-
Call Now
1800-102-2727
Factors Affecting Electron Gain Enthalpy - Atomic Radius, Nuclear Charge, Electronic Configuration, Shielding effect, Practice Problems, FAQs
When I sat down to have lunch today, I could hardly eat two chapatis. My brother, who had just returned from his work and was too hungry (as expressed by him!), stuffed around 5 chapatis. My sister restricted herself to one chapati as she was on diet. I wondered sitting there, how is it that we all are humans and still have different levels of appetite?
The same is the case with atoms. They have different levels of appetite for electrons. And their appetite too, just like our hunger, depends on various factors. (just like how we eat depends on hunger levels, timing, food variety and what not!). In other words, electron gain enthalpy depends on various factors.

TABLE OF CONTENTS
- Factors Affecting Electron Gain Enthalpy
- Atomic Radius
- Nuclear Charge
- Electronic Configuration
- Shielding Effect
- Examples: How various factors determine Electron Gain Enthalpy
- Practice Problems
- Frequently Asked Questions - FAQ
Factors Affecting Electron Gain Enthalpy
Electron gain enthalpy corresponds to the energy change associated when an electron is added to an isolated neutral gaseous atom. We shall discuss in detail the various factors affecting electron gain enthalpy.
Atomic Radius
- Atomic radius is defined as the distance between the centre of the nucleus and the outermost electron of an atom.
- With the increase in atomic radii, the distance between the nucleus and the last shell which receives the incoming electrons, increases.
- Hence, the force of attraction between the nucleus and the additional electron increases. Consequently, electron gain enthalpy becomes lesser negative.
- The greater the size of an atom, the lesser the magnitude of electron gain enthalpy.

Nuclear Charge
- The nuclear charge is equal to the total charge in the nucleus corresponding to the protons present in it.
- The greater the nuclear charge, the greater will be the magnitude of electron gain enthalpy as the electron (added) will have more attraction to the nucleus. Hence, the more the nuclear charge, the more negative the electron gain enthalpy released.
Electron Gain Enthalpy ∝ Nuclear Charge
Electronic Configuration
- Electronic configuration describes the arrangement of electrons present in defined orbitals around the nucleus for an atom.
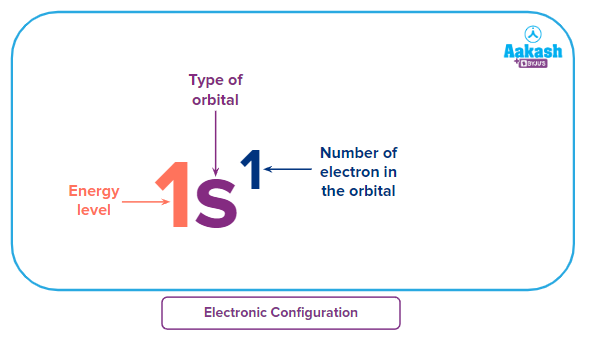
- Elements having half-filled or fully-filled electronic configurations are very stable. Hence, energy is to be supplied to add an electron to such atoms.
- Hence, their electron gain enthalpy has large positive values.
- The electron gain enthalpy becomes less negative on going from top to bottom in a group. It becomes more negative on going from left to right in a period.
Shielding Effect
- The electrons present in the inner shells of an atom act as a screen or protective layer between the nucleus and the electrons in the valence shell. As a result, the nuclear attraction on the outer electrons decreases. This is termed as shielding effect.
- The greater the shielding effect, the lesser will be the effective nuclear charge experienced by the outermost electrons.

- In such a case where there is effective shielding, if an electron is added, it would also experience lesser attraction to the nucleus. Therefore, it will have a low magnitude of electron gain enthalpy.
- Shielding effect is the property of multi-electron species and not of single-electron species.
Electron Gain Enthalpy ∞ 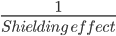
Examples: How various factors determine Electron Gain Enthalpy
Let us consider two metals Magnesium and Sodium.
- Magnesium atoms are smaller than Sodium atoms. So, the attractive force between the nucleus and the electrons in magnesium will be more than that of the attractive force between the nucleus and the electrons in sodium.
- Energy has to be supplied in both the cases and electron gain enthalpy in both the cases will be positive. But it will be less in the case of Magnesium as it is easier to add electrons in Magnesium than Sodium. So, Mg will have less positive electron gain enthalpy as compared to Na.
Let's consider two non-metals, Sulphur and Chlorine.
- The electronic configurations of Chlorine and Sulphur are 3p5 and 3p4, respectively.
- If we add one electron to these non-metals then Chlorine will accept it first. After accepting one electron it will attain noble gas configuration. And so, the stability of Chlorine will be more then.
- Hence the maximum amount of energy gets removed. Hence, the electron gain enthalpy of chlorine is more negative as compared to Sulphur. Let’s verify this by the properties of atomic radius size and nuclear charge.
- Chlorine’s atomic radiiis smaller than that of Sulphur, hence the effective nuclear charge in the case of chlorine will be greater than that of Sulphur. Hence, chlorine will have more negative electron gain enthalpy.
Practice Problems:
Q1. Which has the highest positive electron gain enthalpy?
- Chlorine
- Fluorine
- Neon
- Hydrogen
Answer: Neon being an inert gas, has a stable electronic configuration (octet complete). So, accepting an electron makes it highly unstable and hence has the highest positive E.G.E.
Q2. Elements with high negative values of electron gain enthalpy are:
- Good oxidising agents
- Good reducing agents
- Both A) & B)
- None
Answer: High negative values of E.G.E means on accepting electrons, these elements are highly stabilised. So they readily accept electrons. So they are good oxidising agents.
Q3. Why is the electron gain enthalpy of Be and Mg always positive?
Answer: The electron gain enthalpy of Be and Mg are positive because their last subshell is completely filled. Therefore, to add extra electron energy is provided.
Q4. An element has the electron gain enthalpy +48 kJ mol-1. What will be its apt electronic configuration?
- 1s22s22p1
- 1s22s22p5
- 1s22s22p3
- 1s22s22p4
Answer: The electron gain enthalpy of stable atoms will be positive. We see that option C has a half-filled electronic configuration. Hence, it will have a positive electron gain enthalpy.
Frequently Asked Questions - FAQ
Question 1. Is the shielding effect the same as the screening effect?
Answer: The shielding effect is also known as the screening effect. It refers to the reduction of nuclear charge faced by outer electrons due to guarding of the charge by inner electrons.
Question 2. How to find electron gain enthalpy, if electron affinity is given?
Answer: We can use the formula mentioned below to find the same:
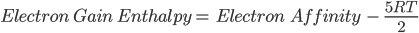
Where R = Universal Gas Constant
T = Temperature.
Question 3. What is the correct order of shielding effect of the various orbitals?
Answer: Since the s-orbital is closest to the nucleus, it has the greatest shielding effect followed by p, d and f -orbitals. Order for shielding effect is s > p > d > f
Question 4. Which element has the least positive and which element has the highest negative electron gain enthalpy?
Answer: Helium has the least positive electron gain enthalpy owing to its smaller size and hence in comparison to other noble gases, it has a higher tendency to easily accept an electron.
Chlorine has the highest negative electron gain enthalpy value owing to its larger size and highest tendency to accept an electron.
Related Topics
|
Successive Electron Gain Enthalpy |
Oxygen |
|
Electron Affinity and Electron Gain Enthalpy |
Metallic and Non-metallic Character |
|
Electron Gain Enthalpy |
Successive Electron gain Enthalpy |
|
Ionisation Enthalpy |
Electronegativity |


