-
Call Now
1800-102-2727
Electrochemical Cells: Definition of Electrochemical Cell, Types of Electrochemical Cell, Difference between Galvanic and Electrolytic Cell, Practice Problems, FAQs:
Imagine there’s a power cut at your place and you have to complete an assignment.
You started searching for a torch but unfortunately, it is not working.
The reason is pretty simple, a battery of the torch is dead.
Have you ever wondered how these small pieces of batteries are generating electricity for their respective devices?

Well, these batteries are made on the principle of electrochemical cells. Let’s understand the concept of electrochemical cell.
Table content
- Definition of electrochemical cell
- Galvanic cell or voltaic cell
- Redox reaction in Zn-Cu galvanic cell:
- Redox reaction between Cu - Ag Galvanic cell:
- Salt bridge
- Importance of salt bridge in Galvanic cell
- Criteria for selecting salt bridge
- Electrolytic cell
- Difference between Galvanic cell and electrolytic cell
- Practice problems
- Frequently asked questions(FAQs)
Definition of electrochemical cell
An electrochemical cell is a device that may either create electrical energy from chemical processes taking place inside it or use an external electrical energy source to facilitate chemical reactions inside it. These devices allow the conversion of chemical energy to electrical energy or vice versa.
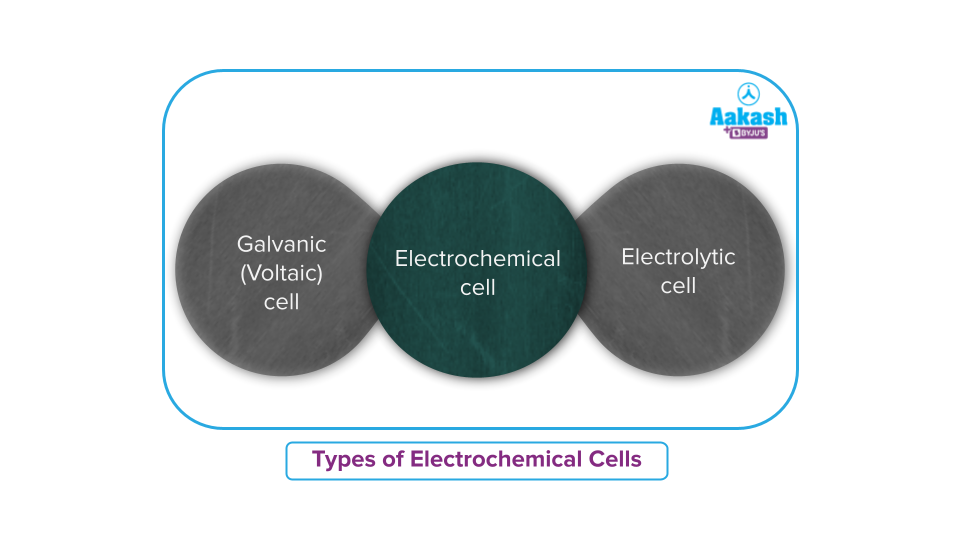
Types of Electrochemical cell
Electrochemical cells are classified into two categories:
- Galvanic or voltaic cell
- Electrolytic cell
Electrochemical cell that generates an electric current from redox chemical reactions that occur within them is called as electrical cell or galvanic cell or voltaic cell. A conventional 1.5-volt cell, which is used to power numerous electrical items such as TV remotes and clocks, is an example of Galvanic cells or Voltaic cells.
On the other hand, electrolytic cells are ones that experience chemical reactions when an electric current is passed through them.
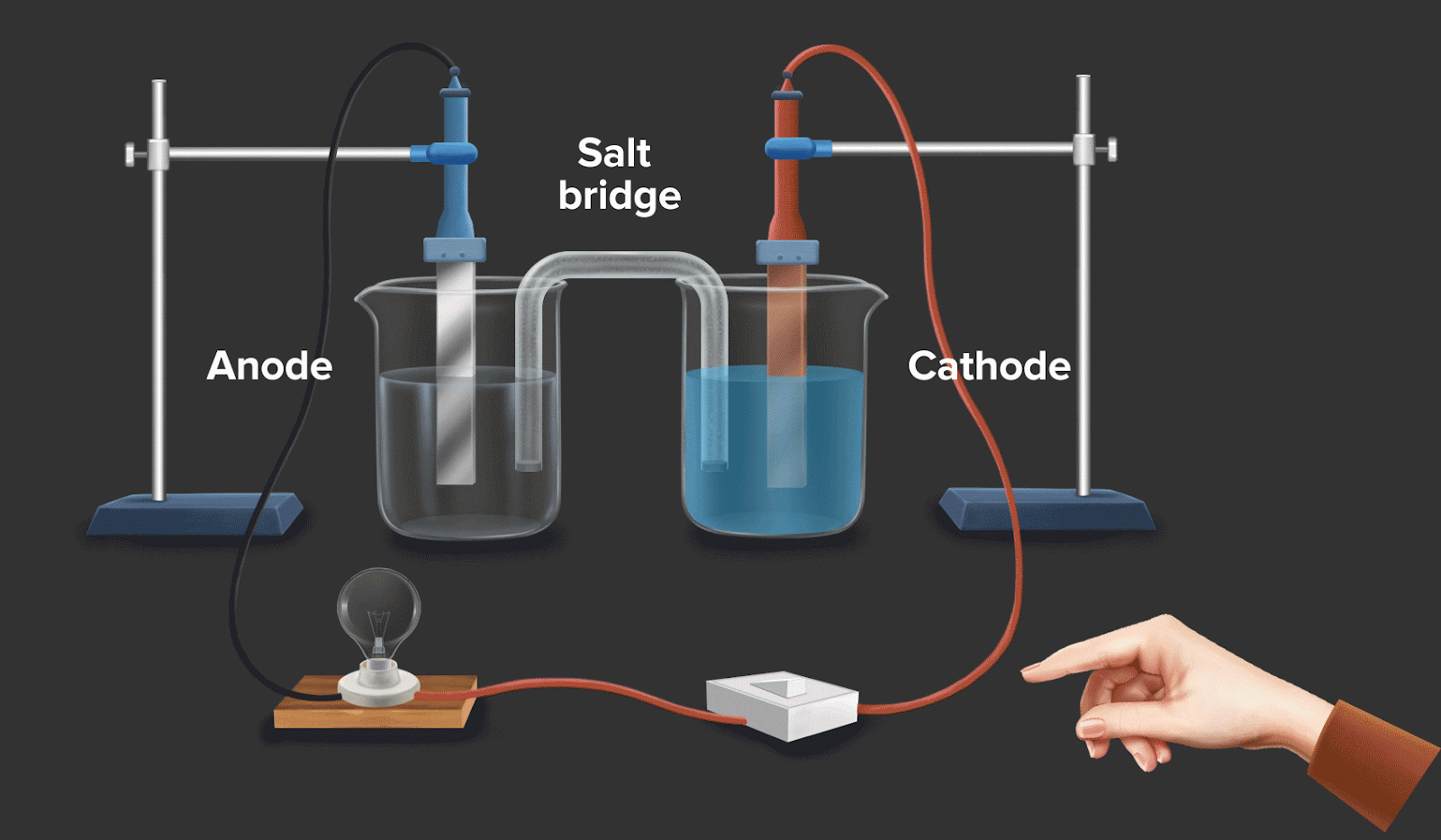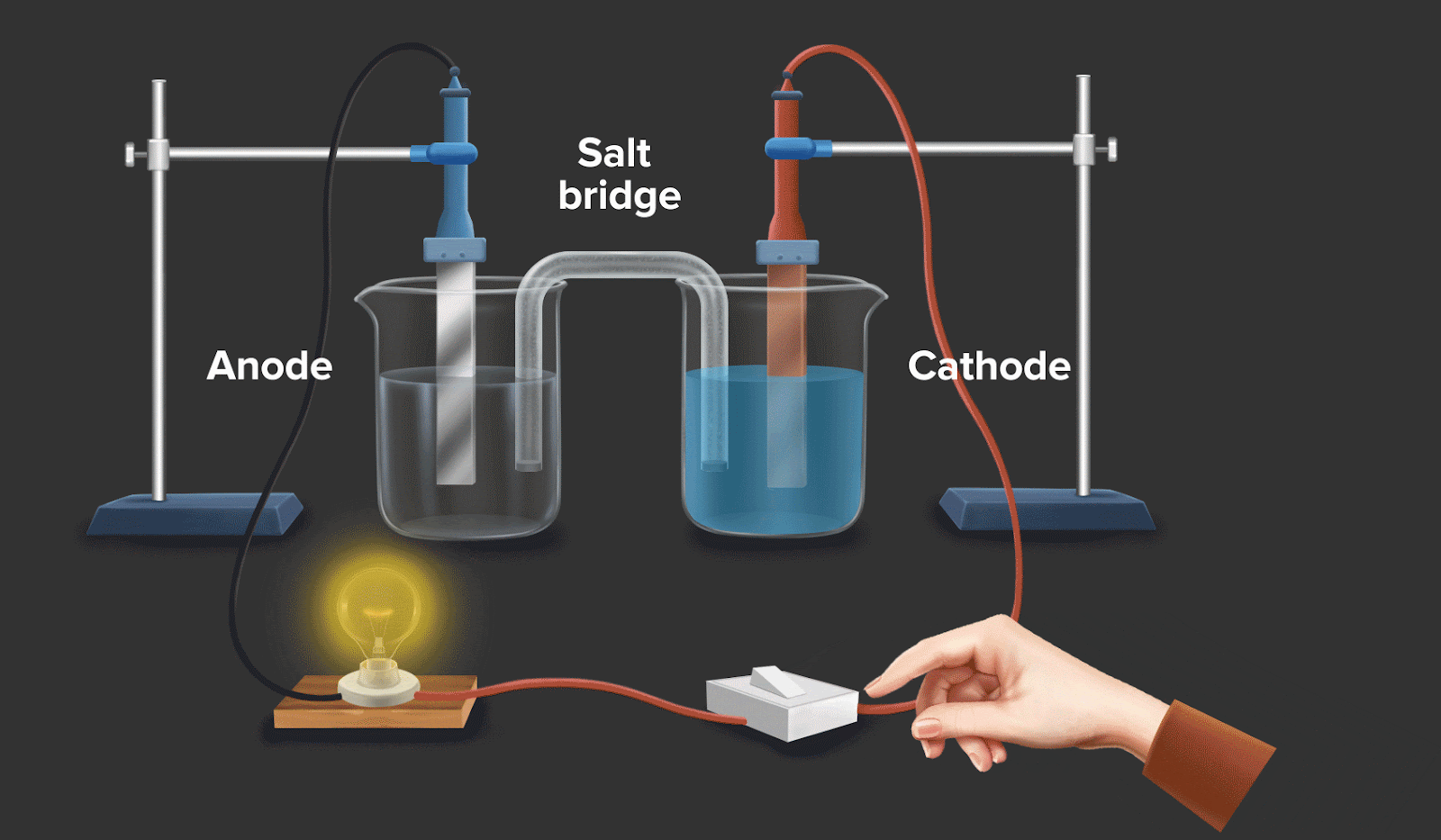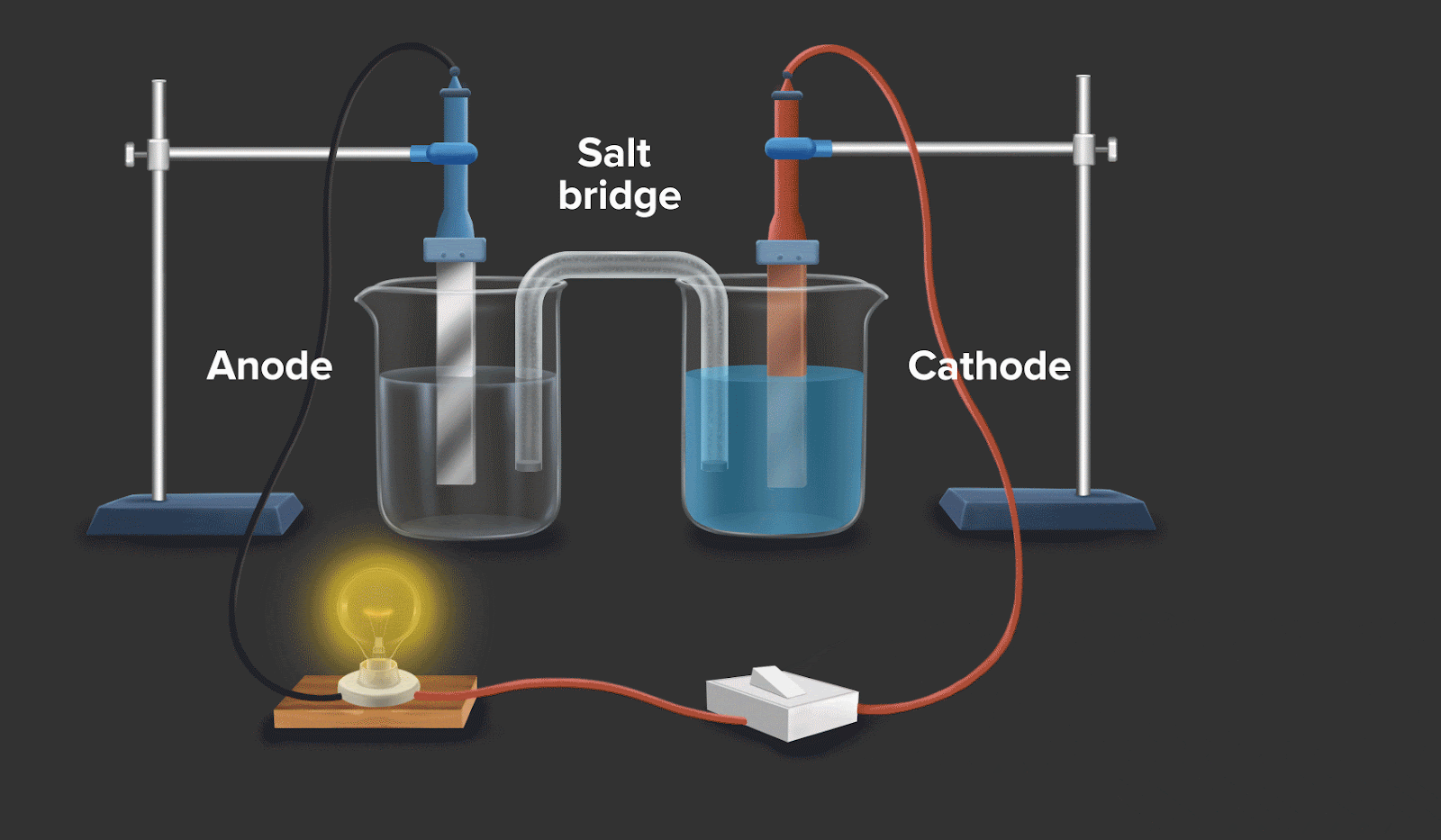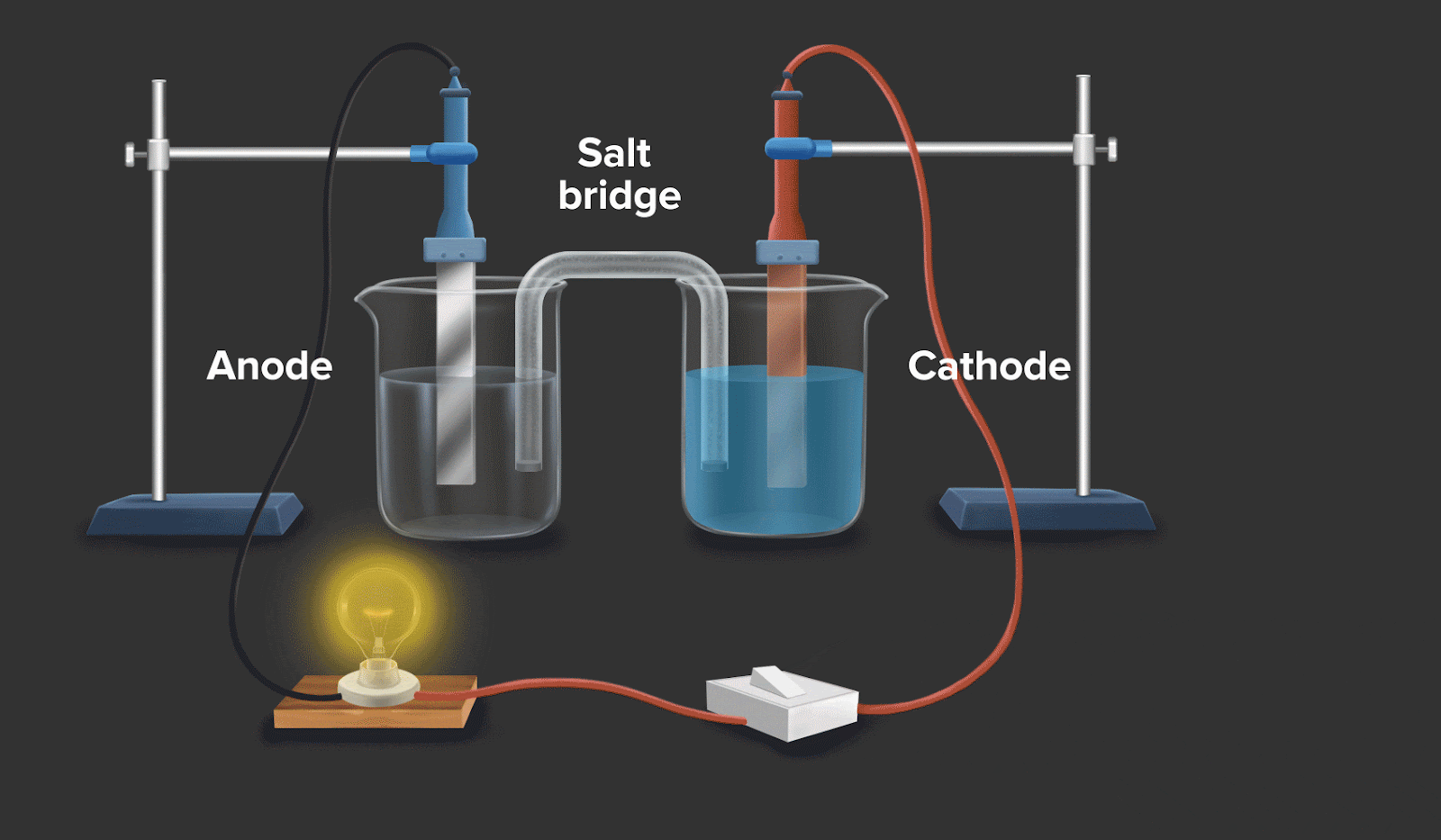
Galvanic cell or voltaic cell
A galvanic cell is an electrochemical cell which uses a spontaneous chemical reaction to produce electrical energy. Example: The batteries in the automobile have the same fundamental mechanism.
Consider a circuit as shown in galvanic cell. In the left container, oxidation takes place by convention, and in the right container, reduction takes place. If the overall reaction in this convention is spontaneous, then it can be said that it facilitates the flow of electrons and electric current.
![]()
-
Redox reaction in Zn-Cu galvanic cell:
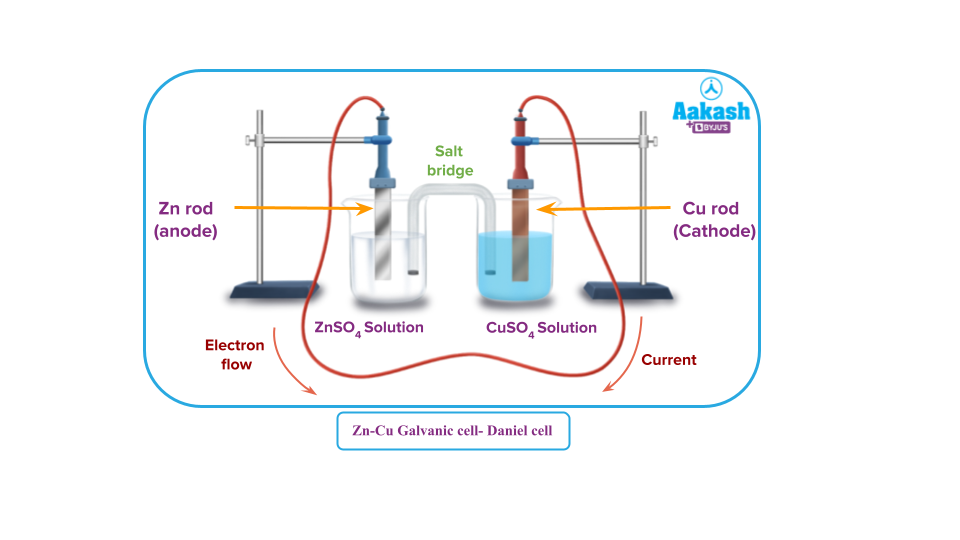
The reaction is
reaction may be split into two half reactions as under:
The first reaction is called the oxidation half reaction and the second is called the reduction half reaction. The reaction obtained by adding the two half reactions is called the overall reaction.
Thus, if a redox reaction is allowed to take place in such a way that oxidation half reaction takes place in one beaker and the reduction half reaction in another beaker, the electrons given out by the former will be
taken by latter and a current will flow. The two portions of the cell are called half-cells or redox couples. The cell is set up as follows:
A zinc rod is inserted into a beaker of zinc sulphate solution. A copper rod is placed in copper sulphate solution taken in another beaker. The two solutions are linked together by a salt bridge, while the two rods are connected by a wire.
2. Redox reaction between Cu - Ag Galvanic cell:
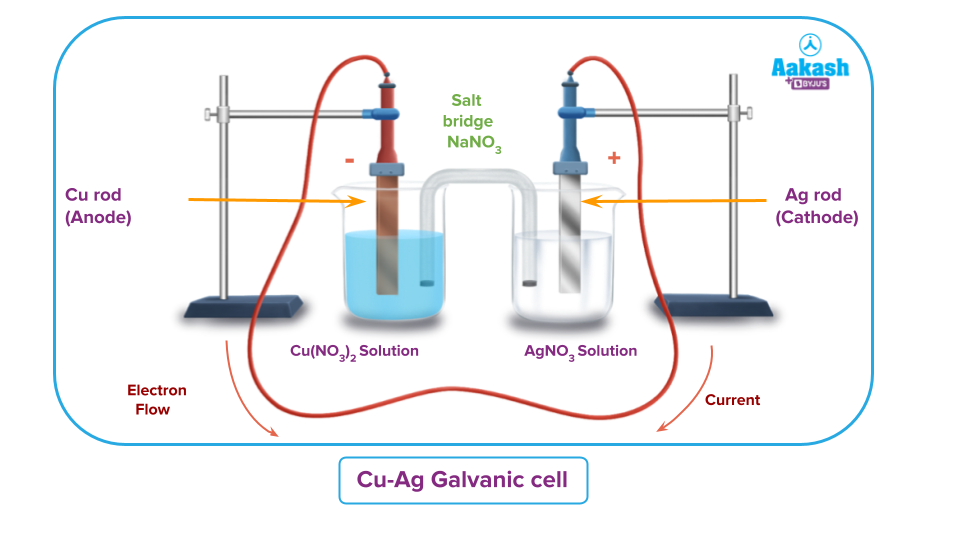
The reaction is:
or it may be written as :
The two half reactions will be:
(Oxidation half reaction)
(Reduction half reaction)
The copper electrode serves as the anode or negative pole because oxidation occurs there. The silver electrode acts as the cathode or positive pole because reduction occurs there. Silver to copper is the traditional direction for current to flow.
In a similar manner, based upon any redox reaction, a galvanic cell can be constructed.
Salt bridge
A salt bridge is an U-shaped tube containing concentrated solution of an inert electrolyte like KCI, KNO3, K2SO4, etc. or solidified solution of such an electrolyte in agar-agar and gelatine.

Importance of salt bridge in Galvanic cell
- If the electrodes are not internally connected, the cell initially produces electrical energy, but after some time the electrodes become polarised, which causes an accumulation of ions with opposing charges near the electrodes. and the electron flow ceases or the cell stops functioning.
- For continuous electrical energy generation, the electrodes must be internally connected, which is done by the salt bridge.
- Let’s consider an electrolyte used in salt bridge that is KCl: Cations (K+) migrate towards the cathode and anions (Cl-) migrate towards the anode and polarise some of the oppositely charged ions in the solution towards themselves. Thus, the cell keeps working and does not stop.
- There will eventually be no electricity flow if there is no salt bridge because the anode half-cell (left beaker) will have an excess positive charge and the cathode half-cell will have an excess negative charge (right beaker).
- After the introduction of a salt bridge, electroneutrality is maintained as the counter ions are provided by the salt bridge and the flow of charge takes place.
Criteria for selecting salt bridge
- Should consist of cations and anions having the same mobility.
- the ions in the solution shouldn't react with.
- Should not get oxidized/reduced at the electrodes.
Electrolytic cell
An electrolytic cell can be defined as an electrochemical device that uses electrical energy to facilitate a non-spontaneous redox reaction (reverse of a spontaneous reaction). If we put a battery in the shown circuit, then the reaction takes place in direction, i.e., the total redox reaction is now a non-spontaneous
reaction being driven by electrical energy.
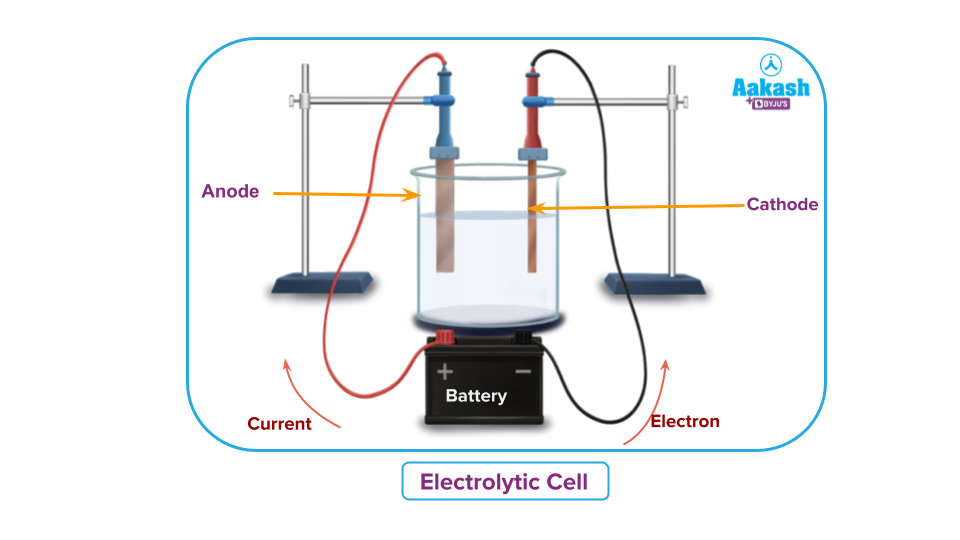
With the use of an electrolytic cell, molten sodium chloride (NaCl) can be electrolyzed. Two inert electrodes are immersed in molten sodium chloride (which contains dissociated Na+ cations and Cl- anions) in this experiment. The cathode becomes rich in electrons and generates a negative charge when an electric current is supplied across the circuit. The negatively charged cathode is now attracted to the positively charged sodium cations. At the cathode, this leads in the formation of metallic sodium.
The chlorine atoms are attracted to the positively charged cathode. As a result, the anode generates chlorine gas (Cl2).
Anode:
Cathode:
Overall reaction:
Hence, In an electrolytic cell, molten sodium chloride can be electrolyzed to produce metallic sodium and chlorine gas as byproducts.
Application of electrolytic cell
- The primary function of electrolytic cells is the production of oxygen and hydrogen gas from water.
- They can also be used to recover aluminum from bauxite.
- Electroplating, which is the process of creating a thin protective layer of a specific metal on the surface of another metal, is another notable application of electrolytic cells.
- Electrolytic cells are used in the electrorefinement of numerous non-ferrous metals.
- Electrochemical cells like these are also employed in electrowinning.
- It should be mentioned that electrolytic cells are virtually usually used in the industrial manufacture of high-purity copper, high-purity zinc, and high-purity aluminum.
Difference between Galvanic cell and electrolytic cell
|
Galvanic Cell |
Electrolytic cell |
|
|
|
|
|
|
|
|
|
|
Practice problems
Q1. Which of the following is not an electrochemical cell?
A. Galvanic cell
B. Voltaic cell
C. Electrolytic cell
D. None of the above
Answer: (D)
Solution: electrochemical cell is a device for the interconversion of chemical energy & electrical energy. Galvanic cell or voltaic cell, and electrolytic cells are different types of electrochemical cell.
Q2. Spontaneous reaction can be observed in which of the following cell?
A. Galvanic cell
B. Electrolytic cell
C. Both A & B
D. None of the above
Answer: (A)
Solution: The redox reactions that occur in galvanic cells are entirely spontaneous.
Q3. Choose the correct statement
A. The redox reactions that occur electrolytic cells are entirely spontaneous.
B. In electrolytic cell, The species that undergoes oxidation is where the electrons come from.
C. In electrolytic cell, chemical energy is converted to electrical energy.
D. A positively charged anode and a negatively charged cathode are used in electrolytic cell.
Answer: (D)
Solution: In electrolytic cell redox reactions are non- spontaneous. In these cells, Electrons are emitted from a source outside of the body (such as a battery), and here electrical energy is converted to chemical energy.
Q4. Which of the following is not a criterion of selecting a salt bridge?
A. Should consist of cations and anions having the same mobility.
B. should not interact with the solution's ions.
C. Should be able to resist the flow of electric current
D. Should not get oxidized/reduced at the electrodes.
Answer:(C)
Solution: A salt bridge is an U-shaped tube containing concentrated solution of an inert electrolyte like KCI, KNO3, K2SO4, etc . or a gelatin- and agar-agar-solidified solution of such an electrolyte.
The important criteria for selecting a salt bridge are
- Should consist of cations and anions having the same mobility.
- should not interact with the solution's ions.
- Should not get oxidized/reduced at the electrodes.
Frequently asked questions(FAQs)
Q1. Can we recharge an galvanic cell?
Answer: By providing an electrical potential in the opposite direction, galvanic cells can be recharged. During the charging process, a rechargeable battery briefly switches from a galvanic cell to an electrolytic cell. Galvanic cells and batteries both use the same fundamental concepts in their design.
Q2. The mobile devices that we use are electrolytic cells or electrochemical cells?
Answer: It is a galvanic cell function while the battery is in operation in the device (using the redox energy to produce electricity). It is an electrolytic cell function while the battery is charging (using outside electricity to reverse the completed redox reaction)
Q3. Is electrolytic cell endothermic or exothermic?
Answer: The chemical process in an electrolytic cell is endothermic. The use of a power source is required because the reaction is not spontaneous. A device that stores energy is called an electrochemical cell. When the cell is allowed to run in a galvanic mode, this energy can be released.
Q.4 What are the two metals that electrochemical cells frequently use?
Answer: Copper and zinc metals can be combined with solutions of their sulphates to create a simple electrochemical cell. Electrons can go from the zinc to the copper during the reaction through an electrically conductive channel as a useful electric current.


