes when the burner is switched in the reverse direction, lowering the vapour pressure. Fuel vapour flow is interrupted as a result of this. In this article we will discuss the vapour pressure, factors affecting the vapour pressure and applications of vapour pressure in detail
Table of Contents:
- Introduction of Vapour Pressure
- Factors affecting Vapour Pressure
- Applications of Vapour Pressure
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Vapour Pressure:
In a sealed chamber, when a liquid evaporates out, some of it turns gaseous and releases vapours to fill the empty space. More liquid molecules are transformed into the vapour phase as the evaporation process advances. These gaseous molecules are in relative motion with one another, and some of them collide with the liquid's surface. The vapours condense as they contact the liquid's surface and the process of condensation proceeds in the opposite direction to that of evaporation. The processes of evaporation and condensation proceed simultaneously as a result, and a point exists when the rates of the two processes are equal. It becomes possible for the liquid and vapour phases to be in equilibrium.
The vapour pressure is the force that the liquid's vapours at equilibrium exert on the surface at a specific temperature.
In comparison to the vapour pressure of the same liquid after the addition of a solute, the vapour pressure of a pure liquid is higher. As an illustration, water will have higher vapour pressure than lemon juice.
Factors Affecting Vapour Pressure:
Vapour pressure depends on the following factors:
- Temperature
- Surface Tension
- Mole fraction
- Nature of the liquid
- Boiling point
- Molecular mass
1. Temperature:
The influence of temperature can also be explained using thermodynamics, as seen below:
Consider liquid evaporation.
Liquid ⇌ Vapour
Heat is needed for this evaporation or conversion. Therefore, evaporation is always endothermic(heat is absorbed) and H (or Hvap) is positive.
Le Chatelier's principle states that a rise in a system's temperature during a state of dynamic equilibrium encourages endothermic processes. As a result, there is more vapour generated as the temperature goes up. The saturated vapour pressure increases as a result. The following chart shows how temperature affects a liquid's vapour pressure.
The following is a quantitative measurement using the Clausius-Clapeyron equation:
Where,
P1= Liquid vapour pressure at temperature T1
P2= Liquid vapour pressure at temperature T2
H= Enthalpy of Vaporisation.
2. Surface Tension:
Most liquid molecules experience forces from all angles, resulting in an overall vector sum of forces acting on the molecules. The total sum of forces for the particles on the majority of the solution (A) will be zero.
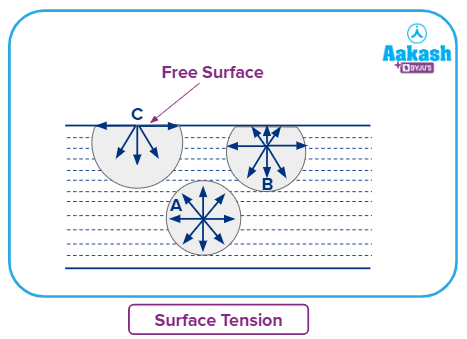
But, there is just one force acting on the particles on the surface (C). Surface tension is the resultant force a particle experiences on a surface per unit length of the surface. The surface tension rises and the vapour pressure decreases as the intramolecular force rises.
The interactions between the particles are all balanced in the bulk, but because the particles on the surface are not completely surrounded by atoms or molecules, they still have residual attractive attractions or unbalanced forces.
3. Mole fraction:
Raoult's law provides the most comprehensive justification for how mole fraction affects vapour pressure.
Raoult's law states that "the partial vapour pressure of each volatile component in a solution of volatile liquids is directly proportional to its mole fraction present in the solution."
Raoult's law can be described mathematically as
PSolution = Vapour pressure of the solution
PSolvento = Vapour pressure of the pure solvent
𝜒Solvent = Mole fraction of the solvent
4. Nature of the liquid:
The characteristics of the liquid are one of the most significant variables affecting vapour pressure. Intermolecular forces can be used to illustrate how the liquid's properties affect the system. The vapour pressure rises in proportion to the strength of intermolecular interactions. The liquid's vapour pressure increases as intermolecular forces become less significant.
At a specific temperature, glycerol, for instance, has a lower vapour pressure than isopentane. The stronger intermolecular forces of hydrogen bonding in glycerol are the reason for this.
Cis and trans isomers of 2-butene have boiling points of 4° and 1° C respectively. Between, geometric isomers of cis and trans-2-butene, trans isomers are generally more stable and expected to have higher stability, and hence higher booking point and lower vapour pressure. But the cis isomer has a higher dipole moment leading to higher molecular interaction and hence a higher boiling point or lower vapour pressure than the trans isomer which has zero dipole moment and small molecular interactions.
5. Boiling point:
The boiling point has an important impact on the vapour pressure. The relationship between a substance's boiling point and vapour pressure is inversely proportional. This implies that if the boiling points of two liquids differ, the higher boiling point liquid will have a lower vapour pressure.
6. Molecular Mass:
Pentane, hexane, and heptane vapour pressures are examined. Pentane, hexane, and heptane all have the same intermolecular interactions, known as London dispersion forces, and only differ in the size of their carbon chains. With a larger molecular size, London forces rise (number of electrons in a molecule). In comparison to the forces between the larger heptane molecules, the London forces between the smaller pentane molecules are substantially less. As a result, heptane has the lowest vapour pressure while pentane has the highest.
Applications of Vapour Pressure:
1. The entire procedure of a pressure cooker is based on the pressure imposed by water vapours. When heat is added to the components of the pressure cooker, water vapours begin to develop. This tends to elevate the pressure on the utensil. The whistle mechanism of the pressure cooker is activated as the pressure produced by the water vapours steadily builds. Only when the heating source is withdrawn does the pressure fall because the absence of heat prohibits the production of new water vapours and causes the existing vapours to condense back to liquid form.
2. Large water bodies have low vapour pressure, which raises the surface tension of the water and prevents water molecules from associating and evaporating quickly, which is why the water in these bodies of water doesn't really evaporate readily. In fact, water molecules found in large quantities of water like lakes, ponds, and seas have a tendency to evaporate and condense simultaneously and can hold a specific amount of water.
3. The best tool for demonstrating vapour pressure is a tea kettle. The quantity of water vapours inside the kettle rises as the water starts to boil. The kettle's lid is forced upward by the pressure of the water vapours.
4. Skin crevices, which are widespread in the winter, are brought on by low vapour pressure. Wintertime air is by nature dry. The amount of water vapour in the air decreases as the vapour pressure value decreases. Skin cells' water has a vapour pressure that is substantially higher than the atmosphere's vapour pressure. Water molecules therefore frequently travel from areas of high pressure to those of low pressure. The skin can't retain water as a result, and cracks begin to develop.
5. Vapour pressure is a crucial phenomenon that is used in the drying of clothing. Clothes are typically exposed to a hot atmosphere to dry them more quickly. For instance, in the past, clothes were dried by placing them outside in the sun. The sun rays tend to raise the temperature when they hit wet clothing, which raises the vapour pressure even more. The water vapour has a tendency to migrate from a low-pressure area to an area of high pressure, or in this case, from wet clothes to the surroundings. The outcome is that the clothes are dried.
6. With the aid of the idea of vapour pressure, it is simple to explain why you are regularly forced to defrost freezers. A freezer's temperature is regulated so that the surface temperature is lower than the freezer's interior temp. This is because the surface of the freezer must be used to expel heat. In proportion to the temperature inside and on the freezer's exterior, a comparable quantity of saturated vapour pressure develops. At the surface, saturation vapour pressure is lower than it is within the freezer, where it is relatively higher.Let's say that the freezer contains an ice cube tray containing water. Water in the ice cube tray starts to evaporate, forming water vapours in the process. These water vapours typically flow from the high-pressure zone, or the centre of the freezer, to the low-pressure zone, or the freezer's surface. Due to the difference in temperature between the surface and interior of the freezer, water vapour molecules start to condense and are then deposited as frost on the freezer's borders.
Practice Problems:
Q1. Among the following, which has higher vapour pressure?
(A) Dimethyl ether
(B) Ethyl alcohol
(C) Both have the same vapour pressure
(D) None of the above
Answer: (A)
Solution: Alcohols contain a functional component called hydroxyl that is extremely polar. The shared pair of electrons of the -OH bond is attracted to the -OH group's oxygen as a result. As a result, the partial charges of the hydrogen and oxygen in the -OH group are opposite.
Positive hydrogen from one molecule interacts with negative oxygen from another. The term for this kind of interaction is hydrogen bonding. As a result, many alcohol molecules are strongly connected.
Therefore, a large amount of energy is required to break this bond. Alcohol has higher boiling points and lowers vapour pressure as a result.
Ethers don't have this kind of hydrogen bonding. Ethers boil at lower temperatures and exhibit high vapour pressure as a result of the weaker intermolecular forces of attraction.
Q2.When a liquid reaches its boiling point, it is in equilibrium with its vapour. Each phase's molecules on average have the same
(A) Kinetic energy
(B) Potential energy
(C) Intermolecular forces
(D) Total energy
Answer: (A)
Solution: In accordance with the kinetic theory of gases the average kinetic energy of gas molecules is directly proportional to the system's absolute temperature
Average kinetic energy K.E
Where T= systems absolute temperature
KB= Boltzmann constant
Thus, kinetic energy is proportional to temperature. The temperatures are the same because the liquid is in equilibrium with its vapour at the boiling point. As a result, molecules in two distinct phases possess the same kinetic energy as when they are at the same temperature.
Looking at the other choices, we can see that the intermolecular forces differ between the two phases, even when they are in equilibrium. Total energy and potential energy are also different.
Q3. Among the following which has the highest vapour pressure?
(A) Methyl alcohol
(B) Mercury
(C) Both have the same vapour pressure
(D) None of these
Answer: (A)
Solution: In comparison to a solvent like a methyl alcohol, mercury has an extremely low vapour pressure because the forces of attraction among its individual metal atoms are far greater than the forces that bind molecules together in alcohols, such as hydrogen bonds.
Q4. The vapour pressures of pure liquids A and B are 350 mm of Hg and 550 mm of Hg, respectively. The resulting mixtures are equal to the sum of the initial volumes when the two liquids are combined. The mixture has a 0.3 mole proportion of liquid B. In the resulting solution, the vapour pressure is
(A) 410 mm of Hg
(B) 450 mm of Hg
(C) 380 mm of Hg
(D) 290 mm of Hg
Answer: (A)
Given:
Frequently Asked Questions(FAQs):
Q1. What is the boiling point’s impact on the vapour pressure?
Answer: The vapour pressure is significantly influenced by the boiling point. A substance's vapour pressure and boiling point are inversely related. It means that if the boiling point of one liquid is greater than the boiling point of another liquid, the higher boiling point liquid will have a lower vapour pressure. For instance, ether has a higher vapour pressure than ethyl alcohol when compared because it has a lower boiling point.
Q2. Why doesn't the amount of space above the liquid affect the vapour pressure?
Answer: The chemical makeup of the liquid and the temperature affect vapour pressure. As long as there is still any liquid when equilibrium is attained, vapour pressure is independent of the liquid's surface area, the volume inside the vessel, and vessel volume. None of the parameters alters the rate of evaporation per unit of surface area, which is why these fractions have no impact.
A good illustration of this is the fact that water has a low vapour pressure due to the hydrogen bonds that connect its molecules. As long as the temperature does not vary, the vapour pressure of water remains constant regardless of its volume.
Q3. What are the differences between boiling and evaporation?
Answer:
|
Evaporation |
Boiling |
|
Evaporation happens on a liquid's surface. |
Even underneath the surface of the liquid, bubbles occur during the boiling process. |
|
It happens on its own at all temperatures. |
It only happens at a particular temperature where the vapour pressure is equal to the pressure applied to the liquid surface. |
|
It is a gradual phenomenon |
It is a quick phenomenon. |
Q4. Why is food cooked at greater elevations in a pressure cooker?
Answer: Since the air pressure at higher elevations is less than 760 mm of Hg or 76 cm of Hg pressure cookers are used there. Higher altitudes have less air pressure than those close to the sea. The water boils as a result at temperatures below 373 K.
This results in a longer cooking time for the food. Food cooks more quickly in a pressure cooker because it stops water from boiling at low temperatures.


